Review
Aristolochic Acid Nephropathy: Molecular Mechanisms, Clinical Impact and Therapeutic Challenges in a Progressive Renal Disease
Nefropatía por ácido aristolóquico: mecanismos moleculares, impacto clínico y desafíos terapéuticos en una enfermedad renal progresiva
Aristolochic Acid Nephropathy: Molecular Mechanisms, Clinical Impact and Therapeutic Challenges in a Progressive Renal Disease
Iberoamerican Journal of Medicine, vol. 7, núm. 2, pp. 53-63, 2025
Hospital San Pedro
Recepción: 13 Febrero 2025
Corregido: 07 Abril 2025
Aprobación: 14 Abril 2025
Abstract: Aristolochic acid nephropathy (AAN) is a rapidly progressive form of interstitial nephritis associated with the use of medicinal plants from the Aristolochiaceae family, which contain aristolochic acid (AA) and environmental pollutants. It was first discovered in Belgium in the 1990s and is associated with progressive renal fibrosis and a high risk of urothelial carcinoma. Development and discussion: AAN is a toxic kidney disease associated with the consumption of medicinal plants with AA or contaminated food. It is characterized by progressive interstitial nephritis, renal fibrosis and an increased risk of urothelial carcinoma. Its pathogenesis includes mitochondrial DNA damage, TP53 mutations, inflammation and fibrosis. Clinically, the disease presents as progressive renal disease in chronic forms, acute renal damage after massive ingestion or mild tubular dysfunction. Diagnosis is based on the history of exposure, clinical findings, renal biopsy and urinary biomarkers. Complications include urothelial carcinoma, severe fibrosis and end-stage renal disease. Treatment requires elimination of AA exposure, oncologic surveillance, and advanced renal support. This emphasizes the need for targeted therapies to slow disease progression and improve prognosis. AAN is a multifactorial disease with severe renal impact and high oncologic morbidity. Prevention, early diagnosis and multidisciplinary management are crucial to improve the prognosis and quality of life of patients.
Keywords: Aristolochic acid, Aristolochic acid nephropathy, Progressive renal disease, Renal fibrosis, Urothelial carcinoma.
Resumen: La nefropatía por ácido aristolóquico (NAA) es una forma rápidamente progresiva de nefritis intersticial asociada al uso de plantas medicinales de la familia Aristolochiaceae, que contienen ácido aristolóquico (AA) y con la presencia de contaminantes ambientales. Esta patología se descubrió por primera vez en Bélgica en la década de 1990 y se asocia a una fibrosis renal progresiva y a un alto riesgo de carcinoma urotelial. El desarrollo y la discusión de este tema se centran en la comprensión de la NAA como una enfermedad renal tóxica asociada al consumo de plantas medicinales que contienen AA o alimentos contaminados. Esta condición se caracteriza por una progresión de la nefritis intersticial, seguida de fibrosis renal y un riesgo aumentado de carcinoma urotelial. La investigación en torno a la patogénesis de la NAA revela daños en el ADN mitocondrial, mutaciones en el gen TP53, inflamación y fibrosis. En términos clínicos, la enfermedad se manifiesta como una enfermedad renal progresiva en las formas crónicas, daño renal agudo tras una ingesta masiva o disfunción tubular leve. El diagnóstico se fundamenta en los antecedentes de exposición, los hallazgos clínicos, la biopsia renal y los biomarcadores urinarios. Las complicaciones asociadas incluyen carcinoma urotelial, fibrosis grave y enfermedad renal terminal. El tratamiento requiere la eliminación de la exposición a la NAA, vigilancia oncológica y soporte renal avanzado, lo que subraya la importancia de las terapias dirigidas para ralentizar la progresión de la enfermedad y mejorar el pronóstico. La NAA es una enfermedad multifactorial con graves repercusiones renales y una elevada morbilidad oncológica. Por lo tanto, se hace evidente que la implementación de medidas preventivas, el diagnóstico temprano y un abordaje multidisciplinario son de suma importancia para mejorar el pronóstico y la calidad de vida de los pacientes afectados.
Palabras clave: Ácido aristolóquico, Nefropatía por ácido aristolóquico, Enfermedad renal progresiva, Fibrosis renal, Carcinoma urotelial.
INTRODUCTION
Aristolochic acid nephropathy (AAN), formerly known as Chinese herbal nephropathy, is a drug-induced progressive interstitial nephritis that can lead to acute kidney injury (AKI), renal fibrosis, and urothelial carcinoma of the upper urinary tract [1]. It is caused by exposure to plants containing aristolochic acid (AA), particularly those from the Aristolochiaceae family—such as Aristolochia fangchi, Aristolochia clematitis, and Aristolochia indica—either through traditional phytotherapies or through environmental contamination in food [2].
In the early 1990s, several cases of young women with rapidly progressing tubulointerstitial nephritis were documented in Belgium, linked to the consumption of diet pills containing Aristolochia fangchi, which was identified as a nephrotoxic agent [3]. In 2002, the International Agency for Research on Cancer (IARC) classified AA as a Class I human carcinogen due to its strong association with urothelial carcinoma [4]. Notably, all documented human exposures were oral, indicating gastrointestinal absorption and systemic distribution [2].
Exposure to AA has been associated with a high incidence of urothelial carcinoma, as these compounds produce specific DNA adducts that induce mutations in tumor suppressor genes such as TP53 [5]. As the use of herbal medicine is widespread in Asia, the use of Aristolochia and other plants with AA has increased the risk of AAN in these regions [6]. Several cases have been documented in different parts of the world, with a particularly high prevalence in Asian countries [7]. More than 300 cases have been reported in China and Taiwan, although the actual number is estimated to be much higher due to widespread exposure in the population [8].
Risk factors include the cumulative dose of AA taken. A study in China found that regular use of nephrotoxic drugs such as analgesics or AA products increased the risk of kidney damage (OR 2.19) [9, 10]. In addition, a cumulative dose of more than 0.5 g of aristolochic acid I (AA-I) was associated with an increased risk of chronic kidney disease (CKD) (OR 5.625) [11]. In recent years, the incidence of AAN has decreased due to strict regulations on the use of AA-containing plants, although sporadic cases are still reported, demonstrating the importance of considering this exposure in the differential diagnosis of kidney disease [12].
A subtype of AAN is endemic Balkan nephropathy, a chronic tubulointerstitial disease that slowly progresses to end-stage renal disease (ESRD) and is associated with urothelial carcinoma of the upper urinary tract (UTUC) [13]. It has been found in rural areas of southeastern Europe and exposure to AA in Aristolochia clematitis, a plant that contaminates wheat for home-baked bread, has been identified as the cause [13]. Similar cases in Belgium have been linked to the consumption of Aristolochia fangchi extracts, where a high prevalence of UTUC was also observed. An important finding was the presence of DNA aristolactam adducts in kidney and UTUC tissues, which not only serve as biomarkers of exposure but also promote specific mutations in TP53, thus contributing to carcinogenesis [13]. Historically, AA have been used in traditional medicine as antiviral, antibacterial, anti-inflammatory, antineoplastic, and weight-loss agents, but their renal toxicity became evident following outbreaks of interstitial nephritis in Europe [14]. AAN is characterized by elevated serum creatinine levels, severe anemia, and histopathological findings including a hypocellular interstitial infiltrate with advanced fibrosis. The disease rapidly progresses to end-stage renal disease, with 40-45% of patients developing CKD in less than two years [15]. Treatment is limited. Glucocorticoids have shown some effect in delaying disease progression in non-randomized studies, but most patients require renal replacement therapy, such as dialysis or kidney transplantation [15]. Given the high risk of malignancy, prophylactic bilateral nephroureterectomy and close surveillance for early-stage upper urinary tract tumors is recommended [15].
AA are found in plants of the globally distributed genera Aristolochia and Asarum from the Aristolochiaceae family [16]. Aristolochia-based herbal remedies have been known to be used for more than 2,500 years in Europe and for at least 1,500 years in China [17]. At least seven species of Aristolochia and four species of Asarum have been identified with medicinal applications. In traditional Chinese medicine, herbs such as Ma Dou Ling (Aristolochiae Fructus), Guan Mu Tong (A. manshuriensis Caulis) and Xi Xin (Asari Radix et Rhizoma) contain AA and have been associated with cases of AAN [16].
The purpose of this literature review is to consolidate the knowledge on AAN and identify the pathogenic mechanisms, risk factors and clinical manifestations. It also aims to provide an up-to-date scientific basis to improve the understanding of the disease, promote prevention and early diagnosis strategies, and emphasize the need for strict regulations on the use of products containing AA, especially in regions where traditional herbal medicines are still widely used.
METHODS
This narrative bibliographic review aims to comprehensively analyze the available scientific literature on Aristolochic Acid Nephropathy (AAN). A systematic search was conducted in PubMed, prioritizing indexed articles to ensure quality and relevance. A total of 35 publications were included, comprising bibliographic reviews, original research articles, meta-analyses, and systematic reviews (Figure 1).

Figure 1
PRISMA flow diagram showing manuscript selection
Among these, one article was from Lippincott Williams & Wilkins, while the remaining studies were classified within impact quartiles: Q1 (25 articles), Q2 (6), Q3 (2), and Q4 (1). This distribution ensures that the selected literature meets high scientific standards.
The search strategy incorporated key terms such as: "Aristolochic Acids - Balkan Nephropathy - Renal Insufficiency, Chronic - Clinical Diagnosis."
Inclusion criteria consisted of English-language publications addressing the epidemiology, etiology, clinical manifestations, and therapeutic approaches of AAN. Only full-text articles were considered. To maintain scientific rigor, studies were excluded if they: employed inadequate methodology, lacked significant clinical insights into AAN, or were not supported by current scientific evidence.
DISCUSSION
AAN is a progressive kidney disease characterized by interstitial fibrosis of the kidneys. Chan and Ham (2021) and Zhang et al., (2022) [17, 18] agree that the etiology of this disease is mainly associated with prolonged exposure to AA, compounds found in certain plants used in traditional herbal medicine and in contaminated foods [18].
Chan and Ham (2021) [17] points out that AA is a class I human carcinogen and its nephrotoxicity is due to several molecular and cellular mechanisms. A key aspect in the pathogenesis is damage to mitochondrial DNA, which significantly impairs energy production in organs with high metabolic demand, such as the kidneys and liver. According to Chen et al., (2024) [19], this mechanism contributes to cellular dysfunction and eventually apoptosis of renal cells. Similarly, some authors have pointed out that this acid binds to proteins essential for mitochondrial metabolism and homeostasis, which alters biosynthetic and energetic processes in renal cells and exacerbates renal injury [18].
Zhang et al., (2022) and Chen et al., (2024) [18, 19] agree and substantiate their approach by pointing out that this disease is inextricably linked to prolonged exposure to toxic compounds contained in certain plants of the Aristolochiaceae family. These plants, traditionally used in herbal medicine in various regions, contain this compound, which has been identified as a carcinogen due to its ability to cause significant damage to kidney and upper urinary tract tissue (18).
On the other hand, Wang et al., (2019) [20] point out that this aggregate generates specific DNA adducts, especially in critical genes such as TP53. These adducts compromise the genetic integrity of the cells and cause mutations that not only contribute to the development of urothelial neoplasms but also impair essential cellular repair mechanisms in the kidneys. Upadhyay and Batuman (2022) [21] support this theory and add that the accumulation of these alterations is an important diagnostic marker and a key factor in the progression of AAN.
In several articles, the authors have focused on the mechanisms of mitochondrial toxicity, as AA-induced nephrotoxicity is associated with extensive damage to mitochondrial DNA (mtDNA). This damage directly affects the bioenergetic function of renal cells, particularly in the proximal tubules, which are highly dependent on mitochondrial energy for their normal function [17, 18, 19, 20]. Pozdzik et al., (2020) [22] point out that mitochondrial dysfunction is characterized by a decrease in ATP production and an increase in reactive oxygen species (ROS) generation, which promotes both oxidative stress and cell apoptosis.
Chan and Ham (2021) [17] describes another important mechanism as the activation of inflammatory pathways and fibrosis because of exposure to AA, which triggers chronic inflammatory responses that contribute to interstitial kidney damage. Activation of signaling pathways such as the NLRP3 inflammasome and the C3a/C3aR axis in macrophages creates an inflammatory microenvironment that promotes the production of proinflammatory cytokines and enzymes such as matrix metalloproteinase-9 (MMP-9) [18]. These molecules play an essential role in the degradation of the extracellular matrix and in the development of renal interstitial fibrosis.
Upadhyay and Batuman (2022) [21] point out that the toxicity of this compound induces metabolic changes that also lead to metabolic disorders, such as alteration of lipid metabolism and overproduction of fat-derived inflammatory mediators. These changes not only exacerbate oxidative stress but also impair cellular homeostasis, perpetuating tubular damage and progression of CKD [21, 22].
It should be mentioned that other mechanisms related to this pathology are genetic and environmental factors, mainly due to susceptibility to AAN, which can be modulated by genetic factors affecting the ability to metabolize and detoxify AA [18, 20]. Chen et al., (2024) [19] describe that polymorphisms in genes such as those encoding cytochrome P450 and glutathione S-transferase enzymes may determine the rate at which these toxic compounds are processed and thus influence the extent of renal damage. In addition, he notes that environmental factors such as repeated exposure to AA-contaminated water or food increase the risk of developing the disease.
Chen et al., (2024) [19] support this theory and clarify that another relevant mechanism is the activation of the C3a/C3aR axis in macrophages, which leads to an increase in the expression of MMP-9, a key factor in the development of renal interstitial fibrosis. Similarly, Wang et al., (2019) [20] emphasize the induction of the enzyme indoleamine 2,3-dioxygenase-1(IDO1)as an important mediator in renal tubulointerstitial fibrosis. In addition, Chen et al., (2024) and Wang et al., (2019) [19, 20] agree that alterations in lipid metabolism and activation of inflammatory and oxidative stress pathways are associated with renal dysfunction induced by this substance. The role of the NLRP3 inflammasome has also been extensively documented as a central mechanism in nephropathy induced by this acid, and its inhibition is proposed as a potential therapeutic strategy, as suggested by the results of these authors [18, 19, 20].
In terms of physiopathology, Upadhyay and Batuman (2022) [21] point out that interstitial nephritis caused by AA is a progressive renal disease characterized by interstitial fibrosis and tubular atrophy as a result of prolonged exposure to this compound contained in certain medicinal plants used in traditional medicine. Conversely, Pozdzik et al., (2020) [22] underscore the multifaceted pathogenesis of this disease, attributing its complexity to the interplay of numerous molecular and cellular mechanisms that collectively contribute to its progression (Figure 2).
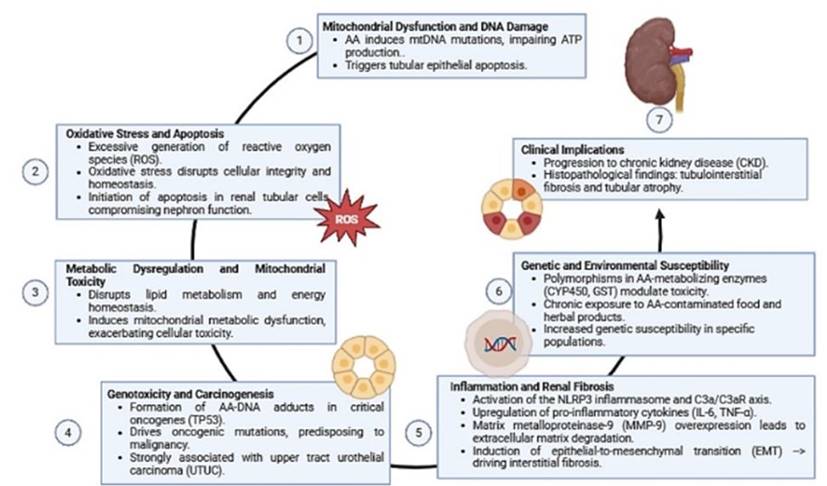
Figure 2
Mechanisms of kidney injury induced by aristolochic acid (AA)
A number of authors have explored and described these mechanisms, providing a detailed overview of the processes involved.One of the central aspects in the pathogenesis of AAN is mitochondrial DNA damage. Zhang et al., (2022) [18] support this theory and emphasize that exposure to this compound induces mutations in mitochondrial DNA, resulting in significant mitochondrial dysfunction. This dysfunction manifests as a reduction in ATP production, affecting the ability of renal cells to maintain essential metabolic functions. Wang et al., (2019) and Upadhyay and Batuman (2022) [20, 21] document that this compound binds to key proteins involved in mitochondrial metabolism, altering biosynthetic processes and promoting renal cell apoptosis. These findings underscore the importance of energy metabolism in the pathogenesis of AAN.
As Pozdzik et al., (2020) and Chen et al., (2024) [19, 22] have demonstrated, metabolic alterations also play a crucial role in the development of this pathology. Recent studies have shown that AA negatively impacts lipid metabolism, which contributes to the development of oxidative stress and activation of inflammatory responses. Consequently, Chan and Ham (2021) [17] underscores the pivotal role of ROS production and NLRP3 inflammasome activation in the progression of renal damage, given their function as mediators in the enhancement of inflammatory processes and fibrosis, which are intricately linked in the pathogenesis of the disease.
In the context of inflammation and fibrosis, the authors delineate the significance of the C3a/C3aR axis in the activation of macrophages, a pivotal factor in the promotion of renal interstitial fibrosis. As posited by Upadhyay and Batuman (2022) [21], this activation culminates in an augmentation of MMP-9 metalloproteinase expression, which is instrumental in the degradation of components of the extracellular matrix and contributes to fibrotic tissue remodeling. Furthermore, Chen et al., (2024) [19] underscore the pivotal role of epitheliomesenchymal transition (EMT), induced by AA, in facilitating the accumulation of fibroblasts and collagen deposition, thereby accelerating the progression of fibrosis and consolidating renal structural damage. Therefore, accumulating evidence suggests that the interrelated mechanisms of mitochondrial dysfunction, metabolic alterations, inflammation, and fibrosis are critical components in the pathogenesis of this disease [19, 21].
Conversely, Pozdzik et al., (2020) and Yang et al., (2012) [22, 25] posit that the contribution of innate and adaptive immune responses plays a crucial role in the exacerbation of renal damage. Recent studies highlight that AA not only activates NLRP3 but also induces the production of chemokines such as CCL2 and CXCL10, which recruit macrophages and T lymphocytes towards the affected renal tissue. This heightened inflammatory response, in turn, fosters the sustained release of proinflammatory cytokines such as IL-6 and TNF-α, amplifying the inflammatory response [26]. Chen et al., (2013) [27] underscore the pivotal role of these molecules in promoting a transition from an acute to a chronic inflammatory state, thereby perpetuating tissue damage and fibrotic remodeling.
Another salient aspect of pathophysiology pertains to the disruption of cell signaling pathways involved in tissue repair, given that AA directly affects signaling cascades mediated by TGF-β1, an essential growth factor in the regulation of the balance between repair and fibrosis [28]. Kim and Tam (2011) [29] observe that under the influence of TGF-β1, tubular epithelial cells adopt mesenchymal phenotypes through the EMT transition process. This phenotypic change increases the population of active myofibroblasts, which are responsible for the excessive production of collagen and other extracellular matrix proteins.This imbalance in the synthesis and degradation of the matrix results in the progressive loss of tissue architecture and renal functionality, consolidating the irreversible damage in this pathology [28, 29].
Luciano and Perazella (2015) [15] posit that the evaluation of the clinical manifestation of AAN poses a significant challenge, primarily due to its predominantly asymptomatic progression, which often remains undetected through routine hematological examinations.Patients exhibiting a symptomatic presentation typically present with nonspecific symptoms, including asthenia, nausea, edema, and anorexia [24]. The clinical signs manifest initially in patients between 40 and 50 years of age; discoveries before the age of 20 years are uncommon, and there is a higher prevalence in the female sex since herbalists are used as weight loss programs [15].
Yang et al., (2012) [25] conducted a study in which 300 patients diagnosed with AAN and a follow-up no less than 156 months were able to establish three clinical subtypes: chronic, acute, and renal tubular dysfunction. These three main clinical patterns exhibited distinctive pathological features. Chronic AAN, which affects more than 90% of patients, is characterized by progressive renal disease, hypertension, anemia, and renal atrophy, associated with extensive interstitial fibrosis with tubular atrophy and dilatation, resulting from long-term cumulative exposure [12].
Acute AAN, a less frequent occurrence, manifests subsequent to excessive ingestions within a brief period, resulting in acute renal injury characterized by severe tubular necrosis, as indicated by areas of bare tubular basement membrane and an absence of cellular regeneration [12]. Luciano and Perazella (2015) [15] assert that AAN renal tubular dysfunction, the least common manifestation, emerges subsequent to intermittent low-dose exposures and is associated with symptoms such as polyuria, Fanconi's syndrome, and gastrointestinal abnormalities. The clinical outcomes associated with AAN exposure are diverse, ranging from rapid progression to end-stage renal disease (ESRD) in the context of chronic AAN, to the potential stabilization of renal function in cases of renal tubular dysfunction [26].
Yang et al., (2012) [25] found that more than 90% of patients with the chronic subtype developed a rapid progression to ESRD within two years of exposure. In contrast, in acute neph In contrast, in acute nephropathy, approximately 5% of patients exhibited non-oliguric acute renal injury, which developed due to excessive use of herbal products within a time span of less than five months.
A greater susceptibility influenced by genetic and environmental factors has been demonstrated [13]. Chen et al., (2013) [27] exposed to racial or ethnic susceptibility, epidemiological studies have shown that certain populations, such as those of Taiwan and endemic regions of the Balkans, are particularly vulnerable. Yang et al., (2012) [25] conducted research between 1997 and 2003, revealing that 39% of the Taiwanese population was exposed to herbs with AA, which resulted in an increased risk of renal injury and UTUC, particularly in women and younger individuals. Less than 3% of patients with intermittent and minimal cumulative intake of AA-I showed varying degrees of renal tubular dysfunction [24]. In animal models, it has been observed that signs of renal damage can appear in as little as 10 days of exposure to high doses of AA [9].
AAN is characterized by nonspecific clinical findings, including elevated creatinine levels detected in routine tests [12]. As the disease progresses to CKD, hypertension, anemia, and decreased glomerular filtration rate become prevalent [26]. This results in proximal tubule impairment, manifested by glycosuria with normal blood glucose and mild proteinuria in the urine study, in addition to sterile pyuria and leukocyte casts in the urinary sediment [18].
Yang et al., (2012) [25] document the presence of urinary markers, such as retinol-binding protein and N-acetyl-β-glucosaminidase, indicative of tubular cell deterioration.Imaging studies reveal that approximately 50% of cases manifest reduced kidney size with irregular contours on ultrasound, reflecting advanced structural alterations [9]. Individuals with AAN are at high risk of developing upper urinary tract carcinomas (UTUC) [28]. Chen et al., (2013) [27] report that approximately 40% of UTUC cases are caused by the creation of specific DNA adducts of AA, which cause specific mutations in tumor suppressor genes, such as TP53, increasing the propensity for urothelial malignancies. The high prevalence of UTUC highlights the importance of oncologic surveillance. The diagnosis of AAN lacks universal criteria and is based on exposure history, clinical manifestations of tubular damage, and histopathological findings [9]. The diagnosis of AAN is distinguished by a combination of clinical, pathological, and laboratory findings that guide its identification. A crucial factor in identifying the disease is a history of contact with AA-containing products, such as certain medicinal plants [24].
The role of renal biopsy is crucial, as evidenced by the presence of severe interstitial fibrosis and hypocellular interstitial infiltrate, in addition to tubular brush membrane ablation in acute situations or tubular basement membrane thickening in chronic situations [12]. Furthermore, Chen et al., (2013) [27] identified aristolactam DNA adducts in renal tissue, corroborating AA exposure, which is also related to specific mutations in the TP53 gene and an elevated risk of urothelial carcinomas. Kim and Tam (2011) [29] exposed monocyte chemoattractant protein 1 (MCP-1) in AAN, which may provide information for the diagnosis and monitoring of this disease.
As posited by Zhang et al., (2022) [18], the presence of metabolic modifications in urine appears to exhibit enhanced sensitivity as a diagnostic marker when compared with conventional biomarkers of kidney damage, such as blood creatinine and blood urea nitrogen (BUN). This observation signifies that a panel of urinary biomarkers possesses the capacity to discern alterations at earlier stages of the disease, a pivotal factor in facilitating timely intervention. Conversely, the study by Wu et al., (2010) [30] underscores the diagnostic value of urinary MCP-1 levels in detecting severe interstitial edema and inflammatory infiltration in drug-induced tubulointerstitial nephritis, signifying its potential benefit in assessing the severity of acute kidney injury.
Gökmen et al., (2013) [9] propose a set of criteria for diagnosing AAN. These criteria include the following: an estimated glomerular filtration rate (eGFR) of 60 ml/min/1.73 m² or less, in conjunction with at least two of the following conditions: a history of consumption of AA-containing products, the presence of tubulointerstitial fibrosis accompanied by a reduction in renal cortex, and the identification of AA DNA adducts in affected tissues (Table 1).
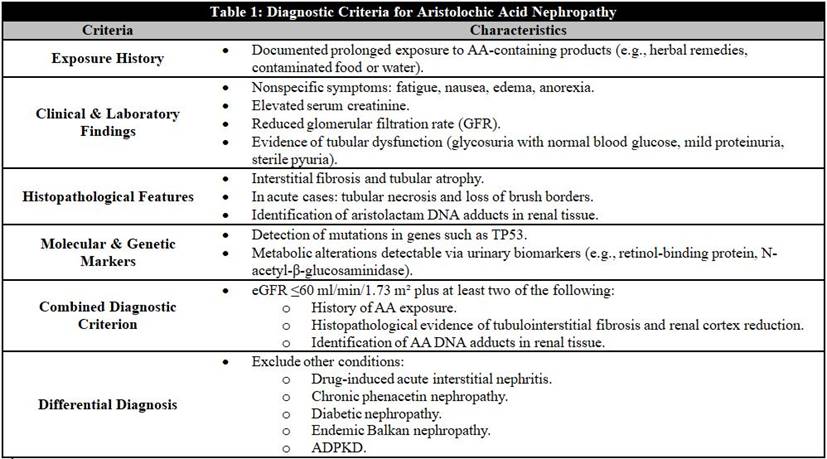
The differential composition of AAN is further investigated in the context of diseases such as drug-induced acute interstitial nephritis (AIN), which can be triggered by antibiotics, NSAIDs, and diuretics [9]. Although AIN can lead to acute kidney injury, it is not associated with the heightened risk of urothelial cancer typically observed in AIN [26].
The development of nephropathy due to chronic phenacetin use has been associated with the occurrence of papillary necrosis of the kidneys and chronic interstitial nephritis. This condition has been shown to heighten the risk of developing transitional urinary cell carcinoma, akin to AAN [9]. Diabetic nephropathy, characterized by glycosuria and a progression of renal dysfunction, differs from AAN in that it does not carry a high risk of urothelial cancer. Additionally, it is associated with proteinuria and diabetic retinopathy [29]. Endemic Balkan nephropathy (BEN), associated with exposure to AA through food contamination, has also been linked to a high risk of urothelial carcinoma, akin to AAN [13].
Finally, autosomal dominant polycystic renal disease (ADPKD), despite its association with progressive renal disease, has not been linked to glycosuria or an elevated risk of urothelial cancer. ADPKD is characterized by the presence of renal and hepatic cysts [12].
Chuanting Xu et al., (2024) [31] emphasize that the therapeutic management of AAN-associated nephropathy remains constrained, with a predominant emphasis on symptom management and disease progression mitigation. Notwithstanding, preliminary uncontrolled studies have indicated that glucocorticoids may offer a therapeutic benefit by decelerating the progression of the pathology. However, this therapeutic approach has not been sufficiently efficacious in halting the progression of the disease, and the majority of patients ultimately progress to end-stage renal disease (ESRD) (Table 2).
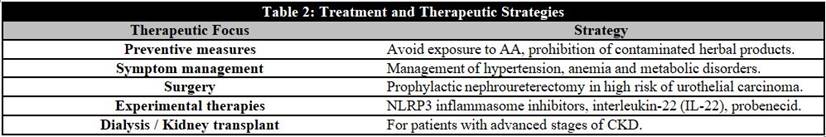
Luciano and Perazella (2015) [15] recommend prophylactic bilateral nephroureterectomies as a key preventive strategy in patients at high risk of developing upper urinary tract cancers, suggesting that this surgical intervention significantly reduces the likelihood of malignant neoplasms in the kidneys and ureters by removing tissues susceptible to malignant tumor transformation.
In preclinical research, the use of probenecid, an inhibitor of organic anion transporters, has been explored as a possible therapeutic strategy. This approach has been shown to effectively prevent acute tubular necrosis induced by AA exposure in animal models, suggesting significant potential in mitigating renal damage associated with this toxin [32].
In a related study, Shaofei Wang et al., (2019) [20] identified the NLRP3 inflammasome as a central mediator in the pathogenesis of AAN-associated nephropathy, highlighting its critical role in the inflammatory processes and renal damage associated with this disease. In this context, interleukin 22 (IL-22) has been observed to possess a beneficial effect by significantly reducing renal tubular damage in preclinical studies in animal models, suggesting its potential as a promising therapeutic tool.
One of the most serious complications associated with this condition is its rapid progression to CKD and eventually to ESRD, mainly due to the presence of severe interstitial fibrosis and pronounced tubular atrophy, which are hallmarks of this pathology. Moreover, these structural alterations play a crucial role in the accelerated and progressive deterioration of renal function [15].
A notable complication is the high incidence of urothelial carcinoma, which affects approximately 40-45% of patients diagnosed with AAN-associated nephropathy. This high prevalence is attributed to the carcinogenic properties of AA, a substance capable of inducing genetic mutations in key elements such as the TP53 gene, facilitating the development of malignant tumors in the upper urinary tract [33].
Renal fibrosis constitutes another of the main complications of AAN-associated nephropathy. This complication is characterized by the activation of resident fibroblasts and epithelial-mesenchymal transition (EMT) processes that lead to an excessive accumulation of collagen in the renal tissue. This pathological phenomenon is driven by chronic inflammatory mechanisms, among which the activation of the NLRP3 inflammasome stands out [20].
While AAN primarily affects the renal interstitium, it can also inflict substantial damage to the glomeruli, manifesting clinically as albuminuria. This phenomenon is closely related to mitochondrial damage in podocytes, and the deterioration of these structures severely compromises the kidney's ability to filter and maintain homeostasis [15, 16].
Jiayun Chen et al., (2022) [34] describe other complications associated with AAN, including renal vascular compromise, which significantly alters organ perfusion and exacerbates ischemic tissue damage. This vascular impairment is also associated with the development of secondary hypertension, a condition that further aggravates renal impairment and increases the risk of cardiovascular events.
The prognosis of AAN can vary considerably depending on the clinical presentation and the amount of AA ingested, since this disease can manifest itself acutely or chronically. In cases of acute presentation, the possibility of renal functional recovery in these patients is extremely low due to the magnitude of structural and functional damage suffered by the renal tissue. This underscores the importance of early diagnosis and preventive or therapeutic interventions [15, 25].
In the chronic presentation, which represents the most common form of AAN, patients usually develop chronic tubulointerstitial nephropathy, which is characterized by a progressive and sustained deterioration of renal function, manifested mainly by a gradual decrease in glomerular filtration rate (GFR). This form of the disease, being chronic in nature, is closely linked to a high rate of progression to end-stage renal disease (ESRD) [15].
An additional significant challenge in AAN is its close relationship with urothelial carcinoma, the incidence of which is estimated to affect between 40% and 45% of patients. This high rate of malignancy is associated with the mutagenic properties of AA, which induce mutations in crucial genes such as TP53 [35].
CONCLUSIONS
AAN is a kidney disease of multifactorial etiology whose pathogenesis involves complex and interrelated mechanisms, such as mitochondrial DNA damage, metabolic alterations, inflammation, and progressive fibrosis. The chemical structure of aristolochic acids is that of nitrophenanthrenic carboxylic acids. AA-I and AA-II differ mainly in the presence of a methoxy group in AA-I, which contributes to its higher absorption and toxicity compared to AA-II. In addition, there are other aristolochic acid analogs that may also contribute to toxicity, such as aristolochic acid IV and several aristolochalactams.
In the United States, the Food and Drug Administration (FDA) has issued warnings about the toxic effects of AAs and has banned the sale of products containing them. Similarly, in other regions, such as Morocco, regulations have been implemented to discontinue the use of Aristolochia species due to their renal toxicity and carcinogenic potential. However, despite these restrictions, products containing AAs are still manufactured and marketed in some parts of the world, indicating inadequate regulation and the need for stricter action to prevent exposure to these compounds.
Prolonged exposure to the acids presents in certain medicinal plants and contaminated foods leads to mitochondrial dysfunction and cell apoptosis, as well as the activation of inflammatory pathways such as the NLRP3 inflammasome and the promotion of EMT transition, which contributes significantly to the development of irreversible renal fibrosis. These findings underscore the necessity of implementing preventive measures to reduce exposure to these compounds and highlight the imperative for the development of targeted therapies that mitigate kidney damage and slow the progression of this debilitating disease. Patients with AAN are at an elevated risk of developing urothelial carcinomas due to mutations in tumor suppressor genes, particularly TP53, highlighting the need for oncologic surveillance. Diagnosis is based on a patient's history of exposure to AA-containing products, compatible clinical findings, a renal biopsy revealing interstitial fibrosis and tubular basement membrane changes, and the detection of AA DNA adducts. Urinary biomarkers such as MCP-1 provide additional information in the early evaluation and follow-up of the disease.
Given the severe impact of AAN on renal function and its association with severe complications, such as end-stage renal disease and a high risk of urothelial carcinoma, a comprehensive approach to its management is required. Treatment is based on the immediate elimination of AA exposure and the management of renal complications through dialysis support or renal transplantation in advanced cases. Early detection is key to mitigating disease progression and reducing the risk of neoplasms, underscoring the importance of continuous oncologic surveillance.
In terms of new treatments, there is growing interest in developing therapies that can remove amyloid deposits from tissues. However, so far, results have been limited and no effective treatment for the removal of amyloid fibrils has been established. The medical literature suggests that, although advances have been made in the management of inflammation, agents capable of clearing amyloid deposits are still needed to further improve patient outcomes. In addition, renal transplantation is an option for patients with renal failure, and recurrence of amyloidosis in the graft has become rare thanks to new therapeutic agents.
CONFLICT OF INTERESTS
The authors have no conflict of interest to declare. The authors declared that this study has received no financial support.
REFERENCES
1.Ji H, Hu J, Zhang G, Song J, Zhou X, Guo D. Aristolochic acid nephropathy: A scientometric analysis of literature published from 1971 to 2019. Medicine (Baltimore). 2021;100(27):e26510. doi: 10.1097/MD.0000000000026510.
2.Jadot I, Declèves AE, Nortier J, Caron N. An Integrated View of Aristolochic Acid Nephropathy: Update of the Literature. Int J Mol Sci. 2017;18(2):297. doi: 10.3390/ijms18020297.
3.Vanherweghem JL, Depierreux M, Tielemans C, Abramowicz D, Dratwa M, Jadoul M, et al. Rapidly progressive interstitial renal fibrosis in young women: association with slimming regimen including Chinese herbs. Lancet. 1993;341(8842):387-91. doi: 10.1016/0140-6736(93)92984-2.
4.IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Some traditional herbal medicines, some mycotoxins, naphthalene and styrene. IARC Monogr Eval Carcinog Risks Hum. 2002;82:1-556.
5.Nortier JL, Martinez MC, Schmeiser HH, Arlt VM, Bieler CA, Petein M, et al. Urothelial carcinoma associated with the use of a Chinese herb (Aristolochia fangchi). N Engl J Med. 2000;342(23):1686-92. doi: 10.1056/NEJM200006083422301.
6.Lai MN, Lai JN, Chen PC, Tseng WL, Chen YY, Hwang JS, et al. Increased risks of chronic kidney disease associated with prescribed Chinese herbal products suspected to contain aristolochic acid. Nephrology (Carlton). 2009;14(2):227-34. doi: 10.1111/j.1440-1797.2008.01061.x.
7.Nortier J, Pozdzik A, Roumeguere T, Vanherweghem JL. [Aristolochic acid nephropathy ("Chinese herb nephropathy")]. Nephrol Ther. 2015;11(7):574-88. doi: 10.1016/j.nephro.2015.10.001.
8.Baudoux T, Jadot I, Declèves AE, Antoine MH, Colet JM, Botton O, et al. Experimental Aristolochic Acid Nephropathy: A Relevant Model to Study AKI-to-CKD Transition. Front Med (Lausanne). 2022;9:822870. doi: 10.3389/fmed.2022.822870.
9.Gökmen MR, Cosyns JP, Arlt VM, Stiborová M, Phillips DH, Schmeiser HH, et al. The epidemiology, diagnosis, and management of aristolochic acid nephropathy: a narrative review. Ann Intern Med. 2013;158(6):469-77. doi: 10.7326/0003-4819-158-6-201303190-00006.
10.Zhang L, Zhang P, Wang F, Zuo L, Zhou Y, Shi Y, et al. Prevalence and factors associated with CKD: a population study from Beijing. Am J Kidney Dis. 2008;51(3):373-84. doi: 10.1053/j.ajkd.2007.11.009.
11.Su T, Zhang L, Li X, Zuo L, Zhang P, Wang H. Regular use of nephrotoxic medications is an independent risk factor for chronic kidney disease--results from a Chinese population study. Nephrol Dial Transplant. 2011;26(6):1916-23. doi: 10.1093/ndt/gfq679.
12.Zhou Q, Jiang L, Su T, Liu G, Yang L. Overview of aristolochic acid nephropathy: an update. Kidney Res Clin Pract. 2023;42(5):579-90. doi: 10.23876/j.krcp.22.211.
13.Jelaković B, Dika Ž, Arlt VM, Stiborova M, Pavlović NM, Nikolić J, et al. Balkan Endemic Nephropathy and the Causative Role of Aristolochic Acid. Semin Nephrol. 2019;39(3):284-96. doi: 10.1016/j.semnephrol.2019.02.007.
14.Ban TH, Min JW, Seo C, Kim DR, Lee YH, Chung BH, et al. Update of aristolochic acid nephropathy in Korea. Korean J Intern Med. 2018;33(5):961-69. doi: 10.3904/kjim.2016.288.
15.Luciano RL, Perazella MA. Aristolochic acid nephropathy: epidemiology, clinical presentation, and treatment. Drug Saf. 2015;38(1):55-64. doi: 10.1007/s40264-014-0244-x.
16.Han J, Xian Z, Zhang Y, Liu J, Liang A. Systematic Overview of Aristolochic Acids: Nephrotoxicity, Carcinogenicity, and Underlying Mechanisms. Front Pharmacol. 2019;10:648. doi: 10.3389/fphar.2019.00648.
17.Chan W, Ham YH. Probing the Hidden Role of Mitochondrial DNA Damage and Dysfunction in the Etiology of Aristolochic Acid Nephropathy. Chem Res Toxicol. 2021;34(8):1903-9. doi: 10.1021/acs.chemrestox.1c00175.
18.Zhang Q, Luo P, Chen J, Yang C, Xia F, Zhang J, et al. Dissection of Targeting Molecular Mechanisms of Aristolochic Acid-induced Nephrotoxicity via a Combined Deconvolution Strategy of Chemoproteomics and Metabolomics. Int J Biol Sci. 2022;18(5):2003-17. doi: 10.7150/ijbs.69618.
19.Chen L, Cheng S, Ying J, Zhang Q, Wang C, Wu H, et al. Aristolochic acid I promotes renal tubulointerstitial fibrosis by up-regulating expression of indoleamine 2,3-dioxygenase-1 (IDO1). Toxicol Lett. 2024;402:44-55. doi: 10.1016/j.toxlet.2024.11.003.
20.Wang S, Fan J, Mei X, Luan J, Li Y, Zhang X, et al. Interleukin-22 Attenuated Renal Tubular Injury in Aristolochic Acid Nephropathy via Suppressing Activation of NLRP3 Inflammasome. Front Immunol. 2019;10:2277. doi: 10.3389/fimmu.2019.02277.
21.Upadhyay R, Batuman V. Aristolochic acid I induces proximal tubule injury through ROS/HMGB1/mt DNA mediated activation of TLRs. J Cell Mol Med. 2022;26(15):4277-91. doi: 10.1111/jcmm.17451.
22.Pozdzik AA, Salmon IJ, Debelle FD, Decaestecker C, Van den Branden C, Verbeelen D, et al. Aristolochic acid induces proximal tubule apoptosis and epithelial to mesenchymal transformation. Kidney Int. 2008;73(5):595-607. doi: 10.1038/sj.ki.5002714.
23.Grollman AP. Aristolochic acid nephropathy: Harbinger of a global iatrogenic disease. Environ Mol Mutagen. 2013;54(1):1-7. doi: 10.1002/em.21756.
24.De Broe ME. Chinese herbs nephropathy and Balkan endemic nephropathy: toward a single entity, aristolochic acid nephropathy. Kidney Int. 2012;81(6):513-5. doi: 10.1038/ki.2011.428.
25.Yang L, Su T, Li XM, Wang X, Cai SQ, Meng LQ, et al. Aristolochic acid nephropathy: variation in presentation and prognosis. Nephrol Dial Transplant. 2012;27(1):292-8. doi: 10.1093/ndt/gfr291.
26.Witkowicz J. [Aristolochic acid nephropathy]. Przegl Lek. 2009;66(5):253-56.
27.Chen CH, Dickman KG, Huang CY, Moriya M, Shun CT, Tai HC, et al. Aristolochic acid-induced upper tract urothelial carcinoma in Taiwan: clinical characteristics and outcomes. Int J Cancer. 2013;133(1):14-20. doi: 10.1002/ijc.28013.
28.Rebhan K, Ertl IE, Shariat SF, Grollman AP, Rosenquist T. Aristolochic acid and its effect on different cancers in uro-oncology. Curr Opin Urol. 2020;30(5):689-95. doi: 10.1097/MOU.0000000000000806.
29.Kim MJ, Tam FW. Urinary monocyte chemoattractant protein-1 in renal disease. Clin Chim Acta. 2011;412(23-24):2022-30. doi: 10.1016/j.cca.2011.07.023.
30.Wu Y, Yang L, Su T, Wang C, Liu G, Li XM. Pathological significance of a panel of urinary biomarkers in patients with drug-induced tubulointerstitial nephritis. Clin J Am Soc Nephrol. 2010;5(11):1954-9. doi: 10.2215/CJN.02370310.
31.Xu C, Wang Q, Du C, Chen L, Zhou Z, Zhang Z, et al. Histone deacetylase-mediated silencing of PSTPIP2 expression contributes to aristolochic acid nephropathy-induced PANoptosis. Br J Pharmacol. 2024;181(9):1452-73. doi: 10.1111/bph.16299.
32.Baudoux TE, Pozdzik AA, Arlt VM, De Prez EG, Antoine MH, Quellard N, et al. Probenecid prevents acute tubular necrosis in a mouse model of aristolochic acid nephropathy. Kidney Int. 2012;82(10):1105-13. doi: 10.1038/ki.2012.264.
33.Zhou Y, Bian X, Fang L, He W, Dai C, Yang J. Aristolochic acid causes albuminuria by promoting mitochondrial DNA damage and dysfunction in podocyte. PLoS One. 2013;8(12):e83408. doi: 10.1371/journal.pone.0083408.
34.Chen J, Luo P, Wang C, Yang C, Bai Y, He X, et al. Integrated single-cell transcriptomics and proteomics reveal cellular-specific responses and microenvironment remodeling in aristolochic acid nephropathy. JCI Insight. 2022;7(16):e157360. doi: 10.1172/jci.insight.157360.
35.Shan H, Tian W, Hong Y, Xu B, Wang C, Yu B, et al. Clinicopathologic characteristics and prognosis of upper tract urothelial carcinoma complicated with aristolochic acid nephropathy after radical nephroureterectomy. BMC Complement Med Ther. 2020;20(1):166. doi: 10.1186/s12906-020-2861-5.
Notas de autor
juan.reinoso.40@est.ucacue.edu.ec
Información adicional
redalyc-journal-id: 6920
