Case Studies
High-grade well-differentiated neuroendocrine tumour of the cecum diagnosed following incisional hernia repair: a case report
AUKŠTO LAIPSNIO GERAI DIFERENCIJUOTAS AKLOSIOS ŽARNOS NEUROENDOKRININIS NAVIKAS, DIAGNOZUOTAS POOPERACINĖS IŠVARŽOS PLASTIKOS METU: KLINIKINIS ATVEJIS
High-grade well-differentiated neuroendocrine tumour of the cecum diagnosed following incisional hernia repair: a case report
Acta medica Lituanica, vol. 27, núm. 1, pp. 17-24, 2020
Vilniaus Universitetas
Recepción: 19 Abril 2020
Aprobación: 29 Mayo 2020
Abstract: High-grade well-differentiated neuroendocrine tumour tumours (NETs) of gastrointestinal tract are rare; they can arise in any part of the digestive system and usually present in advanced stages. Low incidence and wide heterogeneity in the biological behaviour of such lesions pose a diagnostic and therapeutic challenge. Loco-regional NETs should be resected whenever feasible, potentially with a curative intent. Management of a metastatic disease is often more complex, primarily aimed at the alleviation of symptoms, prevention of further complications, and prolongation of survival. Current literature describing the optimal treatment plan for such patients is sparse and further studies are necessary to enhance our understanding of the disease. Here we present a case of high-grade well-differentiated cecal NET with an associated carcinoid syndrome that was diagnosed following the incisional hernia repair.
Keywords: case report, neuroendocrine tumour tumour, cecum, incisional hernia, carcinoid syndrome.
Summary: Aukšto laipsnio gerai diferencijuoti virškinamojo trak- to neuroendokrininiai (NE) navikai yra reti, auga bet kurioje žarnyno dalyje ir dažniausiai diagnozuojami vėlyvose stadijose. Mažas paplitimas ir didelis navikų biologinis heterogeniškumas lemia, jog ši liga pasižymi sudėtinga diagnostika bei gydymu. Į regioninius limfmazgius išplitę NE navikai turėtų būti radikaliai rezekuojami siekiant išgydyti pacientą, tačiau metasta- zavusios ligos operacinio gydymo tikslai yra kuklesni ir dažnai apsiriboja paliatyviomis priemonėmis. Šiuo metu nėra aiškių gairių, kaip reikėtų gydyti ketvirtos stadijos NE naviką, tad dažnai klinikinėje praktikoje pasikliaujama individualia patirtimi. Čia aprašome aukšto laipsnio gerai diferencijuotą NE naviko klini- kinį atvejį, kuris pasireiškė karcinoidinio sindromo klinika ir buvo diagnozuotas pooperacinės išvaržos plastikos metu.
Keywords: klinikinis atvejis, neuroendokrininis navikas, akloji žarna, pooperacinė išvarža, karcinoidinis sindromas.
INTRODUCTION
Neuroendocrine tumours (NETs) of the gastrointestinal (GI) tract represent a minor fraction of all digestive system malignancies. The age-adjusted annual incidence of gastroenteropancreatic NETs in United States is 3.56 cases per 100,000 inhabitants (1). Unfortunately, more than 50% of GI tract NETs are advanced at initial presentation with a disseminated disease (2). Majority of non-pancreatic GI tract NETs are non-functioning. Clinically, such patients are usually asymptomatic or have non-specific symptoms until late into the disease, when metastases of the liver or other distant organs develop. In about 10% of metastatic non-pancreatic GI tract NETs, an associated carcinoid syndrome might be diagnosed (2, 3). Here we present an unusual case of high-grade well-differentiated cecal NET with an unrecognized carcinoid syndrome that was diagnosed after intraoperative discovery of peritoneal carcinomatosis during the incisional hernia repair.
CASE REPORT
A 64-year-old female with a history of well-controlled hypertension and hysterectomy 20 years earlier, presented to an outpatient clinic due to a twomonth history of progressively enlarging tender hypogastric lump, frequent diarrhoea, abdominal cramping, several episodes of vomiting, and episodic facial flushing. Clinical examination revealed a 8 × 10 cm irreducible incisional hernia (M4W2RO by EHS) (4) in the hypogastric region that developed secondary to the hysterectomy incision, a surgical procedure performed 20 years earlier for the diagnosis of multiple uterine leiomyomas. No other abdominal masses besides the hernia itself were palpable. The patient had an abdominal ultrasound investigation 12 months prior to the presentation at different institutions, which was unrevealing. She underwent an elective incisional hernia repair. During the operation, a hernia sac was identified. It contained greater omentum that was covered by multiple whitish carcinomatous lesions. Numerous rigid nodules were palpable during digital investigation of the peritoneal cavity and external surface of the large intestine. Abnormal tissues were sent for pathological investigation. Incisional hernia was repaired with a primary closure, refraining from mesh placement.
During the immediate post-operative period, the patient underwent extensive investigation to identify the primary tumour. Abdominal ultrasound showed multiple hepatic and mesenteric lesions with a 5 cm diameter exophytic mass in the cecum. Whole body contrast-enhanced CT revealed 4.4 × 3.6 × 4.4 cm-sized cecum tumour with a transmural infiltration and multiple hepatic and peritoneal metastases (Fig. 1). Pathological evaluation of metastatic lesions in H & E sections of the greater omentum showed ill-defined eosinophilic epithelial cells with prominent nuclear atypia forming insular, solid, and trabecular-pattern structures with perineoplastic lymphocytic infiltrate and serosal invasion. Mitotic count was 21 per 10 high power fields (HPF). Immunohistochemical (IHC) analysis of Ki67 proliferative index showed up to 22% neoplastic cells nuclear positivity quantified by digital pathological assessment (Aperio Nuclear V9). Immunopositivity for CDX2, synaptophysin/chromogranin A, serotonin and STTR5 was also identified (Fig. 2). The diagnosis was consistent with metastases of well-differentiated high-grade (G3) neuroendocrine tumour. Following pathological diagnosis, a serum chromogranin A level was assessed, and elevated concentration of 4876.1 ng/mL was found. Abdominal somatostatin-receptor-based SPECT showed the highest signal intensity originating from the cecal mass and moderately increased signal intensity from peritoneal and hepatic masses (Fig. 3).
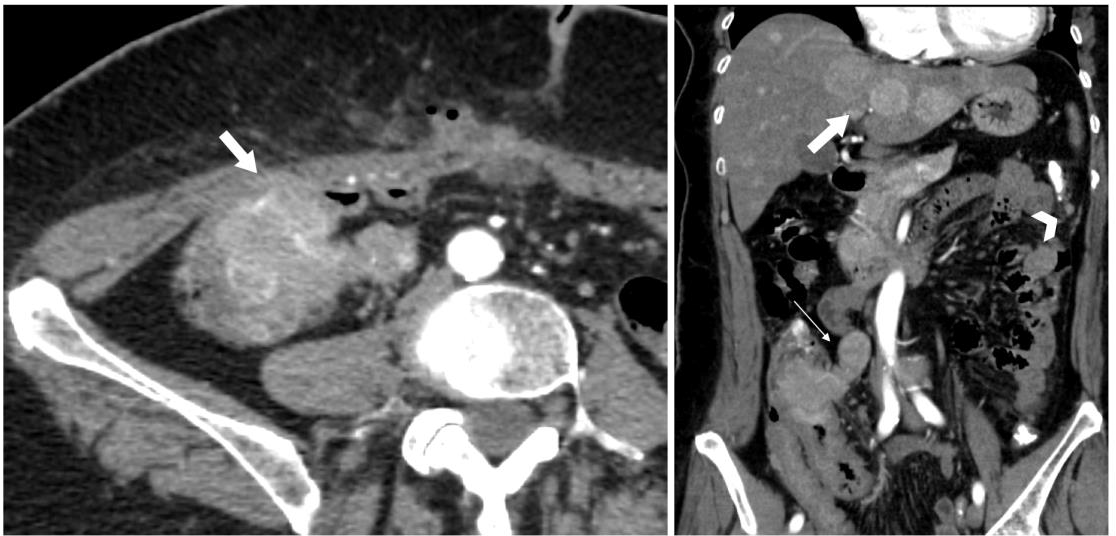
Fig. 1
Cecal neuroendocrine tumour. Left, the transverse contrast-enhanced arterial phase CT image shows a hypervascular mass in the cecum invading the ventral abdominal wall (thick arrow). Note the postoperative air bubbles in the abdominal wall after attempted hernioplasty three days prior. Right, coronal reconstruction of contrast-enhanced arterial phase CT image shows a hypervascular mass in the ileocecal region extending into mesocolon (thin arrow), also multiple hypervascular hepatic (thick arrow) and peritoneal (arrowhead) metastases
After radiological, pathological, and biochemical workup combined with a multidisciplinary team discussion, a decision was made to proceed with a right open hemicolectomy and primary enterocolic anastomosis followed by adjuvant chemotherapy with cisplatin and etoposide. Gross total resection specimen is showed in Fig. 4. Pathological investigation of the primary tumour confirmed the diagnosis of high-grade well-differentiated NET of the cecum (Fig. 5). The post-operative period was uneventful, and the patient was discharged on the sixth postoperative day. Further treatment plan consists of an adjuvant chemotherapy and octreotide to manage carcinoid syndrome symptoms.
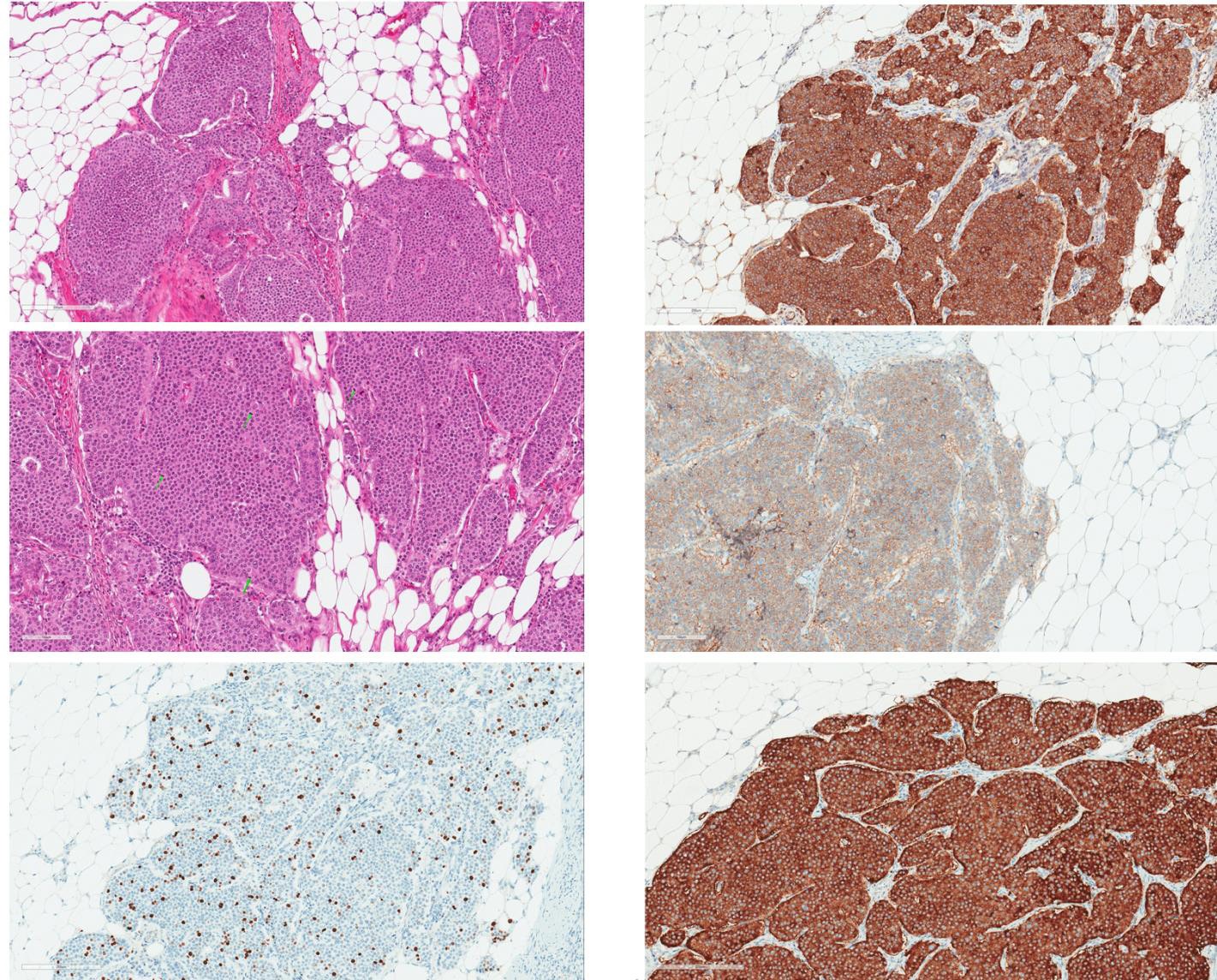
Fig. 2
Microscopic examination of greater omentum metastatic lesions. (a) Alveolar structures of monomorphous round cells with centrally located nuclei and few polymorphous cells distributed randomly. (b) High mitotic ac- tivity with mitotic figures marked by green arrows. (c) Immunohistochemical Ki67 staining. The proliferative index in the metastatic lesion was up to 22% (quantified by digital pathological assessment with Aperio Nuclear V9). (d) Immunohistochemical Chromogranin A staining. The tumour mass shows diffuse cytoplasmic immunopositiv- ity. (e) Weak to moderate membranous staining with STTR5 immunohistochemical reaction. (f) Strongly positive diffuse immunohistochemical reaction for synaptophysin
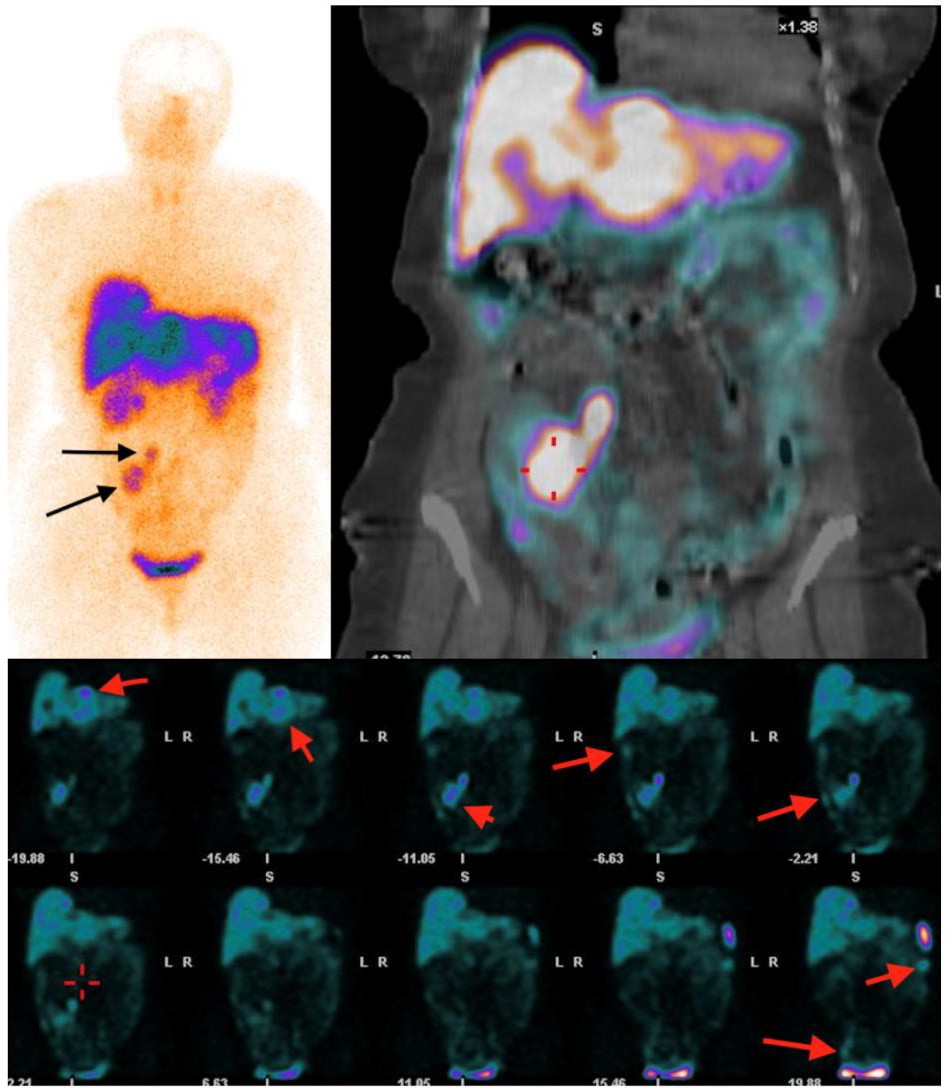
Fig. 3
Top, a planar and SPECT/CT image of SSTR scintigraphy with 99mTc-tectrotyd presenting primary tumour with regional lymph node me- tastases. Bottom, abdominopelvic SPECT/CT images show multiple liver and peritoneal metastases (long arrows) with primary tumour (short arrow)
DISCUSSION
High-grade NETs of GI tract and pancreas consti- tute a heterogenous group of aggressive malignan- cies. Even though pancreatic NETs carry a worse prognosis than GI tract carcinoids, up to 50% of such patients present with a metastatic disease (2).
According to the population-based study investi- gating 64,971 patients with NETs, the annual in- cidence of GI tract NETs in the United States is 3.56 cases per 100,000 inhabitants. The most common sites include the small intestine, the rectum, and the pancreas (1.05, 1.04, and 0.48 per 100,000 individuals, respectively). According to the same study, the cecum is the least common primary lo- cation, accounting for approximately 5% of all GI tract NETs (1).
The clinical course of non-pancreatic GI tract NETs is highly variable. Early diagnosis is tough since majority of patients are asymptomatic or exhibit non-specific symptoms, including weight loss, fatigue, weakness, and/or nausea. More specific symptoms might arise due to increasing tumour mass, distant metastases, and/or polyhormonal hypersecretion. Up to 10% of NETs with hepatic dissemination presents with a carcinoid syndrome. It is characterized by episodic flushing, diarrhea, abdominal pain, vomiting, bronchoconstriction and a carcinoid endocarditis (5). Looking retrospectively, our patient presented with a several carcinoid syndrome symptoms. Anyway, those complaints, namely abdominal cramping and diarrhea, were attributed to the concurrently present incisional hernia.
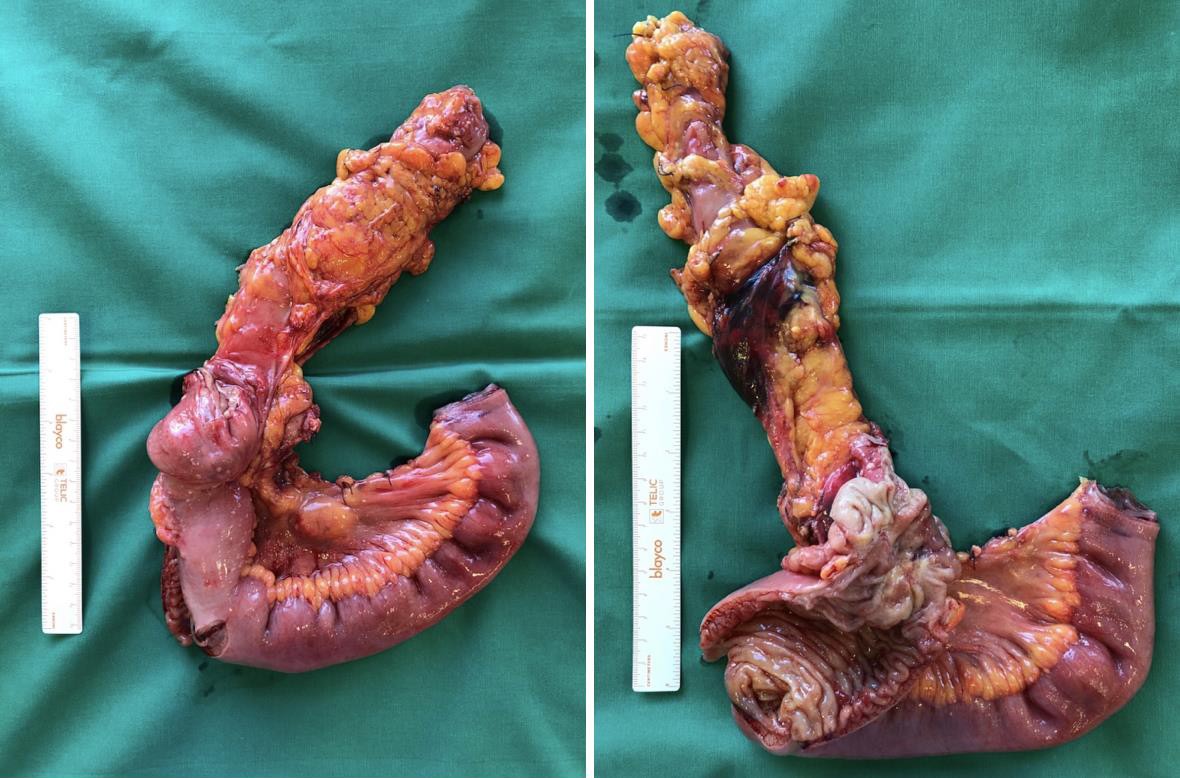
Fig. 4
Gross total resection specimen after right hemicolectomy: a 4 × 4 × 7 cm mass of the cecum infiltrates all of the layers of the large intestine. The tumour extends towards the ascending colon and through the ileocecal valve to reach the distal ileum
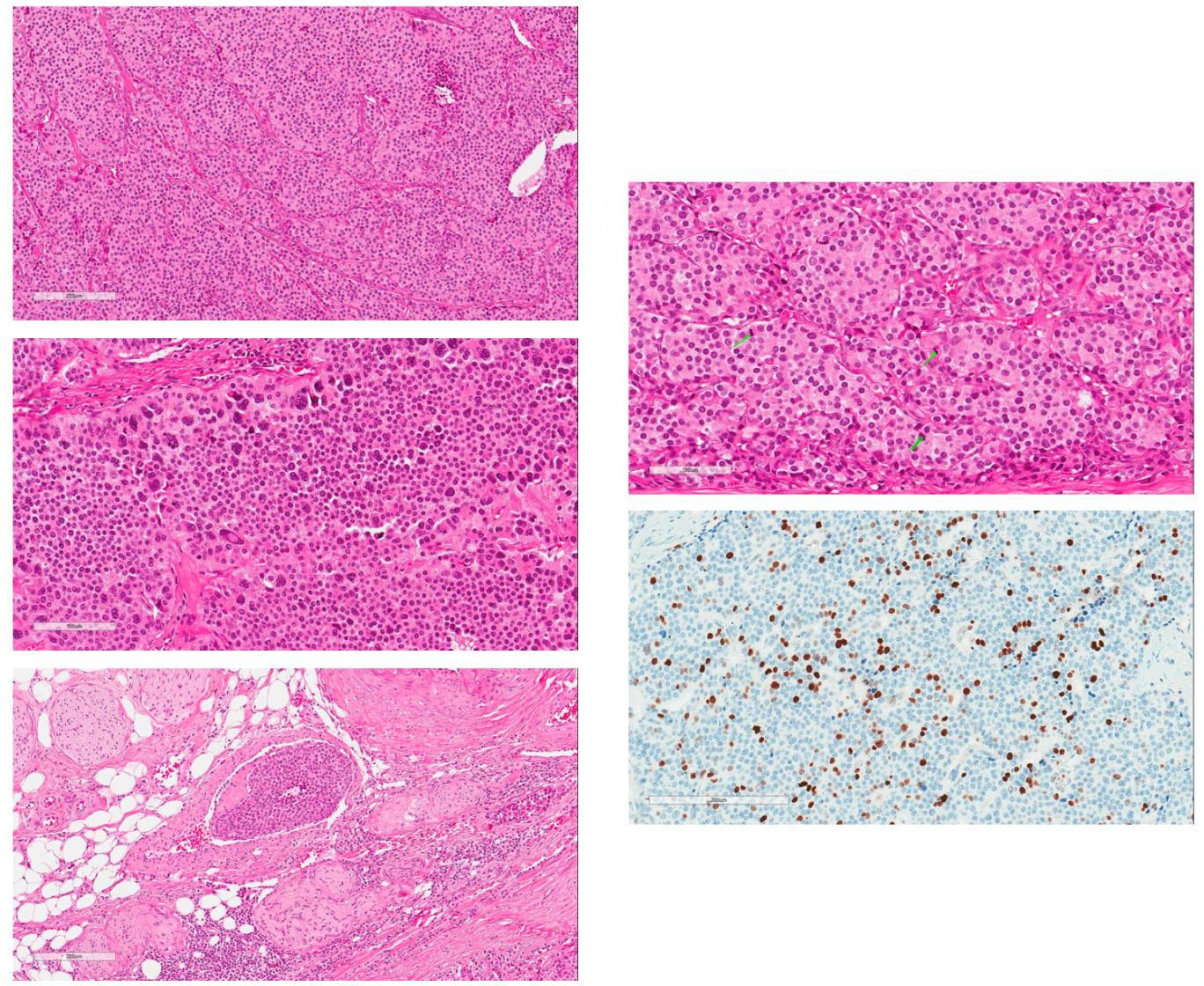
Fig. 5
Microscopic examination of the tumour. (a) H & E section at low magnification. Tumour is composed of confluent islands of relatively monotonous cells with amphophilic granular cytoplasm and centrally located round nuclei. (b) Areas of marked cellular pleomorphism at higher magnification. (c) Evidence of intravascular spreading with tumour embolus in the vein lumen. (d) High mitotic activity with mitotic figures marked by green arrows. (e) Immunohistochemical staining by Ki-67. The proliferative index was up to 22% (quantified by digital pathological assessment with Aperio Nuclear V9)
Survival of such patients is mainly influenced by degree of differentiation, histological grade, stage, and anatomical site of the tumour. Poorly differentiated GI tract NETs are always considered as high grade and carry a particularly poor prognosis. Anyway, well-differentiated NETs are further subdivided based on histological grading by assessing proliferative activity of the tumour, including both mitotic count and Ki67 labeling index. Low grade lesions (G1) exhibit <2 mitoses/10 HPF and <3% Ki67 indexing, intermediate grade (G2) demonstrate from 2 to 20 mitoses/10 HPF and from 3 to 20% Ki67 immu- nopositivity whereas high-grade (G3) well-differentiated NETs display >20 mitoses and >20% proliferative Ki67 activity (6). Therefore, the highgrade NETs consist of two distinct groups of neoplasms: well-differentiated carcinoids with high proliferative activity and poorly differentiated counterparts. Even though majority of high-grade NETs are poorly differentiated (up to 82% of cases), a small subset of well-differentiated G3 NETs poses a therapeutic challenge for the oncologists. Management of such lesions is not standardized and is balanced between low-to-intermediate grade well-differentiated and poorly differentiated NETs treatment modalities, primarily due to rarity of the histological phenotype and lack of well-designed randomized clinical trials aimed at identification of the optimal regimen (7). Currently, platinum-based chemotherapeutic agents are considered as a first-line choice for high-grade NETs, but 2 recent retrospective studies showed that a subgroup of well-differentiated G3 neuroendocrine neoplasms respond much worse to such treatment modalities while comparing with poorly differentiated carcinoids (8, 9). This ques- tion the utility of chemotherapy for such patients and the choice of systemic therapy is based on an individual case-to-case basis, weighting potential risk and benefit ratio. Remarkably, anatomical location of the primary tumour is of great prognostic importance. In advanced metastatic disease with high-grade phenotype, small intestine or appendiceal localization was associated with the best survival (median OS of 30–33 months) in a contrast with colonic and cecal origin lesions, which carries the worst prognosis (median OS of 8 months) (1).
The role of the primary tumour resection in the setting of unresectable metastatic disease remains controversial. The systematic review published in 2012 assessed the survival benefit of primary midgut NETs resection in patients with non-operable liver metastatic disease (10). Six studies were included and showed definitive trend towards better overall survival of surgical group in comparison with no resection (5-year survival 74% versus 36%). Anyway, all studies included were retrospectively designed and carried a high risk of selection bias. Another systematic review conducted in 2016 also evaluated the role of surgery in patients with unresectable hepatic metastases (11). Eight studies were included and all of them demonstrated the better outcome among surgically treated individuals, but retrospec- tive nature of those studies and methodological heterogeneity makes data difficult to interpret. The most recent cohort addressing this issue was published in 2018 (12). Researchers compared the outcome differences between 161 asymptomatic patients with metastatic small bowel NETs who underwent surgical resection of the primary lesion plus standard oncological treatment versus 202 similar patients who received only oncological treatment alone and underwent delayed surgery only in the case of necessity. Primary tumour resection group showed no survival benefit and had a higher rate of complications while comparing to conservatively managed patients. Currently available literature yields conflicting results and clinical decision making in such cases is based solely on clinician opinion and previous experience. There is a great need of randomized clinical trials (RCTs) to assess the utility of primary GI tract NET resection in an advanced disease. An international EUROCRINE registry, which was started in 2013, specifically focuses on data collection about rare endocrine tumours, including gastroenteropancreatic NETs. Acquired information will be used to raise the standards of treatment and reduce differences in practice between participating clinics and countries, hopefully optimizing the care provided.
According to North American Neuroendocrine Tumor Society (NANETS) guidelines published in 2017, surgical treatment in a context of unresectable liver metastases should be advised for patients who have symptoms attributable to the primary lesion and are expected to improve subsequent to the operation (13). Similar recommendations were issued by European Neuroendocrine Tumor Society (ENETS) in 2017, stressing that the resection of primary small intestine NETs in an advanced disease might improve survival and prevent complications related to bowel obstruction, intestinal ischemia, and potentially improve associated paraneoplastic carcinoid syn- drome symptoms (14). On the contrary, the same ENETS consensus guidelines published in 2016 state that surgical intervention is not recommended in case of high grade, well-differentiated NETs and their poorly differentiated counterparts. It is important to emphasize that the evidence to support such recommendations is very sparse and is mainly based on retrospective studies and few minor non-randomized clinical trials (15). Therefore, whether the same principles could be applied to disseminated cecal NETs remains unknown as there are no clear recommendations available regarding the management of such particular cases. Nevertheless, following our MDT discussion we decided to proceed with a right open hemicolectomy and adjuvant chemotherapy in order to im- prove current symptoms, prevent future complications, and, hopefully, prolong survival.
CONCLUSIONS
There is no clear consensus regarding surgical and oncological management of high-grade well-differentiated NETs, especially in a context of met- astatic disease. Currently available guidelines recommend managing such patients on case-tocase basis, preferably following MDT discussion. Lack of well-designed RCTs and novel treatment methods limits clinician’s potential to provide the prime care for such patients. Further research in this field is warranted, aiming at delineation of best surgical candidates and optimal medical therapy regimens.
References
1. Dasari A, Shen C, Halperin D, et al. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017; 3(10): 1335–42. https://doi:10.1001/jamaoncol.2017.0589
2. Öberg KE. Gastrointestinal neuroendocrine tumors. Ann Oncol. 2010; 21(suppl_7): vii72–80. https://doi:10.1093/annonc/mdq290
3. Modlin IM, Oberg K, Chung DC, et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008; 9(1): 61–72. https://doi:10.1016/S1470- 2045(07)70410-2
4. Muysoms FE, Miserez M, Berrevoet F, et al. Classification of primary and incisional abdominal wall hernias. Hernia. 2009; 13(4): 407–14. https://doi:10.1007/ s10029-009-0518-x
5. Schnirer II, Yao JC, Ajani JA. Carcinoid – a comprehensive review. Acta Oncol Stockh Swed. 2003; 42(7): 672–92. https://doi:10.1080/02841860310010547
6. Rindi G, Klimstra DS, Abedi-Ardekani B, et al. A common classification framework for neuroen- docrine neoplasms: an International Agency for Research on Cancer (IARC) and World Health Organization (WHO) expert consensus proposal. Mod Pathol. 2018; 31(12): 1770–86. https://doi:10.1038/ s41379-018-0110-y
7. Coriat R, Walter T, Terris B, Couvelard A, Ruszniewski P. Gastroenteropancreatic well-differentiated grade 3 neuroendocrine tumors: review and position statement. The Oncologist. 2016; 21(10): 1191–9. https://doi:10.1634/theoncologist.2015-0476
8. Sorbye H, Welin S, Langer SW, et al. Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (WHO G3): The NORDIC NEC study. Ann Oncol. 2013; 24(1): 152–60. https://doi:10.1093/ annonc/mds276
9. Heetfeld M, Chougnet CN, Olsen IH, et al. Characteristics and treatment of patients with G3 gastroenteropancreatic neuroendocrine neoplasms. Endocr Relat Cancer. 2015; 22(4): 657–64. https://doi:10.1530/ ERC-15-0119
10. Capurso G, Rinzivillo M, Bettini R, Boninsegna L, Delle Fave G, Falconi M. Systematic review of resection of primary midgut carcinoid tumour in patients with unresectable liver metastases. Br J Surg. 2012; 99(11): 1480–6. https://doi:10.1002/bjs.8842
11. Guo J, Zhang Q, Bi X, et al. Systematic review of resecting primary tumor in MNETs patients with unresectable liver metastases. Oncotarget. 2016; 8(10): 17396–405. https://doi:10.18632/oncotarget.14156
12. Daskalakis K, Karakatsanis A, Hessman O, et al. association of a prophylactic surgical approach to stage IV small intestinal neuroendocrine tumors with survival. JAMA Oncol. 2018; 4(2): 183–9. https://doi:10.1001/jamaoncol.2017.3326
13. Howe JR, Cardona K, Fraker DL, et al. The surgical management of small bowel neuroendocrine tumors: consensus guidelines of the North American Neuroendocrine Tumor Society (NANETS). Pancreas. 2017; 46(6): 715–31. https://doi:10.1097/ MPA.0000000000000846
14. Partelli S, Bartsch DK, Capdevila J, et al. ENETS Consensus guidelines for the standards of care in neuroendocrine tumours: surgery for small intestinal and pancreatic neuroendocrine tumours. Neuroendocrinology. 2017; 105(3): 255–65. https://doi:10.1159/000464292
15. Garcia-Carbonero R, Sorbye H, Baudin E, et al. ENETS consensus guidelines for high-grade gastroenteropancreatic neuroendocrine tumors and neuroendocrine carcinomas. Neuroendocrinology. 2016; 103(2): 186–94. https://doi:10.1159/000443172
Notes
Información adicional
CONFLICT OF INTEREST: The authors declare no conflict of interest.