Originales
Report of presumptive Perkinsus sp. hypnospores in Megapitaria squalida of the Gulf of California with the thioglycollate staining technique
Reporte de presuntas hipnosporas de Perkinsus sp., en Megapitaria squalida del Golfo de California con la técnica de tinción con tioglicolato
Report of presumptive Perkinsus sp. hypnospores in Megapitaria squalida of the Gulf of California with the thioglycollate staining technique
Revista MVZ Córdoba, vol. 25, no. 2, p. 1805, 2020
Universidad de Córdoba

This work is licensed under Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International.
Received: 05 August 2019
Accepted: 06 January 2020
Published: 07 May 2020
Abstract: Objective. To detect the presence of presumptive hypnospores of the protozoan Perkinsus sp. in a wild population of the Mexican chocolata clam Megapitaria squalida in the southeastern Gulf of California, using Ray´s fluid thioglycollate medium (RFTM). Material and Methods. Thirty specimens with size between 56.17 and 69.04 mm were captured each month, during an annual cycle. Infection prevalence and intensity and water parameters were recorded monthly from September 2012 to September 2013. Results.Perkinsus sp. was detected in tissue samples from the Mexican chocolate clam using the RFTM test by the presence of dark round corpuscles that represent parasite´s hypnospores. Monthly samplings revealed a prevalence of 0-43.33% and an infection intensity ranging from 1 to 4 (no infection = 0 hypnospores/entire preparation, to moderate = 34 hypnospores/entire preparation). Conclusions.Perkinsus sp. is reported for the first time in a wild population of M. squalida in the southesternmost Gulf of California. The results indicate that this protozoan is dispersed intraspecifically and would now, potentially, parasiting a new host in the region.
Keywords: Parasitology, bivalves, protozoa, prevalence, Sinaloa, Mexico.
Resumen: Objetivo. Detectar la presencia de presuntas hipnosporas del protozoario Perkinsus sp. en una población silvestre de la almeja chocolata mexicana (Megapitaria squalida) del sureste del Golfo de California, usando el medio fluido de tioglicolato de Ray (RFTM). Materiales y métodos. Cada mes durante un ciclo anual, se capturaron 30 especímenes con una longitud entre 56.17 y 69.04 mm. La prevalencia e intensidad de la infección y los parámetros del agua se registraron mensualmente desde septiembre 2012 a septiembre 2013. Resultados. Se detectó la presencia de presuntas hipnosporas dePerkinsus sp. en muestras de tejido de la almeja chocolata mexicana usando la prueba RFTM, por la presencia de corpúsculos redondos y oscuros que representan hipnosporas del parásito. Los muestreos mensuales revelaron un rango de prevalencia e intensidad de la infección de 0-43.33% y 1-4 (infección negativa = 0 hipnosporas/preparación, a moderada = 34 hipnosporas/preparación), respectivamente. Conclusiones.Perkinsus sp. es reportado por primera vez en una población silvestre de M. squalida en la parte más al sureste del Golfo de California. Los resultados indican que el parásito está disperso intraespecíficamente y, potencialmente, parasitaría un nuevo huésped en la región.
Palabras clave: Parasitología, bivalvos, protozoario, prevalencia, Sinaloa, México.
INTRODUCTION
Several Perkinsus-like organisms are involved in the infection and mortality of wild and cultivated bivalve mollusks causing the disease known as dermo or perkinsosis, which has been reported in different countries and in different species. In oysters, for example, Perkinsus beihaiensis was identified in the tissues of the Pacific cupped Crassostrearhizophorae and the Brazilian oyster Crassostrea brasiliana with high prevalence of the parasite (1). Also in South America, the infection of Perkinsus marinus and Perkinsus olseni in Crassostrea gasar .C. brasiliana) using molecular analysis was recorded (2,3).
Pagenkopp-Lohan et al (4) investigated the distribution of tropical parasites in Panama finding P. marinus infecting C. rhizophorae and Crassostrea virginica in the Atlantic Ocean, and Crassostrea columbiensis on the Pacific coast. The presence of Perkinsus-like organisms in various mollusks from the Great Barrier Reef was recorded in Australia (5), while Perkinsusmediterraneus was detected parasitizing the European flat oyster (Ostrea edulis) (6). For just over two decades, wild populations and cultivated stocks of the eastern oyster (Crassostrea virginica) in the southeastern USA (7) and in the Gulf of Mexico and the Caribbean Sea in Mexico (8), have been affected resulting in mortalities linked to the presence of P. marinus. Meanwhile, in the Pacific Ocean and Gulf of California, P. marinus has been associated with losses in Japanese oyster (Crassostreagigas) production on commercial farms (9).
Protists of the Perkinsus genus are intracellular parasites that infect the bivalve mollusk hemocytes, whose free life is characteristic by the presence of biflagellated zoospores; while in its vegetative form, trophozoites multiply intra or extracellularly within the host. When the trophozoites mature, they divide rapidly to form a hypnospora that subsequently releases biflagellated zoospores (10). So their life cycle allows them to be easily dispersed in the water. Due to its dispersal and transmission capacity among mollusks of different taxonomic groups (11), several species of Perkinsus spp. have been found in clams as well (12). For example, Perkinsusquwadi was linked with mortalities in the Japanese clam Patinopecten yessoensis cultivated in Canada (13), Perkinsus honshuensis was discovered in tissue samples from the Manila clam (Ruditapesphilippinarum) (14).
On the other hand, the presence of Perkinsus chesapeaki in the thin shell clams Mya arenaria and Tagelus plebeius was reported in Chesapeake Bay in the mid-Atlantic USA (15), and the information of perkinsosis in the warty venus clams (Venus verrucosa), the variegated scallop (Chamys varia) and the common cockle (Cerastoderma edule) was updated at new sites on the northwest Mediterranean coast (16). In the coast of Sonora, Mexico, Perkinsusmarinus was identified in the smooth venus clam (Chione fluctifraga) using the staining technique based on thioglycollate medium (17). But so far, there are no reports on the presence of this parasite in clams from the Sinaloa’s coast, which include the Mexican chocolata clam (Megapitaria squalida). To detect the presence of presumptive Perkinsus sp. hypnospores in the tissue of M. squalida in a wild population in the southeasternmost Gulf of California, using Ray’s fluid thioglycollate medium (RFTM), represent the aim of this study.
MATERIALS AND METHODS
Collection site. Clams specimens were collected from Altata Bay (24° 20’-24° 40’ N and 107° 30’-108° 00’ W) on the central coastal line of Sinaloa, Mexico, from September 2012 to September 2013 (Figure 1). Thirty specimens (63.04 ± 6.8 mm) were captured each month by free diving and transported to the laboratory in a 30-L tank containing seawater. At each sampling, the water temperature (°C) and salinity (‰) were recorded.
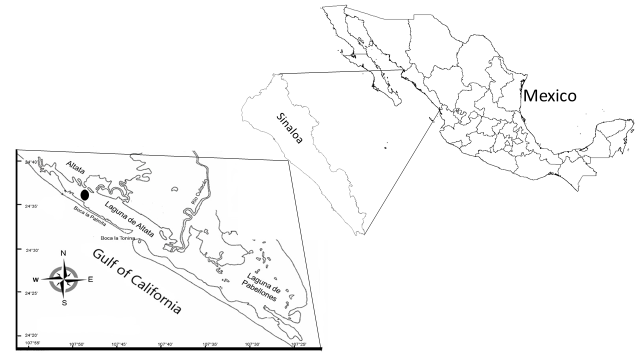
Figure 1
Mexico map indicating Sinaloa state and sampling site • at Altata Bay
Students from the Laboratorio de Malacología, at Instituto Politécnico Nacional-Centro Interdisciplinario de Investigación para el Desarrollo, Integral Regional (IPN-CIIDIR), Unidad Sinaloa, collected and transported the clams following the standard procedures (NOM-031-SSA1-1993, Bienes y Servicios. Productos de la Pesca. Moluscos bivalvos frescos-refrigerados y congelados). This research was approved by the Ethic Committee (College of Teachers) at the IPN-CIIDIR.
Clams processing. The gills, mantle, and digestive gland were removed from each clam to be incubated in RFTM (25°C and seven days at dark conditions), according to the standard specifications (18). Subsequently, they were macerated, stained with Lugol solution and left to rest for 10 minutes before being observed under the microscope (10X and 40X) to detect Perkinsus sp. hypnospores.
Infection analysis. Each month, the prevalence (% of clams that presented presumptive hypnospores) was calculated. Also monthly, the infection intensity (number of presumptive hypnospores observed/entire preparation) was calculated for specimens that were positive for the presence of the parasite with RFTM, and classified based on the five levels of the Mackin’s scale (19): 1 = negative (0 hypnospores), 2=very light (1 to 10 hypnospores/entire preparation), 3=light (11 to 30 hypnospores/entire preparation), 4=moderate (31 to 100 hypnospores/entire preparation), and 5=heavy (>101 hypnospores/entire preparation).
Statistics. Appropriate statistical analyses were applied after examining the normality of the data (Lilliefors). Each month, ANOVA and Tukey test were performed on the infection intensity. The correlations between the infection prevalence and intensity and the water temperature and salinity also were evaluated monthly. All statistical tests were analyzed with the software Statgraphic Plus 5.0; the significance level was set at 95%.
RESULTS
Perkinsus sp. was detected in tissue samples from the Mexican chocolate clam using the RFTM test, based on the presence of presumptive hypnospores (Figure 2). According to the Diagnostic Manual for Aquatic Animals (18), these dark round corpuscles represent vegetative stages of the protozoan.
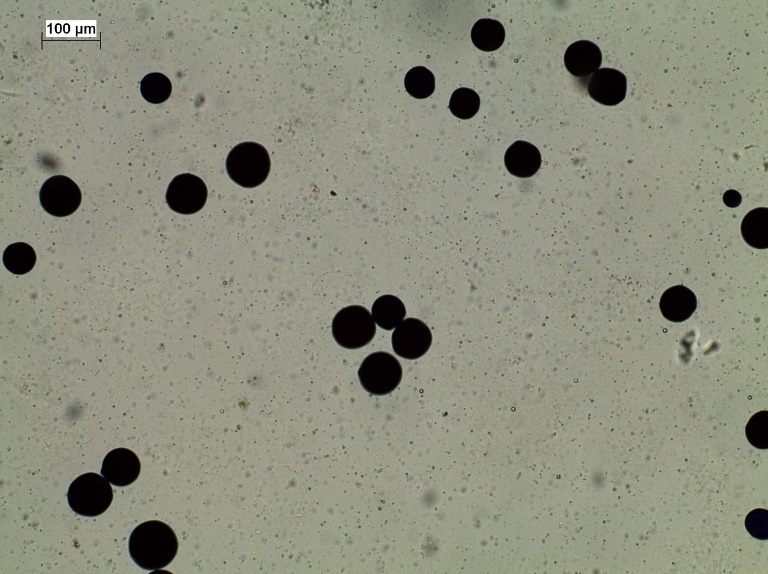
Figure 2
Detection of presumptive Perkinsus sp hipnospores in tissue of Megapitariasqualida by means of the MFTR staining technique 40X Infection intensity in level 2 Mackin´s scale Bar scale 100 μm
The water temperature at the sampling site fluctuated from 16.9°C (January 2013) to 37°C (July 2013), while the salinity varied from 29‰ in October 2012 to 40‰ in April 2013. The monthly prevalence of presumptive hypnospores in M. squalida showed significant differences (F=2.78, p=0.004) and fluctuated from 0% in November 2012, to 43.3% in March and June 2013 (Figure 3), when the water temperature increased without reaching the maximum gradient.
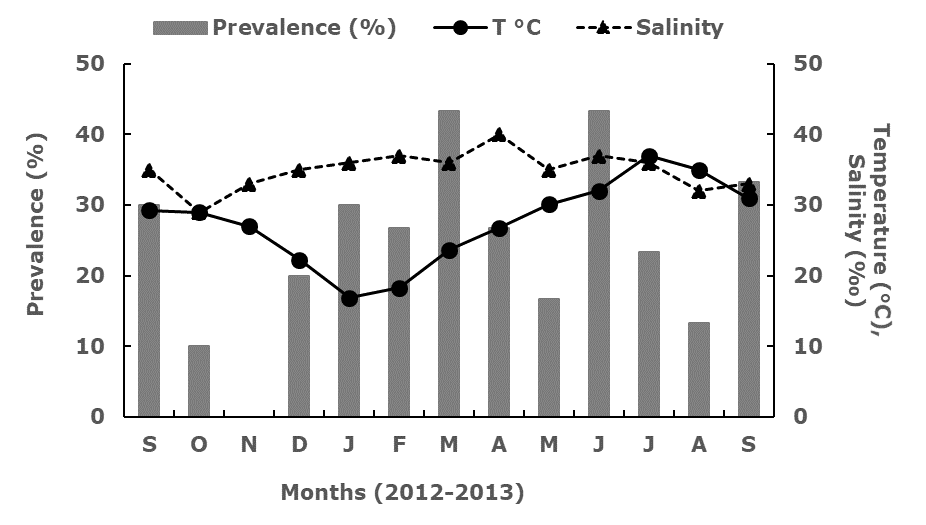
Figure 3.
Prevalence (%) of presumptive Perkinsus sp. hipnospores in Megapitaria squalida detected with MFTR, and temperature (°C) and salinity (‰) in Altata Bay (Sinaloa, Mexico), from September 2012 to September 2013.
The infection intensity varied from 1 to 3 (Mackin’s scale) (19), as the number of hypnospores observed per entire preparation ranged from 0 (November 2012) to 34 (February 2013) (Table 1).
The correlations between the parameters studied and the infection indexes are shown in table 2. Only salinity correlated with prevalence (r=0.56, p=0.04).
| Sep 2012 | Oct | Nov | Dec | Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep 2013 | |
| M | 4.9abc | 2.7abc | ND | 3.0abc | 11.5d | 7.0cd | 5.5bc | 6.5c | 4.4abc | 5.5bc | 1.2ab | 1.5abc | 1.3a |
| SD | 3.4 | 1.1 | ND | 1.6 | 8.7 | 1.1 | 3.8 | 3.3 | 1.7 | 4.0 | 0.7 | 1.0 | 0.4 |
| MML | 2-10 | 2-4 | ND | 2-6 | 2-24 | 2-34 | 1-14 | 2-12 | 2-8 | 2-12 | 1-3 | 1-3 | 1-2 |
| N | 9 | 3 | 0 | 6 | 9 | 8 | 13 | 8 | 5 | 13 | 7 | 4 | 10 |
| M = mean; SD = standard deviation; MML = minimum and maximum limit; N = number of observations; ND = not determined. Different superscript letters show statistical differences; ANOVA, F=2.78, p=0.004. | |||||||||||||
| Sep 2012 | Oct | Nov | Dec | Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep 2013 | |
| M | 4.9abc | 2.7abc | ND | 3.0abc | 11.5d | 7.0cd | 5.5bc | 6.5c | 4.4abc | 5.5bc | 1.2ab | 1.5abc | 1.3a |
| SD | 3.4 | 1.1 | ND | 1.6 | 8.7 | 1.1 | 3.8 | 3.3 | 1.7 | 4.0 | 0.7 | 1.0 | 0.4 |
| MML | 2-10 | 2-4 | ND | 2-6 | 2-24 | 2-34 | 1-14 | 2-12 | 2-8 | 2-12 | 1-3 | 1-3 | 1-2 |
| N | 9 | 3 | 0 | 6 | 9 | 8 | 13 | 8 | 5 | 13 | 7 | 4 | 10 |
| M = mean; SD = standard deviation; MML = minimum and maximum limit; N = number of observations; ND = not determined. Different superscript letters show statistical differences; ANOVA, F=2.78, p=0.004. | |||||||||||||
DISCUSSION
Among all the environmental factors, temperature and salinity are recognized as the most important influencing the infectious expression of Perkinsus spp. in different species of mollusks. Together with the density and type of substrate, the aforementioned environmental factors determined the prevalence and intensity of P. olseni infection in Ruditapes philippinarum from 24 localities in Korea (20). Similar observations were documented for the grooved carpet shell (Ruditapes decussatus) and the Japanese carpet shell (Ruditapes philippinarum) in the northeast Atlantic and the Mediterranean (21). Although some temperatures recorded in the present study were within the optimum range for sporulation of the protozoan (24 to 28°C) (22), the prevalence and infection intensity were not correlated with this parameter. On the other hand, the highest prevalence (43.33%) occurred when salinity exceeded 35‰ (i.e., between March and July).
The climate of the Altata-Ensenada de Pabellones lagoon system, where Altata bay is located, is characterized by being hot with temperatures that vary annually from 19 to 35°C; with rains from June to October and dry from November to May. During the dry season the salinity exceeds 30‰, while 0‰ can be registered in the rainy season, which together with the influence of drains derived from agricultural activity in the area could affect the infectious effect of Perkinsus sp in the callista clam. It is accepted that the prevalence of perkinsosis in wild populations of mollusks increases at high salinities as part of the infection dynamics (10). Although the salinity in March and July were above the optimum maximum limit for the formation of protozoan spores (35‰), as it was proved in vitro (22), it seems that the high prevalence obtained was more the result of the combination of high salinity and temperature, than the sole action of salinity, since the temperature also increased from 23.7 to 37°C during those months. The moderate infection intensity obtained during the 13 months of sampling in Altata Bay, suggests that the seasonal variation of these two parameters did not potentiate the infectious effect of Perkinsus sp. in M. squalida.
Due to its sensitivity, simplicity, and low cost (18,23), the use of the RFTM technique in the identification of presumptive Perkinsus spp. hypnospores is considered a reliable preliminary method for carrying out subsequent molecular assays in processed tissues. The detection of dark round spheres in M. squalida, characteristic of presumptive Perkinsus sp. hypnosporas using RFTM, confirmed the presence of this vegetative stage of the parasite in the clam tissue with an infection intensity ranging from negative to moderate. Whitish nodules (20) or aqueous tissues (21) may indicate injuries caused by the protozoan; however, no tissue damage was observed. Some authors mention that this may be due to a low infection intensity (17), the small size of the tissue sample processed with this technique (24), and/or hypnospores found outside the tissues in the outer layer of the mantle that were incorporated into the stained sample during processing. Only one of 95 clams with positive presence by RFTM had more than 30 hypnospores per entire preparation analyzed, reflecting a low level of infection.
Presumptive Perkinsus sp. hypnospores are reported for the first time in a wild population of the Mexican chocolata clam (M. squalida) from the southeasternmost Gulf of California detected with RFTM, with a moderate prevalence and an infection intensity ranging from negative to moderate, apparently without compromising the health of the clam since no tissue damage was observed visually. For clams in the Gulf of California, there is only one report on the detection of Perkinsus marinus using the RFTM technique in a cultivated population of the smooth venus (Chione fluctifraga) (17). Specifically, the detection of Perkinsus marinus in the bivalves of the Gulf of California has focused primarily on different oyster species (Crassostrea gigas, C. corteziensis, and Saccostrea palmula) due to their commercial importance (10,25,26,27). Considering the species confirmation (P. marinus) and its high incidence in the region, it is possible to assume that the Perkinsus species in this study is the same one that has already been dispersed in different localities and in several species of non-ostreid bivalves. It was detected Perkinsus sp. in the maura pen shell (Atrinamaura) from a locality a few kilometers north of the study area of the present work (28). This reinforces the argument that the parasite in question has been dispersed intraspecifically and has found a new bivalve hosts in the southeastern Gulf of California, thus, the Mexican chocolata clam should be considered in the catalog of infected species with Perkinsus sp. (18).
Regarding the Perkinsus sp. parasitization of M. squalida in the region, it is necessary to carry out more studies on the pathology, host-host interaction, patterns of infection and epidemiology using RFTM together with other parasite detection-confirmation techniques (histology, PCR, genetic sequencing, and phylogenetic analysis), in order to clarify the current health status of this bivalve and implement a permanent monitoring program.
Conflict of interests
The authors declare that there are no conflicts of interest of any kind in the realization and elaboration of this work.
Acknowledgments
The authors deeply thank the logistical and finantial support to the Instituto Politécnico Nacional (IPN), the Secretaría de Investigación y Posgrado (SIP-IPN), the Comisión de Operaciones y Fomento de Actividades Académicas (COFFA-IPN), and the Estímulo para el Desempeño de los Investigadores (EDI-IPN), for the realization of the present study through the proyects: SIP-IPN 20120471 and SIP-IPN 20130858.
REFERENCES
1. Luz MSA, Carvalho FS, Oliveira HC, Boehs G. Perkinsus beihaiensis (Perkinsozoa) in oysters of Bahia State, Brazil. Braz J Biol. 2018; 78(2):289-295. https://doi.org/10.1590/1519-6984.07016
2. Da Silva PM, Scardua MP, Vianna RT, Mendonça RC, Vieira CB, Dungan, et al. Two Perkinsus spp. infect Crassostrea gasar oysters from cultured and wild populations of the Rio São Francisco Estuary, Sergipe, northeastern Brazil. J Invert Pathol. 2014; 119:62-71. https://doi.org/10.1016/j.jip.2014.04.005
3. Queiroga FR, Vianna RT, Vieira CB, Farias ND, da Silva PM. Parasites infecting the cultured oyster Crassostrea gasar (Adanson, 1757) in northeast Brazil. Parasitology. 2015; 142(6):756-766. https://doi.org/10.1017/S0031182014001863
4. Pagenkopp-Logan KM, Hill-Spanik KM, Torchin ME, Aguirre-Macedo L, Fleisher RC, Ruíz GM. Richness and distribution of tropical oyster parasites in two oceans. Parasitology. 2016; 143(9):1119-32. https://doi.org/10.1017/S0031182015001900
5. Waki T, Yoshinaga T. Experimental challenges of juvenile and adult Manila clams with protozoan Perkinsus olseni at different temperature. Fish Sci. 2013; 79:779–786. https://doi.org/10.1007/s12562-013-0651-4
6. Valencia JM, Bassitta M, Picornell A, Ramon C, Castro JA. New data on Perkinsus mediterraneus in the Balearic Archipelago: locations and affected species. Dis Aquat Org. 2014; 112:69-82. https://doi.org/10.3345/dao02795
7. Ford SE, Tripp MR. The Eastern oyster: Crassostrea virginica. 1st. Ed. Maryland (USA): College Park, MD: Maryland Sea Grant College; 1996. https://www.worldcat.org/title/eastern-oyster-crassostrea-virginica/oclc/35164271
8. Huicab-Pech ZG, Curiel-Ramírez S, Castañeda-Chávez M, Lango-Reynoso F, Carrillo-Alejandro P. Variación estacional de Perkinsus marinus en el ostión americano Crassostrea virginica del sistema lagunar Carmen-Machona-Pajonal en Tabasco, México. Trop Subtrop Agroecosys. 2012; 15(Sup 2):S40-S50. http://www.revista.ccba.uady.mx/ojs/index.php/TSA/article/view/1743
9. Enríquez-Espinoza TL, Grijalva-Chon JM, Castro-Longoria R, Ramos-Paredes J. Perkinsus marinus in Crassostrea gigas from the Gulf of California. Dis Aquat Org. 2010, 89:269-273. https://doi.org/10.3354/dao02199
10. Villalba A, Reece KS, Ordás MC, Casas SM, Figueras A. Perkinsosis in molluscs: a review. Aquat Liv Res. 2004; 17:411-432. https://doi.org/10.1051/alr:2004050
11. Percher WT, Alavi MR, Schott EJ, Fernández-Robledo JA, Roth L, et al. Assessment of the northern distribution range of selected Perkinsus species in Eastern oyster (Crassostrea virginica) and hard clams (Mercenaria mercenaria) with the use of PCR-base detection assays. J Parasitol. 2008, 94:410-422. https://doi.org/10.1645/GE-1282.1
12. Choi K-S, Waki T. Perkinsus olseni (Lester and Davis 1981) infection in the Manila clam (Ruditapes philippinarum) in Korea: species identification, impacts and spatio-temporal distribution. Bull Jap Fish Res Edu Agency. 2016, 42:23-27. https://www.fra.affrc.go.jp/bulletin/bull/bull42/42-06.pdf
13. Itoh N, Meyer GR, Tabata A, Lowe G, Abbott CL, Johnson SC. Rediscovery of the Yesso scallop pathogen Perkinsus qugwadi in Canada, and development of PCR tests. Dis Aquat Org. 2013; 104:83-91. https://doi/org/10.3354/dao02578
14. Arzul I, Chollet B, Michel J, Robert M, Garcia C, Joly J-P, et al. One Perkinsus species may hide another: characterization of Perkinsus species present in clam production areas of France. Parasitology. 2012; 139(13):1575-1771. https://doi/org/10.1017/S0031182012001047
15. Glasspie CN, Seitz RD, Ogburn MB, Dungan CF, Hines AH. Impacts of predators, hábitat, recruitment, and disease on soft-shell clams Mya arenaria and stout razor clams Tagelus plebeius in Chesapeake Bay. Biorxiv. 2017; 22:1-55. https://www.biorxiv.org/content/10.1101/224071v2
16. Ramilo A, Carrasco N, Reece KS, Valencia JM, Grau A, et al. Update of information on perkinsosis in NW Mediterranean coast: identification of Perkinsus spp. (Protista) in new locations and hosts. J Invert Pathol. 2015; 125:37-41. https://doi.org/10.1016/j.jip.2014.12.008
17. Enríquez-Espinoza TL, Castro-Longoria R, Mendoza-Cano F, Grijalva-Chon JM. Perkinsus marinus in Crassostrea gigas and Chione fluctifraga from Kino Bay, Sonora, Mexico. Biotecnia. 2015; 17(1):10-13. https://biotecnia.unison.mx/index.php/biotecnia/article/view/6
18. OIE. Manual of diagnostic tests for aquatic animals 2018. World Organization of Animal Health. 2019. Available at: http://www.oie.int/international-standard-setting/aquatic-manual/access-online/
19. Mackin JG. Oyster disease caused by Dermocystidium marinum and other microorganisms in Louisiana. Institute for Marine Science University of Texas. USA. 1962; 7:132-229. https://repositories.lib.utexas.edu/handle/2152/22811
20. Sang H-S, Yang H-S, Reece KS, Cho Y-G, Lee H-M, Kim C-W, et al. Survey on Perkinsus species in Manila clam Ruditapes philippinarum in Korea waters using species-specific PCR. Fish Pathol. 2017; 52(4):202-205. https://doi.org/10.3147/jsfp.52.202
21. Ruano F, Batista FM, Arcangeli G. Perkinsosis in the clams Ruditapes decussatus and R. philippinarum in the Northeastern Atlantic and Mediterranean Sea: A review. J Invert Pathol. 2015; 131:58-67. https://doi.org/10.1016/j.jip.2015.07.015
22. Auzoux-Bordenave S, Vigário AM, Ruano F, Domart-Coulon I, Doumenc D. In vitro sporulation of the clam pathogen Perkinsus atlanticus (Apicomplexa, Perkinsea) under various environmental conditions. J Shellfish Res. 1995; 14:469-475. https://archive.org/details/cbarchive_37449_invitrosporulationoftheclampat1995/page/n2
23. Auderman C, Carnegie RB, Burreson EM. Shellfish tissues evaluated for Perkinsus spp. using the Ray’s fluid thioglycollate medium culture assay can be used for downstream molecular assays. Dis Aquat Org. 2008; 80:235-239. https://doi.org/10.3354/dao01944
24. Villanueva-Fonseca LC, Escobedo-Bonilla CM. Prevalencia del protozoario Perkinsus sp. en un cultivo de ostión japonés Crassostrea gigas en Sinaloa, México. Lat Amer J Aquat Res. 2013; 41(5):996-1002. https://doi.org/103856/vol41-issue5-fulltext-19
25. Cáceres-Martínez J, Vásquez-Yeomans R, Padilla-Lardizábal G, del Río-Portilla MA. Perkinsus marinus in pleasure oyster Crassostrea corteziensis from Nayarit, Pacific Coast of Mexico. J Invert Pathol. 2008; 99(1):66-73. https://doi.org/10.1016/j.jip.2008.03.005
26. Cáceres-Martínez J, Vásquez-Yoemans R, Padilla-Lardizábal G. Parasites and symbionts of the pleasure oyster Crassostrea corteziensis cultured in Nayarit, México. J Aquat Anim Health. 2010; 22:141-151. https://doi.org/10.1577/H09-052.1
27. Cáceres-Martínez J, García-Ortega AM, Vásquez-Yeomans R, Pineda-García TJ, Stokes NA, Carnegie RB. Natural and cultured populations of the mangrove oyster Saccostrea palmula from Sinaloa, Mexico, infected by Perkinsus marinus. J Invert Pathol. 2012; 110(3):321-325. https://doi.org/10.1016/j.jip.2012.03.019
28. Góngora-Gómez AM, Rubio-Zepeda F, Villanueva-Fonseca LC, Álvarez-Dagnino E, Muñoz-Sevilla N., Hernández-Sepúlveda JA, et al. Primer registro de Perkinsus sp. (Protozoa, Apicomplexa) en el callo de hacha Atrina maura en Sinaloa, México. Revista de Biología Marina y Oceanografía. 2016; 51(3):689-694. https://doi.org/10.4067/S0718-1957201600030002
Additional information
How to cite (Vancouver).: óngora-Gómez AM, Villanueva-Fonseca LC, Sandoval-Rivera P, Hernández-Sepúlveda JA, Domínguez-Orozco AL, Villanueva-Fonseca BP, García-Ulloa M. Report of presumptive Perkinsus sp. hypnospores in Megapitaria squalida of the Gulf of California with the thioglycollate staining technique. Rev MVZ Cordoba. 2020; 25(2):e1805. https://doi.org/10.21897/rmvz.1805
