Original article
Sero-Prevalence of Transfusion Transmissible Infections among Prospective Blood Donors in Ekpoma and its Environs
Seroprevalencia de infecciones transmisibles por transfusión entre posibles donantes de sangre en Ekpoma y sus alrededores
Sero-Prevalence of Transfusion Transmissible Infections among Prospective Blood Donors in Ekpoma and its Environs
Iberoamerican Journal of Medicine, vol. 7, núm. 3, pp. 78-85, 2025
Hospital San Pedro
Recepción: 02 Abril 2025
Corregido: 10 Julio 2025
Aprobación: 24 Julio 2025
Abstract:
Introduction: Hepatitis B, C, and HIV are serious public health issues in developing nations. They can be spread through high-risk sexual behavior, vertical transmission, or parental contact, and they can result in both acute and chronic life-threatening illnesses. This study was carried out to determine the sero-prevalence of transfusion-transmissible infections (TTIs) among prospective blood donors in AAU Ekpoma and its environs.
Material and methods: In this cross-sectional study, four hundred and fifty (450) blood samples were aseptically collected from prospective blood donors in Ekpoma and its environs. Participants were screened for hepatitis B surface antigen, HIV, Syphilis and anti-HCV antibody using standard laboratory methods.
Results: The result obtained showed that of the 450 samples screened, 2.33% of the subjects were seropositive for HBsAg, 1.33% were positive for Syphilis, 1.0% were positive for HIV and 0.67% were positive for HCV respectively. The sex related prevalence obtained showed that 0.67% females and 0.33% male was positive for HIV, 0.33% male and 1.00% females positive for Syphilis, 1.00% males and 1.33% females were positive for HBsAg and 0 .33% male and 0.33% female were positive for HCV respectively. There was no significant difference (p>0.05) in the prevalence of HIV, Syphilis, HBV and HCV in the study population with respect to sex and age of the subjects. Risk factors such as unprotected sex accounted for 1.33%, 0.33%, 0.33% and 0.67% of HBV, HCV, HIV and Syphilis infections among seropositive prospective blood donors in Ekpoma, while previous blood transfusion accounted for 0.33%, 0.33% and 0.33% of HBV, HIV and Syphilis infections respectively.
Conclusions: The observed sero-prevalence of TTIs among prospective blood donors in Ekpoma among undergraduates emphasizes the significance of early detection of disease agents in prevention and effective management of disease outbreaks especially in resource-limited settings.
Keywords: Transfusion, Transmissible infections, Blood donors, Hepatitis B virus, HIV, Syphilis, Hepatitis C virus.
Resumen:
Introducción: La hepatitis B y C, y el VIH son graves problemas de salud pública en los países en desarrollo. Pueden propagarse a través de conductas sexuales de alto riesgo, transmisión vertical o contacto parental, y pueden provocar enfermedades graves y crónicas potencialmente mortales. Este estudio se realizó para determinar la seroprevalencia de infecciones transmisibles por transfusión (ITT) entre los posibles donantes de sangre de la UCA de Ekpoma y sus alrededores.
Material y métodos: En este estudio transversal, se recogieron asépticamente cuatrocientas cincuenta (450) muestras de sangre de los posibles donantes de sangre de Ekpoma y sus alrededores. Se realizó una prueba de detección del antígeno de superficie de la hepatitis B, VIH, sífilis y anticuerpos anti-VHC a los participantes mediante métodos de laboratorio estándar.
Resultados: El resultado obtenido mostró que de las 450 muestras examinadas, 2.33% de los sujetos fueron seropositivos para HBsAg, 1.33% fueron positivos para Sífilis, 1.0% fueron positivos para VIH y 0.67% fueron positivos para VHC respectivamente. La prevalencia relacionada con el sexo obtenida mostró que 0.67% mujeres y 0.33% hombres fueron positivos para VIH, 0.33% hombres y 1.00% mujeres fueron positivos para Sífilis, 1.00% hombres y 1.33% mujeres fueron positivos para HBsAg y 0.33% hombres y 0.33% mujeres fueron positivos para VHC respectivamente. No hubo diferencia significativa (p>0.05) en la prevalencia de VIH, Sífilis, VHB y VHC en la población de estudio con respecto al sexo y la edad de los sujetos. Factores de riesgo como las relaciones sexuales sin protección representaron el 1,33 %, el 0,33 %, el 0,33 % y el 0,67 % de las infecciones por VHB, VHC, VIH y sífilis entre los posibles donantes de sangre seropositivos en Ekpoma, respectivamente. Las transfusiones de sangre previas representaron el 0,33 %, el 0,33 % y el 0,33 % de las infecciones por VHB, VIH y sífilis, respectivamente.
Conclusiones: La seroprevalencia observada de ITT entre los posibles donantes de sangre de Ekpoma, entre los estudiantes universitarios, destaca la importancia de la detección temprana de agentes patógenos para la prevención y el manejo eficaz de brotes de enfermedades, especialmente en entornos con recursos limitados.
Palabras clave: Transfusión, Infecciones transmisibles, Donantes de sangre, Virus hepatitis B, VIH , Sífilis, Virus hepatitis C.
INTRODUCTION
According to Erhabor et al. [1], Busayo et al. [2], and Iyoha et al. [3], infectious agents like syphilis, hepatitis A virus (HAV), hepatitis B virus (HBV), hepatitis C virus (HCV), and HIV pose major risks to transfusion recipients' safety and present significant public health issues. According to Nwogoh et al. [4], there is a chance that blood transfusions will spread potentially fatal transfusion-transmittable diseases. Hepatitis B, C, and HIV are serious public health issues in developing nations. They can be spread through high-risk sexual behavior, vertical transmission, or parental contact, and they can result in both acute and chronic life-threatening illnesses [2, 5].
A crucial step in making sure transfusions are as safe as possible is screening for transfusion-transmissible infections (TTIs) to weed out blood donations that could spread illness from donors to receivers [5, 6]. In many developing nations, TTIs continue to pose a serious hazard to blood safety [7]. The risk of transmission can be lowered to extremely low levels by conducting an efficient screening for signs of the presence of the most prevalent and harmful TTIs [1]. Although they do not totally remove danger, donor appraisal, laboratory screening tests, and pathogen inactivation processes are now thought to be essential strategies to lower the risk of TTI [8]. Due to the absence of routine serological testing for donors, there is a growing incidence of HIV, HBV, HCV, and syphilis transmission from donors to recipients in underdeveloped nations [9]. The World Health Organization (WHO) advised routine serological testing for transfusion-transmissible illnesses (HIV, HBV, HCV, and syphilis) in order to prevent the spread of these infections [6, 10]. In Africa, 5–10% of HIV transmissions were caused by contaminated blood transfusions [11]. Due to their substantial mortality, morbidity, and cost impact, HBV and HCV infections are a serious global health concern [11]. According to Diro et al. [7], 12.5% of blood transfusion recipients in Sub-Saharan Africa run the risk of developing post-transfusion hepatitis.
Before blood and blood components are released for clinical or manufacturing use, the World Health Organization (WHO) advises that all blood donations be examined for signs of infection in an effort to promote global blood safety [12]. WHO guidelines state that all blood donors should be required to undergo screening for syphilis, HIV, HBV, and HCV [12]. Countries that routinely screen donors for serologic infections have shown a significant decrease in transfusion-transmissible infections (TTIs) [6, 13]. However, because of the limits of viral detection methods, TTIs are still a concern. Compared to high-income countries, middle- and low-income countries have a much higher median prevalence rate of TTIs in blood donors [12]. The overall seroprevalence of major transfusion-transmissible illnesses (TTIs) among potential blood donors among undergraduates in Ekpoma, where this study is being carried out, is currently poorly documented. By offering concrete data on the frequency of these illnesses in the study population, this study seeks to close this knowledge gap. This study is important because it may help enhance transfusion safety and public health initiatives by identifying the seroprevalence of HIV, syphilis, hepatitis B, and hepatitis C among potential blood donors in the area.
MATERIAL AND METHODS
STUDY AREA
Ambrose Alli University, which is situated in Ekpoma, the administrative center of the Esan West Local Government Area of Edo State, Nigeria, is where this study was conducted. The town, which is in the southern Nigerian rainforest–savannah transitional zone, is home to about 170,123 people. The bulk of the town's residents are students, lecturers and teachers, public servants, farmers, traders, businesspeople, physicians, attorneys, and independent contractors.
STUDY POPULATION
For this study, 450 potential blood donors of both sexes who appeared to be in good health and were between the ages of 17 and 35 were enlisted.
STUDY DESIGN
The study design was a prospective study that was carried out on World Blood Donor Day, June 14, 2022, and June 14, 2023. For this study, people in Ekpoma and the surrounding area who appeared to be in good health were chosen. For convenient evaluation and guidance, the participants were invited to Gilgal Diagnostics, a Medical Laboratory Diagnostic Center located in the center of Ekpoma town. Rapid diagnostic test kits were used to test for the HIV 1 and 2 viruses, syphilis, hepatitis B, and hepatitis C viruses, respectively. The samples were examined at Ambrose Alli University's Research and Diagnostic Laboratory, which is part of the Medical Laboratory Science department at the College of Medical Sciences in Ekpoma, Edo State.
INCLUSION AND EXCLUSION CRITERIA
The study comprised ostensibly healthy people in Ekpoma who gave their assent and were at high risk of contracting hepatitis or were not known to have any chronic illnesses. The study eliminated participants with confirmed HIV, syphilis, HBsAg, and HCV, those who were already on medicine, those who appeared to be in poor health, and those who did not provide their consent.
SAMPLE COLLECTION
Under aseptic conditions, approximately three milliliters of blood were extracted from each participant's antecubital vein and placed in simple bottles. Before being centrifuged using an 800-1 Centrifuge Machine of Halomedicals Systems Limited from Jiangsu, China, at 3000 rpm for 5 minutes at room temperature within 2 hours of collection, they were labeled and left on the bench for at least an hour to allow clot retraction to occur. The serum was isolated for analysis following centrifugation. Diaspot one-step test strips were used to screen them for HIV, Syphilis, HBsAg, and HCV, respectively (Diaspot Diagnostics Inc., U.S.A).
ANALYTICAL METHODS
HBsAg and HCV Screening: Diaspot one step hepatitis B surface antigen test strips and Diaspot one step hepatitis C test strips (Diaspot Diagnostics Inc., U.S.A.) were used to screen the samples for HBsAg and HCV, respectively. With a relative sensitivity of 99.0% and a relative specificity of 98.6%, these qualitative lateral flow immunoassay test kit devices can detect both HCV and HBsAg in plasma. The manufacturer's instructions were followed for conducting the testing. Each batch of testing contained both positive and negative controls to ensure adequate test performance and to confirm the test technique.
HIV 1/2 (using Rapid Test Cassette): The Centers for Disease Control and Prevention's (CDC-UMD) HIV rapid testing serial algorithm II guideline [14] was followed in determining the serostatus of HIV 1 and 2. The first line test kit utilized was the Determine HIV-1/2 kit, which uses an immunochromagraphic approach. The testing was terminated when the Determine HIV-1/2 kit produced negative results, while the Uni-Gold HIV test kit (Trinity Biotech Plc., Bray, Ireland) validated the reactive results from the Determine HIV-1/2 kit (Abbot Diagnostics Medical Co., Ltd., Chiba, Japan). Discordant results were retested before being examined using the Stat-Pak tie-breaker test kit. The tie-breaker result was then used to determine whether the final results were positive or negative. Each series of tests was conducted with both positive and negative controls.
Syphilis (using Ultra Rapid Test strip): The strip was taken out of the foil-wrapped pouch. It was set on a spotless, level surface. A few drops of the serum were added to the sample well on the strip using a Pasteur pipette. Five minutes later, the results were read. Each batch of testing contained both positive and negative controls to ensure adequate test performance and to confirm the test technique.
STATISTICAL ANALYSIS
Simple frequency and percentage were used to display all of the results. The Statistical Package for Social Sciences (SPSS) version 21.0 was used to perform the Student's t-test and the Chi-square test for significant differences. P-values less than 0.05 were regarded as significant.
ETHICS APPROVAL
With approval number AAU/HREC/102/23, ethical approval was acquired from the Ambrose Alli University Health Research Ethic Committee of Ambrose Alli University, Ekpoma. Prior to sample collection, each participant's informed consent was obtained.
RESULTS
SOCIO-DEMOGRAPHIC CHARACTERISTICS OF THE SUBJECTS STUDIED
The sociodemographic details of the subjects under study were displayed in Table 1. For this study, 450 people were enrolled, 288 of whom were male (64%) and 162 of whom were female (36%). The participants came from a variety of age ranges. Of the study population, 21.56% were in the 18–24 age group, 25.78% were in the 25–29 age group, 29.10 percent were in the 30–34 age group, and 23.56% were in the 35+ age group. Likewise, the participants were selected from a variety of professions, including civil servants (15.40%), businesspeople (22.22%), students (26.67%), independent contractors (22.22%), and apprentices (13.56%). 50 (11.11%) of the participants had primary education, 99 (22.0%) had secondary education, 201 (44.67%) had tertiary education, and 100 (22.22%) had no formal education, according to data on participant educational attainment. Ninety percent of the study participants were Christians, and ten percent were Muslims. 168 (56.0%) had unprotected intercourse, 23 (7.67%) had previously received blood transfusions, 15 (5.0%) had scarification markings, 7 (2.33%) had previously undergone surgery, 37 (12.33%) had an intravenous drug use history, 16 (5.33%) had an unsafe injection history, and 34 (11.34%) had no risk factors identified.
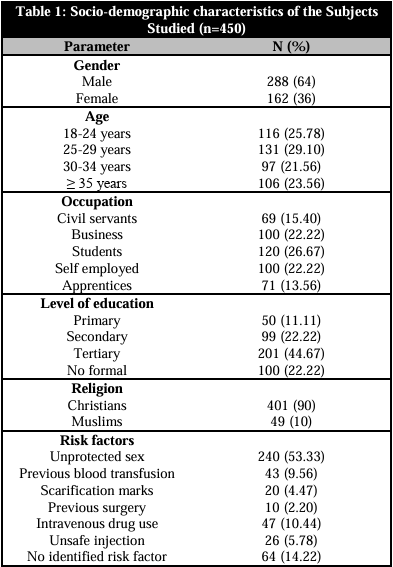
PREVALENCE OF HIV, SYPHILIS, HBsAG AND HCV AMONG THE SUBJECTS
The prevalence of HBsAg, HCV, syphilis, and HIV among the participants was displayed in Table 2. Out of the 450 samples that were evaluated, the results revealed that 9 (2.00%) of the participants tested positive for HBsAg, 6 (1.33%) tested positive for syphilis, 5 (1.0%) tested positive for HIV, and 12 (2.67%) tested positive for HCV.
HBsAg: Hepatitis B surface antigen; HIV: Human immunodeficiency virus; HCV: Hepatitis C virus
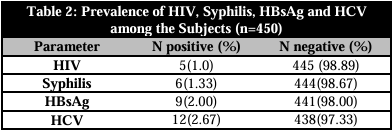
PREVALENCE OF HIV, SYPHILIS, HBsAG AND HCV AMONG THE SUBJECTS WITH RESPECT TO SEX
The prevalence of HIV, syphilis, HBsAg, and HCV among the participants in the study was displayed by sex in Table 3. According to the data, four (0.89%) of the five people who tested positive for HIV were female, and one (0.22%) was male. Likewise, out of the six individuals who tested positive for syphilis, two (0.44%) were men and four (0.89%) were women. Five (1.11%) of the nine individuals who tested positive for HBsAg were female, and four (0.89%) were male. Lastly, three (0.67%) of the twelve individuals who tested positive for HCV were men, and nine (2.00%) were women. The research population's prevalence of HIV, Syphilis, HBV, and HCV did not change significantly (p>0.05) based on sex.
HBsAg: Hepatitis B surface antigen; HIV: Human immunodeficiency virus; HCV: Hepatitis C virus
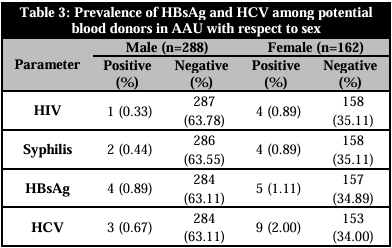
PREVALENCE OF HIV, SYPHILIS, HBsAG AND HCV AMONG THE SUBJECTS IN RELATION TO AGE
The prevalence of HBsAg, HCV, HIV, and syphilis among the participants in the study was displayed by age in Table 4. According to the data, seropositivity for HBsAg was found in the following age groups: 18–24 years old (0.22%), 25–29 years old (0.44%), 30–34 years old (0.67%), and 35 years old and older (0.67%). The age groups of 25–29 years (0.67%) and 30–34 years (0.67%) and 35 and older (1.11%) were included among the subjects who tested positive for HCV. Additionally, syphilis positive was detected in the following age groups: 18–24 years (0.22%), 25–29 years (0.22%), 30–34 years (0.44%), and 35 and older (0.44%). Lastly, HIV positive was discovered in age groups 35 and older (0.67%) and 30–34 years old (0.44%), respectively. In terms of statistics, the study population's prevalence of HIV, syphilis, HBsAg, and HCV did not differ significantly (p>0.05) based on age.
HBsAg: Hepatitis B surface antigen; HIV: Human immunodeficiency virus; HCV: Hepatitis C virus
PREVALENCE OF HIV, SYPHILIS, HBsAG AND HCV INFECTION BY RISK FACTORS
The prevalence of HIV, syphilis, HBV, and HCV infection by risk factors was displayed in Table 5. More over half (53.33%) of the study group had unprotected sex, according to the results. Among seropositive potential blood donors in Ekpoma, risk factors such unprotected sex were responsible for 1.33%, 2.00%, 0.67%, and 0.89% of HBV, HCV, HIV, and syphilis infections, while prior blood transfusions were responsible for 0.22% of HIV infections. Injection errors were responsible for 0.22% of HCV cases. HCV, HBV, HIV, and syphilis infections were caused by no known risk factors in 0.44%, 0.67%, 0.22%, and 0.44% of cases, respectively.
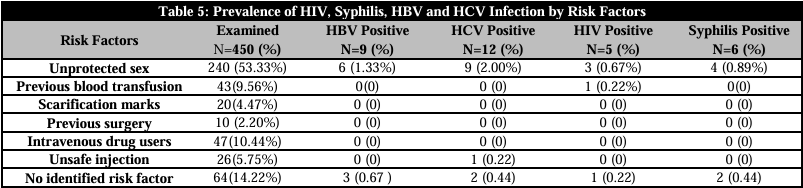
HBV: Hepatitis B virus; HIV: Human immunodeficiency virus; HCV: Hepatitis C virus
DISCUSSION
One of contemporary medicine's life-saving techniques is blood transfusion. Thorough blood screening guarantees the safe supply of blood and blood products while also providing insight into the frequency of transfusion-transmitted infections (TTIs) in healthy populations [15]. Public policy is based on estimates of disease burden that are supported by reliable epidemiological research. In a same vein, a precise assessment of TTI risk is necessary to track blood supply safety and measure the efficacy of currently used screening methods [16]. The purpose of this study was to ascertain the sero-prevalence of WHO TTIs among potential blood donors in Ekpoma and the surrounding area.
Transfusion-transmissible infections (TTIs) were detected in 7.11% of the potential donors in this investigation. The study's finding that the prevalence of TTI is declining is consistent with a global trend brought about by increased control over sexually transmitted infections, mandatory TTI screening, and the implementation of intervention programs [6, 17]. We found lower results than another study in Burkina Faso [18], Ethiopia [19], and Nigeria [20], which had 19.3, 11.5%, and 19.3%, respectively. The disparity in prevalence could be caused by variations in the severity of risk factors for acquiring transfusion-transmittable infections in the various study locations, as well as variations in the health care systems in those countries [6, 19].
The study's findings showed that among young potential blood donors in Ekpoma, the sero-prevalence rate for HBsAg was 2.00%. The prevalence of 2.00% found in this study is lower than the 10.3% reported by Iyoha et al. [3] among apparently healthy individuals in Ekpoma, Edo State. This result is also consistent with Nigeria's designation as a highly endemic nation by the World Health Organization [8]. An adult population is considered endemic if its HBsAg load is greater than 7%. A portion of the 2.00% seroprevalence rate observed in Ekpoma may be explained by the sub-urban setting of the city, where disease prevalence practices that facilitate transmission include sharing drinking cups, unprotected sex, poor vaccination schedule adherence, and relatively low vaccination coverage. Additionally, the seroprevalence values of 4.98% reported by Ejele & Ojule [16] were higher than the 2.00% prevalence of HBV identified in this study. In contrast to the academic community sampled in this study, Ejele & Ojulu [16] each sampled a group that included medical students, potential blood donors, and an apparently healthy pre-vaccination metropolitan population. It is unclear why the prevalence of HBsAg is lower in these areas than in our city and other Nigerian cities. The reduced sensitivity of the latex agglutination method in comparison to the enzyme linked immunosorbent assay (ELISA) approach may be the reason of these variances, as may the fact that some of the studies were carried out on low-risk participants [16].
In contrast, 3739 (14.96%) of the 24,979 screened potential donors over the 2005–2016 study period had at least one infectious agent infection. According to Okoroiwu et al. [21], the prevalences of HIV, syphilis, HBV, and HCV were 4.2%, 3.1%, 3.6%, and 4.1%, respectively. A national survey found that 12.2% of people had hepatitis B [22]. The effectiveness of donor selection procedures is demonstrated by reports of an incidence and prevalence of TTI among Dutch blood donors that are six to sixty times lower than those in the Dutch community at large are. This may help to explain why our studies' TTI prevalence was lower than that of other earlier research and the Nigerian national survey. The variations in HBV viral infection prevalence observed in these regions may have been caused by differences in sample size, the sensitivity and dependability of viral test reagents, the type of individuals examined, the study population's geographic location, and their sociocultural practices [8].
In Ekpoma, potential blood donors had a 2.67% sero-prevalence of the hepatitis C virus. In comparison to studies conducted in Ekpoma, Enugu, Jos, and Kaduna, which reported sero-prevalent rates of 3.0%, 14.9%, 5.2%, and 11.9%, respectively, this conclusion is lower. The documented discrepancies in HCV viral infection prevalence may potentially have been caused by variations in sample size, the sensitivity and dependability of viral test reagents, the category of individuals, the study population's geographic location, and their sociocultural practices. The prevalence of HCV infection was found to be lower than the 15-20% reported in Egypt by Imoru et al. [23] and the 1.0-1.9 percent for countries like the United States of America, Australia, Turkey, Spain, Italy, and Japan, but comparable to reports of 0.1-1.0 percent for the United Kingdom and Scandinavia. The prevalence of hepatitis B and C varies by nation and is impacted by a complex interplay of host, environmental, and behavioral factors. It is higher in nations or regions with lower socioeconomic levels and lowest in those with higher living standards.
In this study, the seroprevalence of syphilis was 1.33%. The prevalence of syphilis among potential blood donors varied, according to reports from earlier researchers. According to a 2001 study conducted in Georgia, 2.3% of blood donors had syphilis [24]. According to a study conducted in North Western Ethiopia, 1.2% of blood donors had syphilis [25]. Compared to 17.6% of volunteer blood donors, 56.6% of commercial and 53.6% of family replacement donors had syphilis and other blood-borne infections [26]. Nigerians with HIV were found to have a 2.0% syphilis sero-positivity and a 14.0% sero-positivity, according to Umeke et al. [10]. While researching syphilis among voluntary blood donors in North Eastern Nigeria, Olokoba et al. [27] found that 1.7% of blood donors had syphilis seropositivity. Methodological variations as well as regional variations in syphilis prevalence could be the cause of the discrepancy in syphilis prevalence seen in this study compared to other studies. Rapid plasma reagin and T. pallidum hemagglutination assays have been found to be less sensitive than T. pallidum particle agglutination assays [28].
In this study, the seroprevalence of HIV was 1.0%. This prevalence is lower than the 2.8%, 4.6%, and 3.8% reported in comparable Nigerian studies in Kaduna [29], Sokoto [1], and Kano [23], but it is comparable to the 1% recorded in Port Harcourt [16]. The 4.9% reported in Ghana has been surpassed by data from other Sub-Saharan regions [30]. Ethiopia, however, reported lower levels of 0.1 [19]. The results of the numerous intervention programs that have been put in place over the years may be regarded as the reason for the decrease in HIV prevalence that this study found.
People between the ages of 26 and 35 had the highest prevalence of transfusion-transmitted infections (TTIs). Likewise, Babatope et al. [6] discovered that TTIs were more common in this age range (30–39 years). A connection between age and health has also been shown in earlier research. According to Iyevhobu & Obodo [31] and Alli et al [32], the highest rate of HIV and HCV infection is seen in those under 40, and those who are between the ages of 24 and 39 are also the ones who have the highest rates of sexual activity. It makes sense to assume that sexual activity is the main mechanism of transmission in this population, given the age group most impacted in our study. High levels of sexual activity during marriage, before marriage, as a widow or widower, because of conventions and traditions, as a means of settling debt, or as a trade or pleasure are all linked to this age group. In the current study, males had a greater prevalence of HBV (1.11%) than females (0.89%). In addition, the prevalence of syphilis was 0.44% in men and over 1.0% in women. HIV prevalence was higher among females (0.89%) than among males (0.22%). The susceptibility of women to HIV, syphilis, HBV, and HCV infections may be attributed to biological, cultural, and socioeconomic factors. A woman's chance of getting a TTI during unprotected vaginal intercourse is up to four times higher than a man. Transfusion-transmitted infections (TTIs) in young potential blood donors did not significantly differ based on sex in our current investigation (p>0.05). The reason why males are more prone to infection than females is unclear. In order to guarantee that every donated blood is appropriately tested TTIs and that only non-reactive blood and blood components are released for clinical and manufacturing use, blood transfusion services should set up effective procedures [4].
CONCLUSIONS
With the potential for viral transmission to a potential susceptible host, the frequency of HBV (2.00%), HCV (2.67%), HIV (1.0%), and syphilis (1.33%) among potential blood donors in the study area indicates their endemicity and potentially rising profile in young potential blood donors. According to the study, prospective blood donors who were between the ages of 26 and 35 had a greater sero-prevalence of TTIs than those who were male. The high prevalence of TTIs (HBV, HIV, Syphilis, and HCV) among potential blood donors in Ekpoma emphasizes the importance of early disease agent diagnosis in the prevention and control of disease outbreaks. For early identification and treatment, young adults should be encouraged to undergo routine screening for TTIs (HBV, HIV, syphilis, and HCV). It is important to promote safe sexual behavior among potential young male and female blood donors.
CONFLICT OF INTERESTS
The authors have no conflict of interest to declare. The authors declared that this study has received no financial support.
REFERENCES
1.Erhabor O, Yakubu A, Usman I, Abubakar AW, Buhari, H, Okwesili A. The risk of transfusion transmissible hepatitis C infection among blood donors in Sokoto, North Western Nigeria. Asian Journal of Science & Technology. 2015;6(2):1051-7.
2.Busayo KA, Olawale SA, Etuvie FA, Iyevhobu KO, Olaniyan OA, Adebunmi OA, et al. Assessing the Impact of Hepatitis B Immunization among Children Aged 1-14 years in Ogbomoso, Oyo State, Nigeria. World Journal of Advanced Research and Reviews. 2024;22(03):1094-104.
3.Iyoha UJ, Iyevhobu K.O, Ebode NO, Iredia QI, Alao BM, Okodua MA, et al. Assessment of the Prevalence of Hepatitis B Virus Infection among people living with HIV visiting a Tertiary Health Institution in Edo State. International Journal of Scientific and Research Publications. 2024;14(1):61-9.
4.Nwogoh B, Ikpomwen OD, Isoa EM. Donor blood procurement and the risk of transfusion transmissible viral infections in a tertiary health facility in South-South Nigeria. Niger Med J. 2011;52(4):227-9. doi: 10.4103/0300-1652.93793.
5.Mujeeb SA, Aamir K, Mehmood K. Seroprevalence of HBV, HCV and HIV infections among college going first time voluntary blood donors. J Pak Med Assoc. 2006;56(1 Suppl 1):S24-5.
6.Babatope IO, Iyevhobu KO, Orukotan RI, Ukhurebor GO. Seroprevalence of Transfusion – Transmissible Infections (T.T.Is) among prospective blood donors in Ambrose Alli University, Ekpoma, Nigeria. North American Academic Research. 2022;5(10):142-53. doi: 10.5281/zenodo.7392373
7.Diro E, Alemu S, G/Yohannes A. Blood safety & prevalence of transfussion transmissible viral infections among donors at the Red Cross Blood Bank in Gondar University Hospital. Ethiop Med J. 2008;46(1):7-13.
8.Ugbaja SC, Omerigwe SA, Ndlovu SMZ, Ngcobo M, Gqaleni N. Evaluating the Efficacy of Repurposed Antiretrovirals in Hepatitis B Virus Treatment: A Narrative Review of the Pros and Cons. Int J Mol Sci. 2025;26(3):925. doi: 10.3390/ijms26030925.
9.Zaheer H, Saeed U, Waheed Y, Karimi S, Waheed U. Prevalence and trends of hepatitis B, hepatitis C and human immunodeficiency viruses among blood donors in Islamabad, Pakistan 2005-2013. Journal of Blood Disorders & Transfusion. 2014;217(5):222-6.
10.Uneke CJ, Ogbu O, Inyama PU, Anyanwu GI, Njoku MO, Idoko JH. Prevalence of hepatitis-B surface antigen among blood donors and human immunodeficiency virus-infected patients in Jos, Nigeria. Mem Inst Oswaldo Cruz. 2005;100(1):13-6. doi: 10.1590/s0074-02762005000100002.
11.Fessehaye N, Naik D, Fessehaye T. Transfusion transmitted infections - a retrospective analysis from the National Blood Transfusion Service in Eritrea. Pan Afr Med J. 2011;9:40. doi: 10.4314/pamj.v9i1.71219.
12.Screening Donated Blood for Transfusion-Transmissible Infections: Recommendations. Geneva: World Health Organization; 2009.
13.Chiavetta JA, Escobar M, Newman A, He Y, Driezen P, Deeks S, et al. Incidence and estimated rates of residual risk for HIV, hepatitis C, hepatitis B and human T-cell lymphotropic viruses in blood donors in Canada, 1990-2000. CMAJ. 2003;169(8):767-73.
14.Joint United Nations Programme on HIV/AIDS (UNAIDS)-WHO. Revised recommendations for the selection and use of HIV antibody tests. Wkly Epidemiol Rec. 1997;72(12):81-7.
15.Khan ZT, Asim S, Tariq Z, Ehsan MA, Malik RA, Ashfaq B, et al.. Prevalence of transfusion transmitted infectious in healthy blood donors in Rawalpindi District, Pakistan: a five-year survey. International Journal of Pathology. 2017;5(1):21-5.
16.Ejele OA, Ojule AC. The prevalence of hepatitis B surface antigen (HBsAg) among prospective blood donors and patients in Port Harcourt, Nigeria. Niger J Med. 2004;13(4):336-8.
17.O'Brien SF, Yi QL, Fan W, Scalia V, Kleinman SH, Vamvakas EC. Current incidence and estimated residual risk of transfusion-transmitted infections in donations made to Canadian Blood Services. Transfusion. 2007;47(2):316-25. doi: 10.1111/j.1537-2995.2007.01108.x.
18.Nagalo MB, Sanou M, Bisseye C, Kaboré MI, Nebie YK, Kienou K, et al. Seroprevalence of human immunodeficiency virus, hepatitis B and C viruses and syphilis among blood donors in Koudougou (Burkina Faso) in 2009. Blood Transfus. 2011;9(4):419-24. doi: 10.2450/2011.0112-10.
19.Mohammed Y, Bekele A. Seroprevalence of transfusion transmitted infection among blood donors at Jijiga blood bank, Eastern Ethiopia: retrospective 4 years study. BMC Res Notes. 2016;9:129. doi: 10.1186/s13104-016-1925-6.
20.Nwankwo E, Momodu I, Umar I, Musa, B, Adeleke S. Seroprevalence of major blood-borne infections among blood donors in Kano, Nigeria. Turkish Journal of Medicine. 2019;42 (5):337-41.
21.Okoroiwu HU, Okafor IM, Asemota EA, Okpokam DC. Seroprevalence of transfusion-transmissible infections (HBV, HCV, syphilis and HIV) among prospective blood donors in a tertiary health care facility in Calabar, Nigeria; an eleven years evaluation. BMC Public Health. 2018;18(1):645. doi: 10.1186/s12889-018-5555-x.
22.Olayinka AT, Oyemakinde A, Balogun MS, Ajudua A, Nguku P, Aderinola M, et al. Seroprevalence of Hepatitis B Infection in Nigeria: A National Survey. Am J Trop Med Hyg. 2016;95(4):902-7. doi: 10.4269/ajtmh.15-0874.
23.Imoru M, Eke C, Adegoke A. Prevalence of hepatitis B surface antigen (HBsAg), hepatitis C virus (HCV) and human immunodeficiency virus (HIV) among blood donors in Kano state, Nigeria. Journal of Medical Laboratory Science. 2013;12(5):59-63.
24.Butsashvili M, Tsertsvadze T, McNutt LA, Kamkamidze G, Gvetadze R, Badridze N. Prevalence of hepatitis B, hepatitis C, syphilis and HIV in Georgian blood donors. Eur J Epidemiol. 2001;17(7):693-5. doi: 10.1023/a:1015566132757.
25.Azene D, Bayah A, Fissaba M. Sero-prevalence of major blood Borne Infections Among Blood Donors at Felege Hiwott Referal Hospital, North Western Ethopia. Ethopian Journal of Health Science. 2017;21(7):68-9.
26.Osime OE, Ogho OM. Prevalence of Syphilis. An Emerging public Health Problem among Blood Donors. Annals of Biomedical Science. 2019;8(1):73-9.
27.Olokoba AB, Olokoba LB, Salawu FK. Syphilis in Voluntary Blood Donors in North Eastern Nigeria. European Journal of Science Research. 2019;31(1):335-40.
28.Tessema B, Yismaw G, Kassu A, Amsalu A, Mulu A, Emmrich F, et al. Seroprevalence of HIV, HBV, HCV and syphilis infections among blood donors at Gondar University Teaching Hospital, Northwest Ethiopia: declining trends over a period of five years. BMC Infect Dis. 2010;10:111. doi: 10.1186/1471-2334-10-111.
29.Hassan A, Muktar HM, Mamman AI, Ahmed AJ, Isa AH, Babadoko AA. The incidence of HIV among blood donors in Kaduna, Nigeria. Afr Health Sci. 2008;8(1):60.
30.Walana W, Ahiaba S, Hokey P, Vicar EK, Acquah SEK., Der EM. Seroprevalence of HIV, HBV and HCV among blood donors in Kintampo municipal hospitals, Ghana. British Microbiology Research Journal. 2014;4(12);1491-9.
31.Iyevhobu KO, Obodo BN. Prevalence of Parasitic Infections in Relation to CD4+ and Antiretroviral (ART) Usage of HIV Sero-Positive Patients Attending Irrua Specialist Teaching Hospital (ISTH) Irrua, Edo State, Nigeria. Research and Reviews Journal of Microbiology and Biotechnology. 2020;1(2):34-41.
32.Alli JA, Okonko OA, Abraham AF, Kolade PN, Ogunjobi AO, Salako MO. Survey of blood parasites (Plasmodium, HIV, HBsAG, HCV Antibodies) in Prospective Nigerian Blood Donors. Research Journal of Medical Science. 2019;4(4):255-75.
Notas de autor
kennylamai@yahoo.com
Información adicional
redalyc-journal-id: 6920
