Case Report
Abdominal Cocoon: a rare case of small bowel obstruction, a case report
Capullo abdominal: un caso raro de obstrucción del intestino delgado, informe de un caso
Abdominal Cocoon: a rare case of small bowel obstruction, a case report
Iberoamerican Journal of Medicine, vol. 6, núm. 4, pp. 126-131, 2024
Hospital San Pedro
Recepción: 06 Julio 2024
Corregido: 12 Agosto 2024
Aprobación: 22 Agosto 2024
Abstract: Sclerosing encapsulating peritonitis (SEP), or abdominal cocoon syndrome (ACS), is an uncommon disorder marked by a thick fibrotic membrane that completely or partially envelops the small bowel. For surgeons, preoperative diagnosis is difficult and challenging. The majority of documented, diagnosis were made during a laparotomy. We present a case of SEP that was diagnosed preoperatively. The patient had a complete small bowel obstruction that necessitated urgent surgery. We review the literature and discuss the causes, clinical manifestation, and management of the condition.
Keywords: Abdominal cocoon , Bowel obstruction , Conservative management , Adhesiolysis.
Resumen: La peritonitis esclerosante encapsulante (PES) o síndrome del capullo abdominal (SCA) es un trastorno poco frecuente caracterizado por una membrana fibrótica gruesa que envuelve total o parcialmente el intestino delgado. Para los cirujanos, el diagnóstico preoperatorio es difícil y desafiante. La mayoría de los diagnósticos documentados se realizaron durante una laparotomía. Presentamos un caso de PES que se diagnosticó preoperatoriamente. El paciente tenía una obstrucción completa del intestino delgado que requirió cirugía urgente. Revisamos la literatura y discutimos las causas, la manifestación clínica y el manejo de la afección.
Palabras clave: Capullo abdominal , Obstrucción intestinal , Manejo conservador , Adhesiólisis.
INTRODUCTION
Sclerosing encapsulating peritonitis (SEP) commonly known as abdominal cocoon syndrome (ACS) is considered one of the rare causes of intestinal obstruction. A chronic inflammatory disease results from a recurrent low-grade or subclinical peritonitis without specific abdominal signs. It eventually progresses to sclerosis and membrane formation with subsequent cocooning. SEP may be primary or secondary to conditions causing inflammation of the peritoneum and fibroblastic proliferation [1]. Small bowel obstruction occasionally can be caused by an abdominal cocoon, which is often only discovered after laparotomy. SEP could be secondary to a number of diseases or primary of unknown etiology. We report a 36-year-old woman who presented with clinical and radiological evidence of complete small bowel obstruction. A CT scan of the abdomen suspected SEP, which was confirmed by surgical exploration.
CASE REPORT
A 36-year-old woman from Bangladesh came to A&E because of central abdominal pain for several hours. She was also suffering from nausea and vomiting but was not constipated as she had gone to the toilet the day before. She had never had surgery and had no known previous medical problems. She stated that she did not have a persistent cough and had not been in contact with anyone who had tuberculosis. On examination, she looked ill and dehydrated. Her blood pressure was 140/86 mm of mercury, her pulse was 94 beats per minute and her oxygen saturation was 99% on room air. Her abdomen was noticeably tender in the periumbilical area and slightly distended. Bowel sounds were slow. A leukocytosis of 17.6x103 was the only abnormality noted on blood tests. A chest radiograph showed no free air under the diaphragm, abdominal radiograph showed dilated loops of small bowel without multiple fluid levels [Figure 1].
Several dilated small bowel loops with a maximum diameter of 3.2 cm were visible on the abdominal CT scan. The transition zone is located in the center of the ileum. Adjacent to the dilated loops of small bowel were strands of mesenteric fat. The bowel appeared to be enveloped by a thick membrane with moderate peritoneal thickening. The bowel wall enhancement in both the arterial and portal venous phases was normal. There was no free or localized intra-abdominal fluid or pneumoperitoneum, however there were several enlarged mesenteric and retroperitoneal lymph nodes. The CT scan suggested an adhesive small intestinal obstruction, most likely because of ACS [Figure2].
Following initial fluid resuscitation, an urgent exploratory laparotomy was performed on the patient. During surgery, a fibrous membrane was discovered to surround the entire small bowel, apart from 100 cm of the proximal jejunum. The membrane was divided and the small bowel adhesions were carefully released using a combination of blunt and sharp dissection without resection of the bowel (Figure 3).
Histopathological analysis of the cocoon wall revealed non-caseating epithelioid cell granulomas without Langhans-type giant cells, and the test for acid-fast bacilli was negative.
Subsequent examinations and investigation of the patient by the chest physician ruled out tuberculosis. The patient was discharged on the fifth postoperative day and was followed up in the outpatient clinic for more than twelve months and she remained well and healthy.
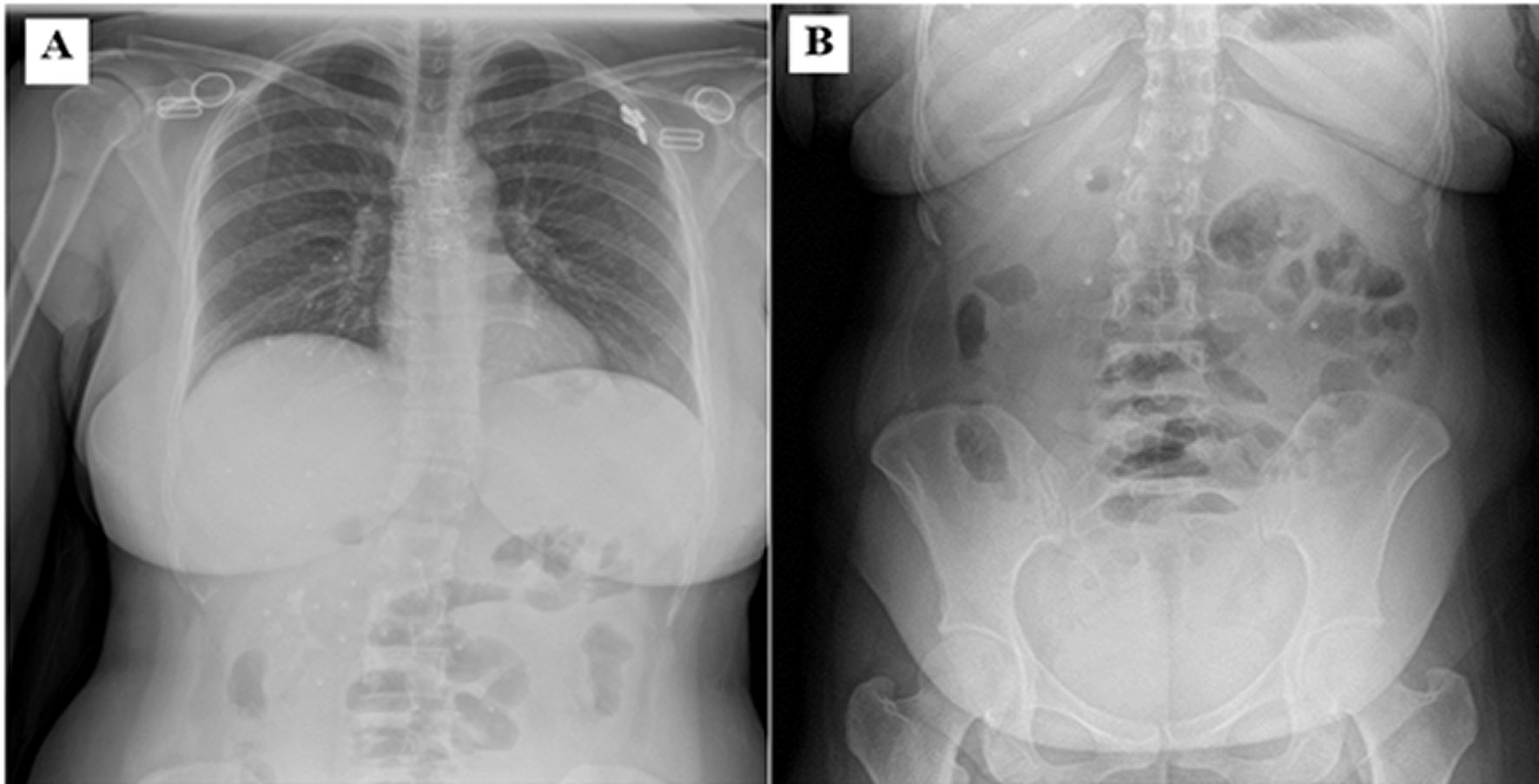
Figure 1
(A) The X-ray of the chest reveals clear lungs fields and no free air beneath the diaphragm and (B) abdominal radiograph showed dilated loops of small bowel without multiple fluid levels
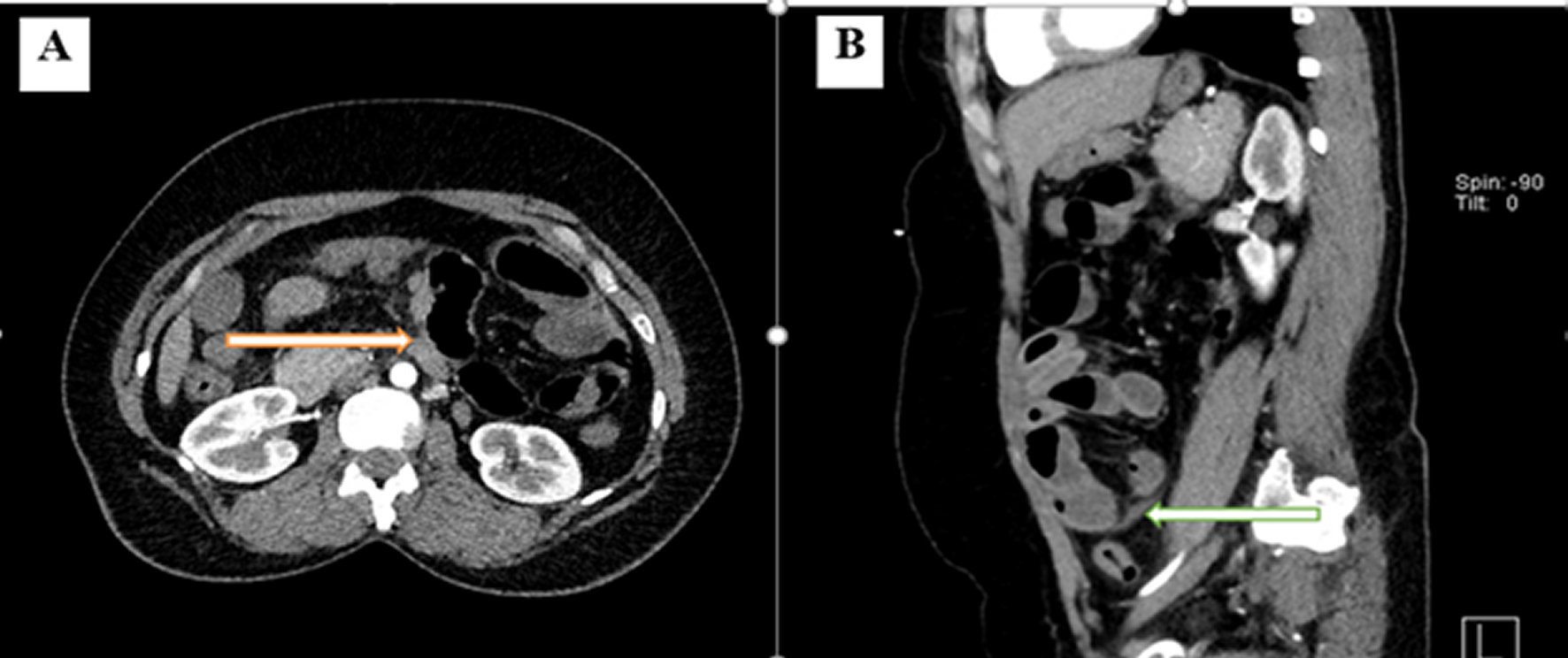
Figure 2
(A) An axial cut of the abdominal CT scan reveals the small bowel's dilated loops, which are surrounded by a thick membrane (The arrow) and B a sagittal cut of the abdominal CT scan reveals multiple dilated small bowel loops surrounded with a thick fibrous membrane (The arrow)
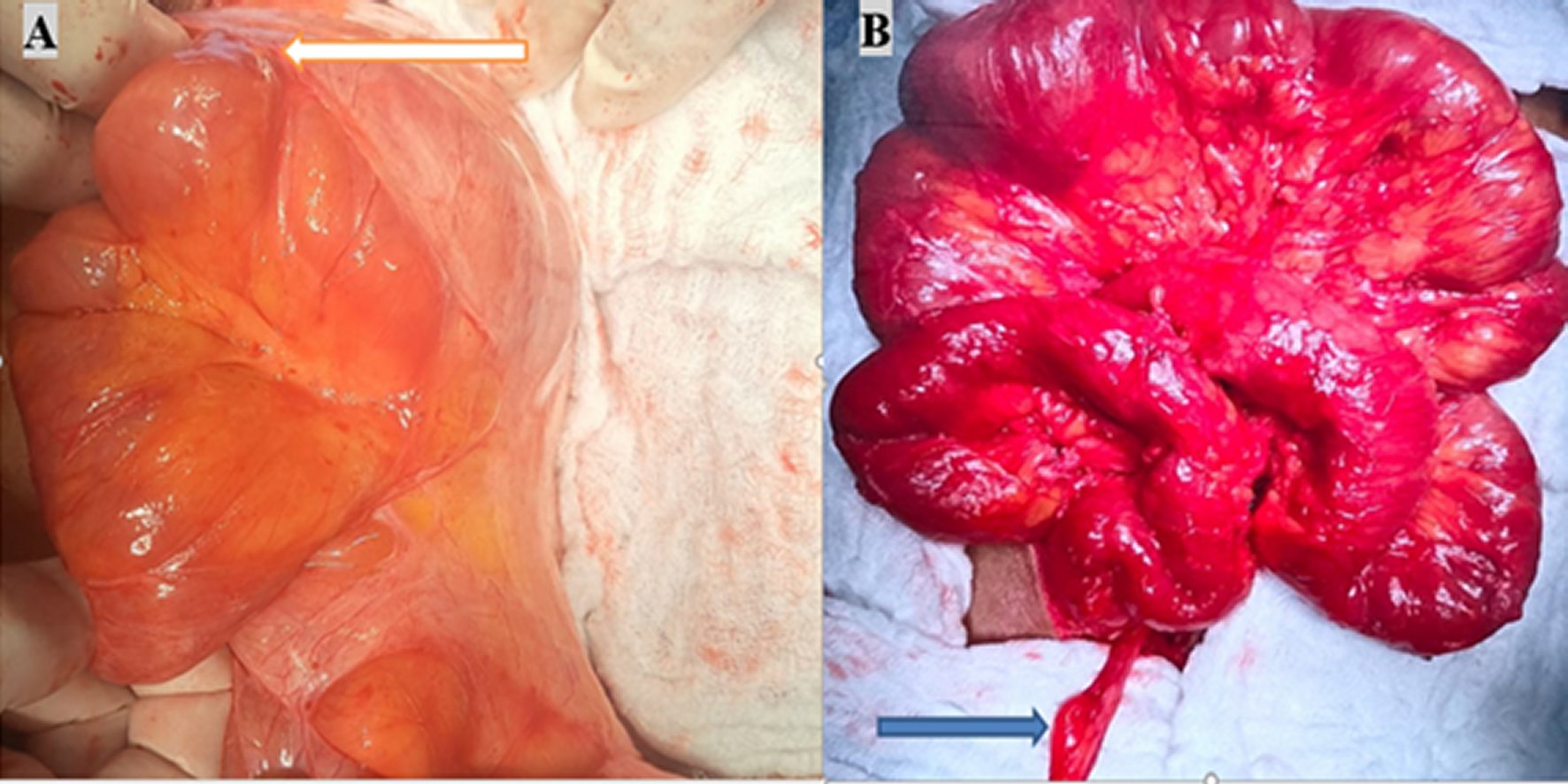
Figure 3
(A) An intraoperative picture displaying small intestinal loops covered in fibrous membranes. The membrane is shown by (the arrow) and (B) showing the Small bowel after the adhesiolysis. The arrow is pointing to the appendix
DISCUSSION
The disorder known as SEP or (ACS) is defined by the small intestine being entirely or partially encased in a fibro collagenous sac that resembles a cocoon. [2] Foo et al. coined the term "abdominal cocoon" in 1978, but the disease was first diagnosed as "peritonitis chronica fibrosa incapsulata" by Ovchinnikov in 1907 [3] and was later referred to as "sclerosing encapsulating peritonitis" by Deeb et al. in 1998 [4]. The terms "sclerosis" and "encapsulation" refer to the progressive formation of layers of dense collagenous tissue, "encapsulation" describes the sheath of new fibrous tissue that covers the small intestine and restricts its motility, and "peritonitis" denotes a persistent inflammatory process and the presence of a mononuclear inflammatory infiltrate within the new fibrous tissue [1, 5].
SEP is a chronic clinical syndrome with an insidious onset that manifests as chronic undernourishment accompanied by sporadic, acute, or subacute gastrointestinal obstruction symptoms [6].
The classification of a disease as primary or secondary depends on whether it is idiopathic or has a known cause. A genetic predisposition may also be a contributing component in secondary SEP, which is also known as multifactorial [7].
Based on the clinical presentation, SEP is classified into four stages [8]:
· Pre-encapsulating peritoneal sclerosis stage: Mild ascites, no inflammation, and no symptoms.
· Inflammatory stage: The patient's symptoms, which include intestinal swelling and partial encapsulation of the bowel, nausea and diarrhoea. There is an exudation of fibrin and mild inflammation.
· Encapsulation: Signs of intestinal blockage brought on by the encapsulation-causing fibrous cocoon. It may be linked to inflammation, ranging from mild to severe.
· Chronic stage of ileus: The thickening of the encapsulating fibrous cocoon causes patients to experience complete bowel obstruction. At this point, there is not much, if any, inflammation.
Idiopathic SEP’s pathogenesis is still unknown. Although case reports of adults from temperate zones have been documented in the literature. SEP has been traditionally reported in young, adolescent women from tropical and subtropical countries. In secondary SEP, a local or systemic factor can be identified as the trigger for peritoneal inflammation. Infection as Tuberculosis, non-tuberculosis, mycobacteria, bacterial peritonitis, cytomegalovirus, fungi, parasites, mechanical or chemical intraperitoneal irritants: Ventriculoperitoneal shunt Peritoneovenous shunt, Intraperitoneal iodine, Abdominal trauma, Intra-abdominal surgery, Foreign body, Talcum powder, Asbestos, Silica, Cirrhosis, Organ transplant, Endometriosis, Gynaecological neoplasms, Dermoid cyst rupture, Systemic rheumatologically and inflammatory disorders, including Sarcoidosis and Systemic Lupus Erythematosus [9].
The most common secondary SEP is tuberculosis. In India and other countries where tuberculosis is more frequent, the disease is more prevalent. It has also been reported as a major side effect of continuous ambulatory peritoneal dialysis and in connection with some drugs, most notably the beta-adrenergic blocker Practolol [10, 11, 12, 13].
It is very challenging to accurately diagnose SEP preoperatively, and a high index of clinical suspicion is needed. Patients with recurrent subacute small intestinal obstruction should be suspected of having SEP, particularly if they have a history of tuberculosis, continuous ambulatory peritoneal dialysis, or long-term use of B-blockers.
Preoperative diagnosis of SEP is now feasible due to the growing use of computerized tomography in the examination of patients with intestinal blockage. Previously, the diagnosis of SEP could only be made at laparotomy. In order to prevent surprises during surgery and to enable proper treatment planning, preoperative diagnosis is essential. According to some research, computed tomography (CT) is the gold standard for diagnosing SEP, and it can now be used to definitively diagnose SEP [14]. When ESP is detected on CT, the entire small bowel is usually concentrated in the middle of the abdomen and covered in a layer of soft tissue with a high density. Agglutination and fixation of bowel loops, wall thickening, ascites, localized fluid collections, peritoneal thickening, and enhancement, peritoneal or mural calcifications, and reactive adenopathy are additional CT characteristics of SEP [15, 16]. Complex loculations may indicate intra-abdominal hemorrhage, but if they contain gas, there is reason to suspect sepsis or a perforation [17]. Elevated thickening or enhancement of the colon wall suggests transmural fibrosis or persistent inflammation [18]. Although it has not been used as much for diagnosis, magnetic resonance imaging probably produces similar results. Benefits include better bowel encasement and peritoneal thickening delineation, as well as the avoidance of ionizing radiation [19].
Because case reports make up the majority of SEP -related publications to date, there is no evidence-based consensus regarding the best course of treatment for patients with SEP. Despite debate over the best time, type, and criteria for surgery, it is still the most effective treatment for SEP, although some patients may develop new adhesions and small intestinal obstructions after surgery. [9]. The most appropriate surgical procedure is to detach the membrane from the intestinal surface and excise the dense adhesions between the intestinal loops [20]. Membrane excision and adhesiolysis should be performed on all affected bowel segments if there are no other contraindications to this procedure. The risk of recurrence is quite low if the membrane can be completely removed from the bowel surface [21]. Inserting an anti-adhesive substance between the bowel loops before closing the abdomen may prevent the development of postoperative adhesive obstruction of the small bowel [22]. Numerous substances have been used to prevent postoperative adhesions, including hyaluronic acid, which acts as a mechanical barrier and promotes the dissolution of fibrin and the proliferation of mesothelial cells. Oxidized regenerated cellulose and carboxymethylcellulose are cellulose derivatives commonly used to prevent intra-abdominal adhesions. Oxidized regenerated cellulose has excellent adhesive properties and is fully biocompatible. It forms a gel-like membrane within twenty-four hours of application, which degrades after about fourteen days. Polylactide and its copolymers are often used to prevent adhesions, mainly due to their excellent biodegradability, biocompatibility and good mechanical properties [23]. However, these mechanical barrier agents should not replace careful surgical technique, which includes gentle handling of the tissue, avoidance of excessive bleeding and excessive use of diathermy.
Individuals who exhibit recurrent subacute small intestinal obstruction and who respond to conservative treatment may benefit from pharmacological medications that include azathioprine, colchicine, tamoxifen, and steroids [9]. Corticosteroids are hypothesized to prevent inflammation within the peritoneal membrane, which in turn prevents collagen formation and maturation [24]. Tamoxifen is a selective modulator of the estrogen receptor that prevents fibroblasts from producing the probiotic cytokine transforming growth factor beta. Colchicine has an anti-inflammatory effect by preventing the transforming growth factor beta's mRNA expression [1] Apart from a few documented examples; there is not enough evidence in the literature to support the use of such drugs in patients with SEP.
CONCLUSIONS
SEP is a very uncommon disorder that can develop as a subsequent disease or as a primary cause of unknown etiology. SEP can be classified as primary (idiopathic) or secondary, where a trigger for the inflammatory process can be identified. SEP is most commonly secondary to tuberculosis and usually present with complete obstruction of the small intestine that does not respond to conservative treatment. It is considered one of the rare causes of intestinal obstruction. Preoperative SEP diagnosis is quite challenging and necessitates a high index of suspension. Nowadays, CT can be used to diagnose SEP with certainty, as it is the gold standard for diagnosing SEP. The most successful course of treatment for small intestinal obstruction in patients who have experienced an acute and total obstruction and have not responded to conservative measures is surgery. Surgical options include membrane excision and adhesiolysis, resection and anastomosis, protective enterostomy and exploratory laparotomy. Patients who experience recurrent small intestinal obstruction may benefit from medication that blocks the formation of fibroblasts and collagen synthesis and maturation.
CONFLICT OF INTERESTS
The authors have no conflict of interest to declare. The authors declared that this study has received no financial support.
REFERENCES
1.Tannoury JN, Abboud BN. Idiopathic sclerosing encapsulating peritonitis: abdominal cocoon. World J Gastroenterol. 2012;18(17):1999-2004. doi: 10.3748/wjg.v18.i17.1999.
2.Foo KT, Ng KC, Rauff A, Foong WC, Sinniah R. Unusual small intestinal obstruction in adolescent girls: the abdominal cocoon. Br J Surg. 1978;65(6):427-30. doi: 10.1002/bjs.1800650617.
3.Lalloo S, Krishna D, Maharajh J. Case report: abdominal cocoon associated with tuberculous pelvic inflammatory disease. Br J Radiol. 2002;75(890):174-6. doi: 10.1259/bjr.75.890.750174.
4.Deeb LS, Mourad FH, El-Zein YR, Uthman SM. Abdominal cocoon in a man: preoperative diagnosis and literature review. J Clin Gastroenterol. 1998;26(2):148-50. doi: 10.1097/00004836-199803000-00013.
5.Honda K, Oda H. Pathology of encapsulating peritoneal sclerosis. Perit Dial Int. 2005;25 Suppl 4:S19-29.
6.Augustine T, Brown PW, Davies SD, Summers AM, Wilkie ME. Encapsulating peritoneal sclerosis: clinical significance and implications. Nephron Clin Pract. 2009;111(2):c149-54; discussion c154. doi: 10.1159/000191214.
7.Kawaguchi Y, Kawanishi H, Mujais S, Topley N, Oreopoulos DG. Encapsulating peritoneal sclerosis: definition, etiology, diagnosis, and treatment. International Society for Peritoneal Dialysis Ad Hoc Committee on Ultrafiltration Management in Peritoneal Dialysis. Perit Dial Int. 2000;20 Suppl 4:S43-55.
8.Nakamoto H. Encapsulating peritoneal sclerosis--a clinician's approach to diagnosis and medical treatment. Perit Dial Int. 2005;25 Suppl 4:S30-8.
9.Akbulut S. Accurate definition and management of idiopathic sclerosing encapsulating peritonitis. World J Gastroenterol. 2015;21(2):675-87. doi: 10.3748/wjg.v21.i2.675.
10.Eltringham WK, Espiner HJ, Windsor CW, Griffiths DA, Davies JD, Baddeley H, et al. Sclerosing peritonitis due to practolol: a report on 9 cases and their surgical management. Br J Surg. 1977;64(4):229-35. doi: 10.1002/bjs.1800640402.
11.Brown P, Baddeley H, Read AE, Davies JD, McGarry J. Sclerosing peritonitis, an unusual reaction to a beta-adrenergic-blocking drug (practolol). Lancet. 1974;2(7895):1477-81. doi: 10.1016/s0140-6736(74)90218-9.
12.Windsor WO, Durrein F, Dyer NH. Fibrinous peritonitis: a complication of practolol therapy. Br Med J. 1975;2(5962):68. doi: 10.1136/bmj.2.5962.68.
13.Mohamed A, Kuhit M, Emran F, Riaz M. Abdominal Cocoon, a Rare Complication of Continuous Ambulatory Dialysis Catheter. The Internet Journal of Surgery. 2008;21(2).
14.Tombak MC, Apaydin FD, Colak T, Duce MN, Balci Y, Yazici M, et al. An unusual cause of intestinal obstruction: abdominal cocoon. AJR Am J Roentgenol. 2010;194(2):W176-8. doi: 10.2214/AJR.09.3083.
15.Wig JD, Gupta SK. Computed tomography in abdominal cocoon. J Clin Gastroenterol. 1998;27(3):259-60. doi: 10.1097/00004836-199810000-00018.
16.Krestin GP, Kacl G, Hauser M, Keusch G, Burger HR, Hoffmann R. Imaging diagnosis of sclerosing peritonitis and relation of radiologic signs to the extent of the disease. Abdom Imaging. 1995;20(5):414-20. doi: 10.1007/BF01213261.
17.Upponi S, Butler AJ, Watson CJ, Shaw AS. Encapsulating peritoneal sclerosis--correlation of radiological findings at CT with underlying pathogenesis. Clin Radiol. 2014;69(1):103-9. doi: 10.1016/j.crad.2013.09.004.
18.Ti JP, Al-Aradi A, Conlon PJ, Lee MJ, Morrin MM. Imaging features of encapsulating peritoneal sclerosis in continuous ambulatory peritoneal dialysis patients. AJR Am J Roentgenol. 2010;195(1):W50-4. doi: 10.2214/AJR.09.317.
19.Jovani M, Baticci F, Bonifacio C, Omodei PD, Malesci A. Abdominal cocoon or idiopathic encapsulating peritoneal sclerosis: magnetic resonance imaging. Dig Liver Dis. 2014;46(2):192-3. doi: 10.1016/j.dld.2013.08.136.
20.Meshikhes AW, Bojal S. A rare cause of small bowel obstruction: Abdominal cocoon. Int J Surg Case Rep. 2012;3(7):272-4. doi: 10.1016/j.ijscr.2012.03.016.
21.Thakur SK, Agrawal T. The abdominal cocoon. J Indian Med Assoc. 2012;110(3):192.
22.Rajagopal AS, Rajagopal R. Conundrum of the cocoon: report of a case and review of the literature. Dis Colon Rectum. 2003;46(8):1141-3. doi: 10.1007/s10350-004-7295-5.
23.Sirovy M, Odlozilova S, Kotek J, Zajak J, Paral J. Current options for the prevention of postoperative intra-abdominal adhesions, Asian J Surg. 2024;47(1):77-82. doi: 10.1016/j.asjsur.2023.10.001.
24.Habib SM, Betjes MG, Fieren MW, Boeschoten EW, Abrahams AC, Boer WH, et al. Management of encapsulating peritoneal sclerosis: a guideline on optimal and uniform treatment. Neth J Med. 2011;69(11):500-7.
Notas de autor
abbasmoh323@gmail.com
Información adicional
redalyc-journal-id: 6920