Revisión de Literatura
Environmental factors related to Influenza A virus occurrence in wild birds
Factores ambientales relacionados con la presentación de virus influenza A en aves silvestres
Environmental factors related to Influenza A virus occurrence in wild birds
Revista MVZ Córdoba, vol. 25, no. 2, pp. 1-19, 2020
Universidad de Córdoba

This work is licensed under Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International.
Received: 04 November 2019
Accepted: 06 April 2020
Published: 08 June 2020
Abstract: Research and interest in avian influenza virus have increased considerably in recent decades in response to highly pathogenic avian influenza outbreaks in poultry and its zoonotic potential. Wild waterfowl are the main reservoir of the virus, therefore studying the dynamics of influenza A virus (IAV) infection in these populations is essential in order to understand its potential persistence in the environment and transmission to poultry and humans. It has been identified that environmental factors (such as temperature, rainfall, vegetation and landscape characteristics, among others) can play an important role in the maintenance and dissemination of the virus in the areas of concentration of wild birds. However, studies that include ecological aspects of the virus and explore the interaction between the prevalence of IAV in wild birds and environmental factors remain scarce. This review summarizes research efforts that have been made to identify the environmental factors involved in the persistence and transmission of IAV in areas of wild bird concentration and how these factors may influence the prevalence of the virus in these populations, generating differences in the presentation of the infection among different geographical areas.
Keywords: Orthomyxovirus, bird disease, environment.
Palabras clave: Orthomyxovirus, enfermedad de las aves, medio ambiente
INTRODUCTION
The influenza A virus (IAV) is considered a worldwide threat to both animal and public health, having caused the four largest human pandemics in the last hundred years (1): The “Spanish Influenza” in 1918 was one of the most destructive pandemics of humanity caused by a H1N1 subtype, the “Asian Influenza” in 1957 caused by an H2N2 subtype, the “Hong Kong Influenza” caused by a H3N2 subtype and the “Pandemic Influenza of 2009” which arose from an H1N1 subtype with a combination of genes from three different strains (avian H1N1, seasonal human H3N2 and swine H1N1 from North America and Asia) (2).
Since the beginning highly pathogenic avian influenza (HPAI) epidemic in 1997 caused by the H5N1 subtype in Hong Kong, more than 60 countries belonging to the 5 continents have been affected (Figure 1). In addition, the number of strains reported worldwide has increased alarmingly with the emergence of new HPAI variants H5N8 in East Asia and Europe, the H5N6 subtype in Asia and the introduction of an H5Nx virus of Eurasian origin in North America, all belonging to the new clade 2.3.4.4 (3). Furthermore, the emergence of new subtypes affecting humans (such as pandemic H1N1 and avian H7N9 introduced into the human population in Asia) highlights the need to address this disease from a comprehensive approach, understanding the ecological and environmental factors that affect host-pathogen interactions and that may influence virus presentation patterns.
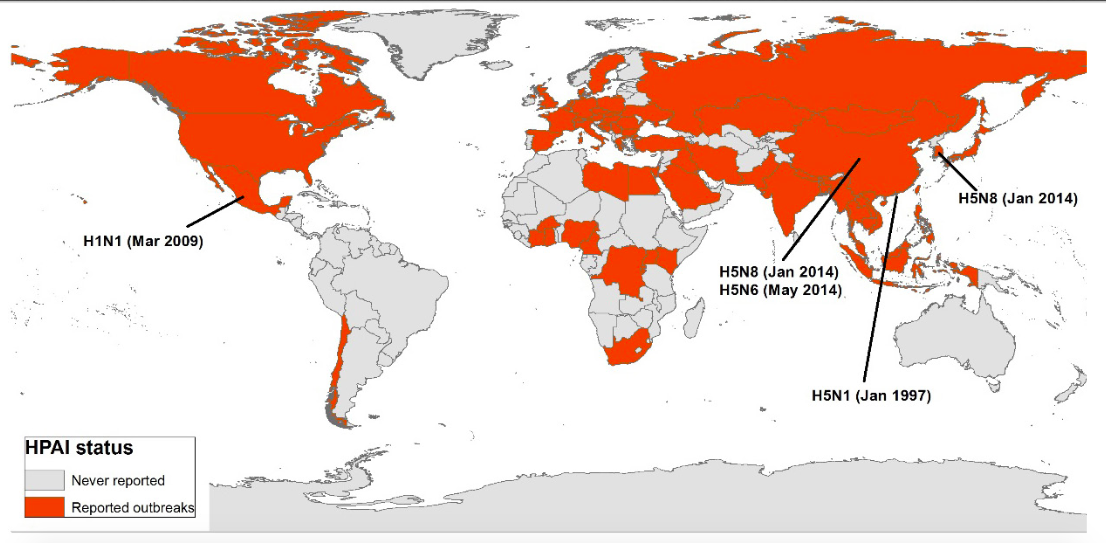
Figure 1.
Countries affected by highly pathogenic avian influenza virus (IAAP) from 1997 to date and places of emergency of the main subtypes of influenza A virus.
The influenza virus can be classified in two groups according to its clinical presentation in poultry. HPAI viruses present multi-basic cleavage sites, replicating in multiple tissues causing systemic infection and high mortality. So far, only subtypes H5 and H7 have been associated with highly pathogenic presentations. On the other hand, low pathogenic avian influenza viruses (LPAI) present a monobasic cleavage site, therefore, they are restricted to replicate in respiratory and gastrointestinal tissues, causing a mild disease. However, it is described that LPAI strains may mutate to a HPAI strain after a period of circulation in poultry populations, causing severe disease with high mortality and morbidity (4).
Wild waterfowl have a key role in the IAV ecology because they are recognized as natural reservoirs of most of the subtypes described in the form of LPAI (5), having the potential to disseminate it when they migrate within and between continents, representing a risk for the emergence of HPAI outbreaks in domestic birds (6). Therefore, the epidemiology, prevalence and diversity of IAV are closely related to the behavior of these reservoir species, including their feeding, habitats and migratory patterns, among others (7). Environmental factors, such as temperature, rainfall, amount of vegetation and water characteristics, may also play a role in the maintenance and spread of the virus in areas of concentration of wild birds (8,9,10,11).
Few studies have investigated the ecological and environmental factors that affect the prevalence of the influenza virus in wild birds, mainly in the northern hemisphere (12,13,14,15) and in some regions of Africa (11,16,17).
The objective of this review is to summarize the research efforts that have been made to identify the main environmental factors that affect the persistence and transmission of the IAV in wild waterfowl and how these factors may influence the prevalence of the virus in these populations, generating differences in the presentation of the infection among different geographical areas.
Dynamics of LPAI virus infection in wild birds
In wild waterfowl, especially those belonging to the orders Anseriformes (ducks, geese, swans) and Charadriiformes (shorebirds, gulls, terns), all influenza virus subtypes described have been isolated, with the exception of subtypes H17N10 and H18N11, only described in bats to date (18).
In these natural hosts, the IAV replicates preferentially in the gastrointestinal tract, excreting in high concentrations through the faeces. Therefore, circulation of LPAI virus in wild bird populations depends on transmission between these reservoir species and susceptible hosts, mainly through the faecal-oral route (5). However, the prevalence and dynamics of LPAI virus infection in wild birds may vary depending on geographic location, season and bird species (7).
In North America and Europe, prevalence of IAV in waterfowl is greater when many birds of different ages and origins congregate at their breeding sites, and where there are also many immunologically naïve young individuals. In these areas, prevalence can reach 30% during late summer and early autumn, prior to migration, followed by a decline during winter (7,12). In contrast, in the southern hemisphere, although the dynamics of virus presentation has been scarcely studied, research in Australia and Africa has shown that there is no seasonality, suggesting that the factors influencing infection dynamics in southern hemisphere may differ from those in the northern hemisphere (11,19,20).
In tropical Africa, for example, the prevalence of IAV infection in wild birds is low but constant throughout the year (11,17) which could be explained by the fact that the breeding period of waterfowl is more extended than in the northern hemisphere, with immunologically immature individuals throughout the year, which could perpetuate the infection (11). This situation is different from that found in an IAV surveillance study of wild birds conducted in Chile between 2012 and 2015, where a difference in prevalence was observed between seasons, being greater during the summer/autumn months than in winter/spring (21).
Although these geographic differences can be attributed to factors related to hosts, such as: species present, feeding behavior, habitat use, migratory pattern, population size and seasonal congregation among others; environmental conditions and landscape structure in different geographic contexts can play a preponderant role in the dynamics of virus infection in these populations (11,22).
According to research carried out using mathematical models, the persistence of the virus in aquatic environments is fundamental for the maintenance and circulation of the virus in wild birds (23,24,25). This is because infected birds eliminate large concentrations of virions to the environment through faeces, which can survive in water for long periods of time, maintaining the infection within the population through the faecal-oral transmission route (25). The importance of this transmission route may be influenced by population size. Thus, in large populations, where there is a greater density of individuals and a higher contact rate between them, transmission by direct contact may be more relevant than indirect (environmental) transmission. On the other hand, in small populations, environmental transmission plays a key role in maintaining the virus within the bird community (23).
Therefore, it is important to emphasize the role of the environment as a determining factor in the epidemiology of IAV in wild birds and the need for consideration of ecological and environmental factors in prevalence studies (25).
Persistence of influenza virus in aquatic environments
Among the environmental factors that influence the persistence and circulation of the influenza virus in aquatic ecosystems, both biotic and abiotic elements are included, and surface water plays a fundamental role (8). This has been demonstrated by numerous studies in which it has been determined that the virus can survive in water for long periods of time (Table 1).
| Study | Year | Country | Persistence of VIA according to different parameters | |||
| T° | pH | Salinity | Others | |||
| Webster et al. | 1978 | USA | - 4 days at 22°C - More than 30 days at 0°C | --- | --- | --- |
| Stallknecht et al | 1990 | USA | -100 days at 17°C -9 days at 28°C | -100 days at pH 8.2 | -100 days at 0 parts per trillion (ppt) -9 days at 20 ppt | --- |
| Brownet al | 2009 | USA | More stable at low temperatures (<17°C) | More stable at a basic pH (7.4–8.2) | More stable between 0–20000 parts per million (ppm) | --- |
| Nazir et al | 2010 | Germany | -Days between 30 y 20°C -Weeks at 10°C - Months between 0 y -10°C | --- | --- | Distilled water: Longer persistence (up to 642 days) Normal saline solution: Less persistence (up to 321 days) Surface water: lowest persistence (up to 55 days) |
| Lebarbenchon et al. | 2011 | USA | Stable between 17 y 23°C | Stable at pH 7.2 | --- | --- |
| Keeler et al | 2013 | USA | All viruses were tested at 17°C | --- | --- | Distilled water: Longer persistence (more than 80 days) Filtered surface water: Less persistence (up to 60 days) Intact surface water: lowest persistence (less than 20 days) |
| Keeler et al | 2014 | USA | High viability at low temperatures (<17°C) | Stable between 7.0- 8.5 | Stable at 0.5 ppt | Stable at low ammonia concentrations (<0.5 mg/L) |
In 1978, Webster et al (26) published the first information related to IAV persistence in aquatic environments using an A/Duck/Memphis/546/74 (H3N2) virus, proving that it can remain infective in lake waters for up to 4 days at 22°C and more than 30 days at 0°C. Subsequent studies, conducted under laboratory conditions, have shown that the persistence of the virus in water can vary widely depending on the viral strain and the characteristics of the water (temperature, pH and salinity) (9,27).
These studies, using a modified distilled water model, determined that low temperatures (4 to 17°C), neutral to basic pH (7.0-8.5) and low salinity (<20000 ppm) favour the persistence of the IAV in water, while acidic conditions (pH <6.6), high temperatures (>32°C) and high salinity (>25000 ppm) significantly reduce it. Subsequently, Lebarbenchon et al (28) found similar results when evaluating two viral subtypes (H4N6 and H3N8) isolated from ducks in the United States.
Both subtypes persisted for several months in distilled water at constant temperatures (between 17 and 23°C) and at neutral pH. Another study by Nazir et al (29), evaluated the persistence of three LPAI virus strains (H4N6, H5N1, and H6N8) under different temperatures and in waters with different physicochemical properties (distilled water, normal saline, and surface water obtained from Constance Lake). Virus persistence was greater in distilled water, followed by normal saline water. Virus strains had a lower persistence in surface water. In addition, results showed that virus survival time was inversely proportional to temperature, remaining infectious for a few days at temperatures between 20 and 30°C and for months at temperatures between -10 and 0°C. This situation is consistent with that reported by Keeler et al (30), where the persistence of the virus was greater in distilled water (more than 80 days) and notably less in intact surface water (less than 20 days).
It is important to note that all the above studies were performed under laboratory conditions, using modified distilled water to obtain different physicochemical parameters. A study that more closely replicate natural water bodies properties was performed by Keeler et al (31). In this study, surface water samples collected from 38 waterfowl habitats in the United States were used to evaluate the persistence of different virus subtypes. Results agree to those described above and also demonstrating that low concentrations of ammonia (<0.5 mg/L) favor the persistence of the virus. Shoham et al (32), also evaluated the persistence of several IAV subtypes in artificially frozen environmental water, most of which were stable over a year at -30 and -20°C, suggesting that arctic regions could represent an important environmental reservoir of the virus for wild bird populations.
However, since there are other variables in natural water bodies that can interact with those mentioned above, it is important to implement future studies under field conditions, considering factors such as: temperature variations between day and night; water flow and depth; UV exposure; turbidity; adherence of IAV to organic and inorganic substrates present in the water; and biological diversity (bacteria, biofilms, filtering organisms and other invertebrates) among others (8,33). Biological diversity has been considered relevant in some studies, since it has been determined that the biotic community present in the water would have an important role in the concentration and/or inactivation of the virus in the environment (34).
Effect of environmental factors on IAV prevalence in wild birds.
Empirical research on environmental and ecological factors that influence the dynamics of IAV presentation in wild birds is still in full development (10,11,13,14). Studies conducted so far agree on those factors that could have an important role on the prevalence of IAV in ecosystems, within which abiotic elements are described, such as rainfall and temperature; biotics, such as bird species present and amount of vegetation; and anthropogenic factors, such as land use and type of land cover (15,19,22).
Temperature, rainfall and vegetative cover measured by the Normalized Differential Vegetation Index (NDVI) have been identified as key environmental factors related to the occurrence of HPAI H5N1 outbreaks in Europe (10). The NDVI is the most widely used index for remote detection of vegetation, which estimates the amount of photosynthetic mass from the intensity of radiation from certain bands of the electromagnetic spectrum emitted by vegetation. Elevated NDVI values, low precipitation and an increase in minimum temperatures during the cold season were shown to be related to outbreaks of HPAI. An increase in NDVI in combination with an increase in minimum temperatures during the winter provides greater food availability and shelter for birds in cold seasons, increasing bird congregation and viral spread (10).
Something similar was observed in Africa and the Middle East, where the occurrence of HPAI H5N1 has been associated with high levels of NDVI, indicating that the spatial distribution of HPAI H5N1 cases in these areas would be related to specific environmental characteristics, generating an “environmental fingerprint” for the presentation of the virus (35).
Research in North America also indicates temperature as an important factor influencing LPAI virus prevalence in wild birds, associating a higher probability of infection with lower temperatures (15,36,37). This situation is consistent with a predictive model carried out by Herrick et al (38), where temperature and precipitation were identified as important predictors for IAV outbreaks in wild birds. Regions with low annual precipitation and low temperatures were indicated as the primary environmental niche for IAV outbreaks in wild birds, emphasizing the importance of boreal regions in the epidemiology of the virus. In Mediterranean climate regions, low temperatures and low rainfall have also been associated with a higher prevalence of IAVs in wild birds (13).
Conversely, research in Australia and tropical regions of Africa indicates that temperature would not have a major influence on IAV prevalence in wild birds, suggesting that there are other factors influencing virus presentation in these regions (11,16,19). Gaidet et al (11) used a set of wetland data from 15 African countries to assess the influence of a wide range of ecological factors on IAV prevalence in wild birds. This study found that variations in prevalence were related to host ecological factors rather than temperature conditions, being positively associated with the arrival of migrant birds from Eurasia and with the density of birds in wetlands.
Climate variables (temperature and humidity), on the other hand, were poorly related to prevalence. This differs from that found in boreal regions, since Afro-tropical regions are characterized by temperatures greater than or equal to 20°C throughout the year, which is not favourable for the maintenance of the virus in the environment, suggesting that in these areas transmission by direct contact would have greater relevance than environmental transmission for the maintenance of infection in populations (16).
These results are similar to others found on the African continent, where an increase in prevalence was related to the arrival season of migrant birds (17,39). In addition, Fuller et al (39) also determined that rainy seasons would be associated with an increase in the prevalence of IAVs in wetlands in Central Africa. This is probably due to two mechanisms: [1] that rainfall triggers an increase in food availability and hence a greater abundance of birds and [2] that the onset of the rainy season coincides with the onset of the bird breeding period, favouring congregation at sites by increasing the rate of contact.
Based on these results, authors suggest that rainfall determines a breeding opportunity, influencing the age structure within the population (greater number of juveniles), which affects the dynamics of infection of the IAV (39). This is consistent with the findings of Van Dijk et al (12), who demonstrated that the peak prevalence of IAV in collared ducks (Anas platyrhynchos) in the Netherlands was associated with an increase in susceptible young individuals during late summer/early autumn. This time coincides with the time of arrival of migrants, providing strong evidence of the role that migratory birds would have as disseminators in seasonal epizootics of LPAI viruses in wild birds.
In a study conducted in south-eastern Australia (19), the prevalence of IAVs in wild duck faeces samples and their relationship to biotic (number of birds) and abiotic (climatic variables) factors was assessed. These results also demonstrated a positive effect of rainfall on prevalence, both immediately and in the long term as a result of the southern child oscillation phenomenon (ENSO). Temperature, unlike in the northern hemisphere, did not have a significant effect on the data, nor did the number of birds present at the time of sampling, indicating that the dynamics of the IAV is not simply a function of the number of birds in the wetland, but a combination of many factors involved.
Another investigation under field conditions corresponds to a study carry on in Spain in which the influence of various environmental factors (climatic conditions, density and diversity of birds, physical-chemical properties of water and vegetation structure) on the dynamics of IAV in various wetlands was evaluated (13). In this study, explanatory variables were grouped into factors. Results determined that the combination of spatial and temporal variables (sampling location, year and sampling period) may mostly explain the variation in IAV prevalence over the study period (36.8%), followed by meteorological variables (21.5%) and finally the combination of the composition of the wild bird community (Anseriformes density and species richness) with the vegetation structure (21.1%).
Although these results are similar to those found in other regions of the northern hemisphere , it is important to note that none of the variables included in the factor “physical-chemical characteristics of water” were retained in the final model, probably because under field conditions there are other variables such as the effect of dilution and other biotic factors, which could have been potentially more important than the degree of variation of the physical properties evaluated (8).
Anthropogenic factors related to agricultural land use have also been indicated as important factors affecting IAV presentation. Studies have already determined that there is a strong association between HPAI H5N1 outbreaks and rice-intensive areas in Asia, due to a large congregation of domestic ducks foraging in rice paddies, corresponding to a critical factor in the persistence and spread of HPAI on that continent (40,41,42). Something similar was found in Madagascar, where serological data of poultry were combined with environmental factors obtained by remote sensing.
In this study, LPAI virus seroprevalence was higher on farms adjacent to rice fields and wetlands than on those surrounded by savanna landscape, reaffirming the role of ecosystems composed of rice fields and high densities of ducks in the epidemiology of the virus (43). In the same way, the use of agricultural land close to wetlands would favor the aggregation of wild birds in search of food, increasing the rate of contact between individuals, creating ideal conditions for the transmission of the virus (10). This was previously determined in a study conducted in the United States in which a spatial risk map of IAV in wild birds showed that the amount of land cultivated in each county had a significant relationship with the prevalence of IAV found in birds (36).
In addition, land use change resulting from intensive agriculture has led to fragmentation and loss of wetlands, altering wild bird populations. Many bird populations have decreased, while others have adapted to landscapes altered by humans, causing a greater density of birds in the sites, stress and proximity to domestic birds, favouring the transmission of the virus within flocks and between different species (44).
In addition, climate change also plays an important role in causing significant alterations in the ecology of the IAV. An increase in temperatures could potentially affect bird migration patterns, creating new species assemblages and new opportunities for viral transport and reassortment of viral genes (45). In addition, climate change could modify the survival of the virus in the environment. Water temperature and its physicochemical characteristics (pH and salinity) may also be altered, resulting in increased viral persistence, pathogenicity and transmissibility (44,45).
However, research models that include the interaction of several variables (such as, survival of the virus in the environment, change in land use, existence of smaller wetlands, greater density of birds and increased contact rates, among others) are needed in order to predict the impacts of climate change on the ecology of IAV (44,45). Improved understanding of climatic influences on virus ecology will be critical to improve IAV prevention and surveillance strategies at the global level.
In conclusion, despite the considerable effort made worldwide for the surveillance of VIA in wild birds, the existing information about the ecology of the virus and the environmental factors that favor its presentation in these populations, remains very scarce.
Research has focused primarily on the identification and characterization of subtypes, focusing on survival and viral transmission under laboratory conditions or through mathematical models. However, few studies have delved into relationship between the pattern of occurrence and environmental factors under field conditions. In addition, this empirical knowledge is derived mainly from studies conducted in the northern hemisphere and in some regions of Africa, therefore, it could not be extrapolated directly to regions of the southern hemisphere, where there are differences in the hosts, in climatic factors and the seasonality.
To date, there are no studies in South America that explore ecological and environmental factors related to the presentation of IAVs in wild birds, despite the fact that a wide diversity of subtypes circulating in the region have been described, both of North American origin and lineages that are unique to South America (21,46,47,48). Therefore, it would be interesting to generate information regarding the dynamics of virus presentation and the environmental factors involved at the local level. For this, it is necessary to improve field level research and conduct prevalence studies that include ecological and environmental factors in different geographical contexts. This would better assess the risk associated with virus transmission from wildlife reservoirs to domestic animals, improve surveillance programs, and direct sampling efforts to habitats and seasons that are more favourable for viral maintenance and transmission.
Acknowledgments
This research was supported by ANID Grant N°21150579 to SRP, Fondecyt Grant 1191747 to CHW and Fondecyt Grant 11190755 to PJB.
REFERENCES
1. Taubenberger JK, Morens DM. Pandemic influenza - including a risk assessment of H5N1. OIE Rev Sci Tech. 2009; 28(1):187–202. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2720801
2. Smith GJD, Vijaykrishna D, Bahl J, Lycett SJ, Worobey M, Pybus OG, et al. Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza a epidemic. Nature. 2009; 459(7250):1122–1125. https://www.nature.com/articles/nature08182
3. De Vries E, Guo H, Dai M, Rottier PJM, Van Kuppeveld FJM, De Haan CAM. Rapid emergence of highly pathogenic avian influenza subtypes from a subtype H5N1 hemagglutinin variant. Emerg Infect Dis. 2015; 21(5):842–846. https://www.ncbi.nlm.nih.gov/pubmed/25897518
4. Capua I, Alexander DJ. Avian influenza infection in birds: A challenge and opportunity for the poultry veterinarian. Poult Sci. 2009; 88(4):842–846. https://academic.oup.com/ps/article-lookup/doi/10.3382/ps.2008-00289
5. Yoon SW, Webby RJ, Webster RG. Evolution and ecology of influenza a viruses. In: Current Topics in Microbiology and Immunology. Springer Verlag; 2014. https://doi.org/10.1007/82_2014_396
6. Lee D-H, Torchetti MK, Winker K, Ip HS, Song C-S, Swayne DE. Intercontinental Spread of Asian-Origin H5N8 to North America through Beringia by Migratory Birds. J Virol. 2015; 89(12):6521–6524. https://jvi.asm.org/content/89/12/6521.short
7. Munster VJ, Fouchier RAM. Avian influenza virus: Of virus and bird ecology. Vaccine. 2009; 27(45):6340–6444. https://www.sciencedirect.com/science/article/pii/S0264410X09003740
8. Stallknecht DE, Goekjian VH, Wilcox BR, Poulson RL, Brown JD. Avian Influenza Virus in Aquatic Habitats: What Do We Need to Learn? Avian Dis Dig. 2010; 5(s1):e99–100. https://www.aaapjournals.info/doi/abs/10.1637/8760-033109-Reg.1
9. Brown JD, Goekjian G, Poulson R, Valeika S, Stallknecht DE. Avian influenza virus in water: Infectivity is dependent on pH, salinity and temperature. Vet Microbiol. 2009; 136(1–2):20–26. https://www.sciencedirect.com/science/article/pii/S0378113508004884
10. Si Y, Wang T, Skidmore AK, de Boer WF, Li L, Prins HHT. Environmental factors influencing the spread of the highly pathogenic avian influenza H5N1 virus in wild birds in Europe. Ecol Soc. 2010; 15(3):26. https://www.ecologyandsociety.org/vol15/iss3/art26/
11. Gaidet N, Caron A, Cappelle J, Cumming GS, Balança G, Hammoumi S, et al. Understanding the ecological drivers of avian influenza virus infection in wildfowl: A continental-scale study across Africa. Proc R Soc B Biol Sci. 2012; 279(1731):1131–1141. https://royalsocietypublishing.org/doi/full/10.1098/rspb.2011.1417
12. Dijk JGB van, Hoye BJ, Verhagen JH, Nolet BA, Fouchier RAM, Klaassen M. Juveniles and migrants as drivers for seasonal epizootics of avian influenza virus. J Anim Ecol. 2014; 83(1):266–275. https://besjournals.onlinelibrary.wiley.com/doi/abs/10.1111/1365-2656.12131
13. Pérez-Ramírez E, Acevedo P, Allepuz A, Gerrikagoitia X, Alba A, Busquets N, et al. Ecological Factors Driving Avian Influenza Virus Dynamics in Spanish Wetland Ecosystems. PLoS One. 2012; 7(11):e46418. https://dx.plos.org/10.1371/journal.pone.0046418
14. Torrontegi O, Alvarez V, Acevedo P, Gerrikagoitia X, Höfle U, Barral M. Long-term avian influenza virus epidemiology in a small Spanish wetland ecosystem is driven by the breeding Anseriformes community. Vet Res. 2019; 50(1):4. https://veterinaryresearch.biomedcentral.com/articles/10.1186/s13567-019-0623-5
15. Farnsworth ML, Miller RS, Pedersen K, Lutman MW, Swafford SR, Riggs PD, et al. Environmental and demographic determinants of avian influenza viruses in waterfowl across the contiguous United States. PLoS One. 2012; 7(3). https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0032729
16. Gaidet N. Ecology of Avian Influenza Virus in Wild Birds in Tropical Africa. Avian Dis. 2016; 60(1s):296–301. https://www.aaapjournals.info/doi/abs/10.1637/11149-051115-Review
17. Caron A, Abolnik C, Mundava J, Gaidet N, Burger CE, Mochotlhoane B, et al. Persistence of low pathogenic avian influenza virus in waterfowl in a southern African ecosystem. Ecohealth.2011; 8(1):109–115. https://link.springer.com/article/10.1007/s10393-010-0356-4
18. Tong S, Zhu X, Li Y, Shi M, Zhang J, Bourgeois M, et al. New World Bats Harbor Diverse Influenza A Viruses. PLoS Pathog. 2013; 9(10). https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1003657
19. Ferenczi M, Beckmann C, Warner S, Loyn R, O’Riley K, Wang X, et al. Avian influenza infection dynamics under variable climatic conditions, viral prevalence is rainfall driven in waterfowl from temperate, south-east Australia. Vet Res. 2016; 47(1):23. http://www.veterinaryresearch.org/content/47/1/23
20. Mundava J, Caron A, Garine Wichatitsky M, Abolnik C, Mundy P, Gaidet N. The role of breeding phenology and aggregation of waterfowl on avian influenza dynamics in southern Africa. Ibis. 2016;158(4):762–775. https://onlinelibrary.wiley.com/doi/abs/10.1111/ibi.12404
21. Jiménez-Bluhm P, Karlsson EA, Freiden P, Sharp B, Di Pillo F, Osorio JE, et al. Wild birds in Chile Harbor diverse avian influenza A viruses. Emerg Microbes Infect. 2018; 7(1):1–4. https://www.tandfonline.com/doi/full/10.1038/s41426-018-0046-9
22. Cumming GS, Abolnik C, Caron A, Gaidet N, Grewar J, Hellard E, et al. A social–ecological approach to landscape epidemiology: geographic variation and avian influenza. Landsc Ecol. 2015; 30(6):963–985. https://link.springer.com/article/10.1007/s10980-015-0182-8
23. Breban R, Drake JM, Stallknecht DE, Rohani P. The Role of Environmental Transmission in Recurrent Avian Influenza Epidemics. PLoS Comput Biol. 2009; 5(4):e1000346. https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1000346
24. Rohani P, Breban R, Stallknecht DE, Drake JM. Environmental transmission of low pathogenicity avian influenza viruses and its implications for pathogen invasion. Proc Natl Acad Sci U S A. 2009; 106(25):10365–10369. https://www.pnas.org/content/106/25/10365.short
25. Roche B, Lebarbenchon C, Gauthier-Clerc M, Chang CM, Thomas F, Renaud F, et al. Water-borne transmission drives avian influenza dynamics in wild birds: The case of the 2005-2006 epidemics in the Camargue area. Infect Genet Evol. 2009; 9(5):800–805. https://www.sciencedirect.com/science/article/pii/S156713480900077X
26. Webster RG, Yakhno M, Hinshaw VS, Bean WJ, Copal Murti K. Intestinal influenza: Replication and characterization of influenza viruses in ducks. Virology. 1978; 84(2):268–278. https://www.sciencedirect.com/science/article/pii/0042682278902477
27. Stallknecht DE, Brown JD. Tenacity of avian influenza viruses. Rev sci tech Off int Epiz. 2009; 28(1):59–67. https://pdfs.semanticscholar.org/4556/0521cf6246e4852e1ccffd0936e9fadc80bc.pdf
28. Lebarbenchon C, Yang M, Keeler SP, Ramakrishnan MA, Brown JD, Stallknecht DE, et al. Viral Replication, Persistence in Water and Genetic Characterization of Two Influenza A Viruses Isolated from Surface Lake Water. PLoS One. 2011; 6(10):e26566. https://dx.plos.org/10.1371/journal.pone.0026566
29. Nazir J, Haumacher R, Ike A, Stumpf P, Böhm R, Marschang RE. Long-Term Study on Tenacity of Avian Influenza Viruses in Water (Distilled Water, Normal Saline, and Surface Water) at Different Temperatures. Avian Dis. 2010; 54(s1):720–724. http://www.bioone.org/doi/abs/10.1637/8754-033109-ResNote.1
30. Keeler SP, Lebarbenchon C, Stallknecht DE. Strain-related variation in the persistence of influenza A virus in three types of water: distilled water, filtered surface water, and intact surface water. Virol J. 2013; 10(1):13. https://virologyj.biomedcentral.com/articles/10.1186/1743-422X-10-13
31. Keeler SP, Dalton MS, Cressler AM, Berghaus RD, Stallknecht DE. Abiotic factors affecting the persistence of avian influenza virus in surface waters of waterfowl habitats. Appl Environ Microbiol. 2014; 80(9):2910–2917. https://aem.asm.org/content/80/9/2910.short
32. Shoham D, Jahangir A, Ruenphet S, Takehara K. Persistence of Avian Influenza Viruses in Various Artificially Frozen Environmental Water Types. Influenza Res Treat. 2012; 2012:1–11. https://www.hindawi.com/journals/irt/2012/912326/abs/
33. Dalziel AE, Delean S, Heinrich S, Cassey P. Persistence of Low Pathogenic Influenza A Virus in Water: A Systematic Review and Quantitative Meta-Analysis. PLoS One 2016; 11(10):e0161929. https://dx.plos.org/10.1371/journal.pone.0161929
34. Meixell BW, Borchardt MA, Spencer SK. Accumulation and inactivation of avian influenza virus by the filter-feeding invertebrate Daphnia magna. Appl Environ Microbiol. 2013; 79(23):7249–7255. https://aem.asm.org/content/79/23/7249.short
35. Williams RA, Peterson AT. Ecology and geography of avian influenza (HPAI H5N1) transmission in the Middle East and northeastern Africa. Int J Health Geogr. 2009; 8(1):47. http://ij-healthgeographics.biomedcentral.com/articles/10.1186/1476-072X-8-47
36. Fuller TL, Saatchi SS, Curd EE, Toffelmier E, Thomassen HA, Buermann W, et al. Mapping the risk of avian influenza in wild birds in the US. BMC Infect Dis. 2010; 10(1):187. https://bmcinfectdis.biomedcentral.com/articles/10.1186/1471-2334-10-187
37. Papp Z, Clark RG, Parmley EJ, Leighton FA, Waldner C, Soos C. The ecology of avian influenza viruses in wild dabbling ducks (Anas spp.) in Canada. PLoS One. 2017; 12(5):e0176297. https://dx.plos.org/10.1371/journal.pone.0176297
38. Herrick KA, Huettmann F, Lindgren MA. A global model of avian influenza prediction in wild birds: the importance of northern regions. Vet Res. 2013; 44(1):42. http://www.veterinaryresearch.org/content/44/1/42
39. Fuller TL, Ducatez MF, Njabo KY, Couacy-Hymann E, Chasar T, Aplogan GL, et al. Seasonality dynamics of avian influenza occurrences in Central and West Africa. bioRxiv. 2014; 007740. https://www.biorxiv.org/content/10.1101/007740v1.abstract
40. Tiensin T, Ahmed SSU, Rojanasthien S, Songserm T, Ratanakorn P, Chaichoun K, et al. Ecologic Risk Factor Investigation of Clusters of Avian Influenza A (H5N1) Virus Infection in Thailand. J Infect Dis. 2009; 199(12):1735–1743. https://academic.oup.com/jid/article-lookup/doi/10.1086/599207
41. Gilbert M, Newman SH, Takekawa JY, Loth L, Biradar C, Prosser DJ, et al. Flying over an infected landscape: Distribution of highly pathogenic avian influenza H5N1 risk in South Asia and satellite tracking of wild waterfowl. Ecohealth. 2010; 7(4):448–458. https://link.springer.com/article/10.1007/s10393-010-0672-8
42. Paul M, Tavornpanich S, Abrial D, Gasqui P, Charras-Garrido M, Thanapongtharm W, et al. Anthropogenic factors and the risk of highly pathogenic avian influenza H5N1: prospects from a spatial-based model. Vet Res. 2010; 41(3):28. http://www.vetres.org/10.1051/vetres/2009076
43. Guerrini L, Paul MC, Leger L, Andriamanivo HR, Maminiaina OF, Jourdan M, et al. Landscape attributes driving avian influenza virus circulation in the Lake Alaotra region of Madagascar. Geospat Health. 2014; 8(2):445–453. https://geospatialhealth.net/index.php/gh/article/view/33
44. Vandegrift KJ, Sokolow SH, Daszak P, Kilpatrick AM. Ecology of avian influenza viruses in a changing world. Ann N Y Acad Sci. 2010; 1195:113–128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2981064/
45. Morin CW, Stoner-Duncan B, Winker K, Scotch M, Hess JJ, Meschke JS, et al. Avian influenza virus ecology and evolution through a climatic lens. Environ Int. 2018;119:241–249. https://www.sciencedirect.com/science/article/pii/S0160412018302629
46. de Araujo J, de Azevedo Júnior SM, Gaidet N, Hurtado RF, Walker D, Thomazelli LM, et al. Avian Influenza Virus (H11N9) in Migratory Shorebirds Wintering in the Amazon Region, Brazil. PLoS One. 2014; 9(10):e110141. http://dx.plos.org/10.1371/journal.pone.0110141
47. Xu K, Ferreri L, Rimondi A, Olivera V, Romano M, Ferreyra H, et al. Isolation and characterization of an H9N2 influenza virus isolated in Argentina. Virus Res. 2012; 168(1–2):41–47. https://www.sciencedirect.com/science/article/abs/pii/S0168170212002079
48. Rimondi A, Xu K, Craig MI, Shao H, Ferreyra H, Rago M V., et al. Phylogenetic Analysis of H6 Influenza Viruses Isolated from Rosy-Billed Pochards (Netta peposaca) in Argentina Reveals the Presence of Different HA Gene Clusters. J Virol. 2011; 85(24):13354–13362. https://jvi.asm.org/content/85/24/13354.short
Additional information
How to cite (Vancouver).: Ruiz PS, Baumberger C, Jimenez-Bluhm P, Marambio V, Salazar C, Hamilton-West C. Environmental factors related to Influenza A virus occurrence in wild birds. Rev MVZ Cordoba. 2020; 25(2):e1845. https://doi.org/10.21897/rmvz.1845
