Artículo Original
Evaluation of the Implantation of the Good Practices Guideline for Vascular Access in the Euskadi Regional Host of Osakidetza-Basque Health Service: A Descriptive Studio of the Process
Evaluación de la implantación de la guía de buenas prácticas de acceso vascular en el Host Regional Euskadi de Osakidetza-Servicio Vasco de Salud: estudio descriptivo de proceso
Avaliação da implementação do guia de boas práticas para acesso vascular no Host Regional Euskadi de Osakidetza-Serviço Vasco de Saúde: estudo descritivo do proceso
Evaluation of the Implantation of the Good Practices Guideline for Vascular Access in the Euskadi Regional Host of Osakidetza-Basque Health Service: A Descriptive Studio of the Process
MedUNAB, vol. 26, núm. 2, pp. 187-199, 2023
Universidad Autónoma de Bucaramanga

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial 4.0 Internacional.
Recepción: 09 Febrero 2023
Aprobación: 19 Septiembre 2023
Abstract: Introduction. Intravenous treatments are necessary in multiple situations; however, they do have complications. The vascular access guideline provides assessed and synthesized evidence to be transferred to care. The aim of this study is to describe the process of implementation of the vascular access guideline in the organizations of Euskadi Regional Host by monitoring the use of alternative catheters, early removal and the presence of complications. Methodology. Descriptive study of process. The cut-off times are 3 months pre- implementation and 6, 12, 48 months post- implantation. Descriptive and comparative analysis with Chi- square. Results. A total of 7,074 patients and 12,363 devices were evaluated; The short peripheral catheter is the most frequently used. The use of alternative catheters increased over time, showing a significant difference in the 3 months pre- implementation period with the 48-month post-implantation period (from 6.81% pre- implementation to 10.58% post 48 months). The percentage of short intravenous catheters removal in less than 48 hours increased from 53.25% to 57.54% of the pre-implantation of the 48 months post-implantation. Discussion. The results obtained after the implementation of the guideline highlight the impact of the use of alternative catheters and their early removal. Further work needs to be done to reduce complications and rates of peripheral venous catheter phlebitis. Conclusions. The implementation of the good practices guidelines for vascular access care improves the results in the use of alternative catheters or the reduction in peripheral venous catheters in days.
Keywords: Implementation Science, Vascular Access Devices, Practice Guideline, Evidence-Based Practice, Nursing.
Resumen: Introducción. Los tratamientos endovenosos son necesarios en multitud de situaciones; sin embargo, presentan complicaciones. La guía de acceso vascular ofrece evidencia valorada y sintetizada para ser trasladada al cuidado. El objetivo del estudio es describir el proceso de implantación de la guía de acceso vascular en las organizaciones del Host Regional Euskadi monitorizando uso de catéteres alternativos, retirada precoz y presencia de complicaciones. Metodología. Estudio descriptivo de proceso. Se establecen como momentos de corte 3 meses preimplantación y 6, 12 y 48 meses postimplantación. Análisis descriptivo y de comparación con Chi cuadrado. Resultados. Se evaluaron 7,074 pacientes y 12,363 dispositivos; siendo el catéter periférico corto el de uso más frecuente. La utilización de catéteres alternativos aumentó a lo largo del tiempo, mostrando diferencias significativas en el periodo 3 meses preimplantación con el de 48 meses postimplantación (del 6.81% preimplantación al 10.58% post 48 meses). El porcentaje de catéteres venosos periféricos cortos retirados en menos de 48h aumentó del 53.25% al 57.54% de la preimplantación a los 48 meses postimplantación. Discusión. Los resultados obtenidos tras la implantación de la guía ponen de relieve el impacto de la utilización de catéteres alternativos y la retirada precoz de estos. Se ha de seguir trabajando en disminuir las complicaciones y las tasas de flebitis de catéter venosos periféricos. Conclusiones. La implantación de la guía de buenas prácticas para el cuidado del acceso vascular mejora los resultados en el uso de catéteres alternativos o la disminución en días de los catéteres venosos periféricos.
Palabras clave: Ciencia de la Implementación, Dispositivos de Acceso Vascular, Guía de Práctica Clínica, Práctica Clínica Basada en la Evidencia, Enfermería.
Resumo: Introdução. Os tratamentos intravenosos são necessários em muitas situações; porém, apresentam complicações. O guia de acesso vascular oferece evidências avaliadas e sintetizadas para serem transferidas para o cuidado. O objetivo do estudo é descrever o processo de implementação do guia de acesso vascular nas organizações do Host Regional Euskadi, monitorando o uso de cateteres alternativos, remoção precoce e presença de complicações. Metodologia. Estudo descritivo de processo. São estabelecidos como momentos de corte 3 meses pré-implantação, e 6, 12 e 48 meses pós-implantação. Análise descritiva e comparativa com Qui-quadrado. Resultados. Foram avaliados 7,074 pacientes e 12,363 dispositivos, sendo o cateter periférico curto o mais utilizado. O uso de cateteres alternativos aumentou ao longo do tempo, apresentando diferenças significativas no período de 3 meses pré-implantação com o de 48 meses pós-implantação (de 6.81% pré-implantação para 10.58% pós 48 meses). A porcentagem de cateteres venosos periféricos curtos removidos em menos de 48 horas aumentou de 53.25% para 57.54% desde a pré-implantação até 48 meses pós-implantação. Discussão. Os resultados obtidos após a implementação do guia destacam o impacto do uso de cateteres alternativos e sua remoção precoce. O trabalho deve continuar para reduzir complicações e taxas de flebite por cateter venoso periférico. Conclusões. A implementação do guia de boas práticas para cuidados com acesso vascular melhora os resultados no uso de cateteres alternativos ou na redução de dias de cateteres venosos periféricos.
Palavras-chave: Ciência da Implementação, Dispositivos de Acesso Vascular, Guia de Prática Clínica, Prática Clínica Baseada em Evidências, Enfermagem.
Introduction
Intravenous treatments are necessary in a multitude of patients and clinical situations. There are venous devices of different materials for different routes or anatomical locations, for varied durations, and for physicochemical characteristics of varied pharmaceuticals. Healthcare focused on quality and safety implies the use of scientific knowledge in daily practice, and the selection and adaptation of materials implemented; the application of care that avoids complications is fundamental.
The vascular accesses allow for a high bioavailability of the pharmaceuticals at the systemic level, avoiding problems of inadequate absorption, but it can produce complications (1,2).
Around 60% of hospitalized patients have venous access, as well as 90% of the patients who visit the emergency services, with an estimated 70% of these patients experiencing catheter-related complications (1).
Besides phlebitis, complications include thrombophlebitis, infiltration and extravasation, pneumothorax, hemothorax, infections, thrombosis, and catheter-related bacteremia (3).
Before 2018, finding variability in vascular care, different service organizations began working on implementing recommendations for selecting the appropriate vascular device, introducing intravenous therapy devices, and monitoring maintenance care and complications, but in a not very structured manner, without a rigorous implementation methodology and systematic monitoring from the health service.
The national study EPINE (4) shows that 76.57% of the hospitalized people used a peripheral vascular catheter (CVP), and 12.34% a central venous catheter (CVC) finding a bacterial prevalence and catheter-associated infection 1.41%, which was acquired at the health center.
Besides the impact on the patient, complications from vascular access devices increase costs, hospital stays and morbidity and mortality rates (5-7). Therefore, promoting knowledge and scientific evidence is vital to reduce variability and the risk of complications.
To reduce this problem, various initiatives have emerged, such as the introduction of intravenous therapy teams in expert vascular access centers, new types of devices like the PICC (Peripherally Inserted Central Catheter), new insertion techniques like ultrasound-guided microseldinger, and materials like third-generation polyurethane and silicone. Finally, it is worth noting that there is a growing body of evidence on venous access care, as outlined in various clinical practice guidelines on vascular care (6,8-11).
Health professionals are called upon to use the results of scientific research in the care they provide. However, there is a gap between evidence and practice (5), variability, and non-use of updated knowledge daily. One of the transfer strategies is the Clinical Practice Guidelines, which, based on the level of evidence found, propose action guidelines to facilitate the transmission of evidence to practice since they offer synthesized and critically evaluated evidence for the management of different aspects of a health problem and, in addition, incorporate practice recommendations, which helps to create a bridge between practice and research results (12).
Applying the Good Practice Guidelines supports the improvement of clinical practice by using consistent results and promoting the reduction of variability. However, evidence alone may not be sufficient for its translation, and many professionals are unfamiliar with how to effectively, efficiently, and sustainably implement interventions in clinical practice (13). Therefore, it is necessary to facilitate access to Good Practice Guidelines and set up an implementation methodology that promotes translating of knowledge into daily practice (5).
The Registered Nurses’ Association of Ontario (RNAO) develops, supports the implementation, and evaluates Good Practice Guidelines and healthy work environments. Currently, RNAO has 54 guidelines for good clinical practices and healthy work environments, including the one for vascular access.
RNAO designated the Research Unit in Health Care, Investén-isciii, as the BPSO Host in Spain to implement, evaluate, disseminate, and sustain/expand RNAO’s Good Practice Guidelines through the Centers Committed to Excellence in Care / Best Practice Spotlight Organization (CCEC/BPSO.), developing the functions of BPSO Host- Spain since 2010. This initiative promotes activities for creating and supporting a network of CCEC/BPSO in Spain, as well as coordinating the creation and development of Regional BPSO Hosts in Spain (5,14)(Figure 1).

Figure 1. Guidelines implementation network
Figure 1. Guidelines implementation network
*The figure shows the relationships of the global BPSO community: RNAO-CCEC®/BPSO® Spain - Euskadi regional host.
taken and adapted from https://rnao.ca/bpg/bpso and https://www.bpso.es/In Spain, the Regional BPSO Host-regional model emerged from the expressed need by some Autonomous Communities to expand the model and adopt the Centers Committed to Excellence in Care. program (CCEC./ BPSO. Spain), as the program has shown improvement in clinical processes, patient health results, and healthcare structures of participating institutions (5,14). The Regional Hosts, typically based in the health service directorates of each autonomous community, coordinate the convocations, leader training, and program monitoring in collaboration with other Hosts, ensuring its replicability across their healthcare services. Specifically, the Euskadi Regional Host started in 2018, was designated in 2021, and coordinates the BPSO program in 8 healthcare organizations of Osakidetza-Basque Health Service.
The implementation methodology is based on the tool developed by RNAO (15), which combines the implementation methodology framework of Knowledge to Action with the phases of problem identification, analysis of the local context, evaluation of recommendations, implementation of strategies, knowledge monitoring, evaluation of results, and dissemination within the framework of the Social Action Movement (16)(Figure 2).
Figure 2. Conceptual frameworks for evidence implementation
Figure 2. Conceptual frameworks for evidence implementation
*The figure shows the two frameworks to guide the application of evidence and its sustainability
Leading Change Toolkit https://rnao.ca/leading-change-toolkitIn implantation and sustainability, evaluation and feedback constitute one of the fundamental phases. The evaluation system allows for understanding the degree of development of the implantation and the magnitude of the results obtained, both in terms of health outcomes and changes in the care process within organizations (14).The NHS sustainability model (17) also highlights the importance of having systematic tools to demonstrate impact, monitor progress, and communicate results that can continue beyond implantation.
One of the fundamental challenges assumed by Host Euskadi has been to design and build an indicator system to evaluate the evolution of the implantation of the RNAO guidelines in Osakidetza-Basque Health Service. The objective was to have a dashboard that met the following criteria:
● To exclusively use data from the clinical history collected in daily clinical practice, without depending on other parallel or additional methodologies.
● Provide measurable and homogeneous results for the whole Basque Health Service for any period and different care areas or units.
● Ability to explore each anonymized case individually.
● Accessible, exploitable, and easily usable for the organization’s users.
Host Euskadi has made progress in this development by adapting and modifying elements of the unified electronic health record for the whole Basque Health Service, operationalizing specific indicators from the guidelines into dashboards in the Oracle Analytics tool, and finally making these dashboards available for result evaluation to the whole health system, for any organization and period of analysis.
The RNAO “Vascular Access” guideline (6) is being implemented in five healthcare organizations in the Euskadi Regional Host. Specifically, in the year 2022, a total of 158,092 patients were carriers of a venous device in our health service.
The current Vascular Access guideline (6) updates previous practice guidelines for The Care and Maintenance of vascular access to reduce complications (9), and Assessment and Selection of vascular access devices (10).
Among the recommendations of the current guideline (6) for the management and use of venous catheters are training for professionals and daily review of the devices, monitoring device-associated complications or their adequacy.
Therefore, the objective of this study is to describe the implementation process of the clinical practice guideline for vascular access in the organizations of the Euskadi Regional Host, monitoring the use of alternative catheters, early removal, and the presence of complications.
Methodology
Design: Descriptive study of an implementation process.
Study context: Osakidetza-Basque Health Service hospitalization units that are BPSO institutions, designated or candidates, are implementing the Vascular Access Guide. These units are within five hospitals: three tertiary hospitals, one regional hospital (second-level hospital with medium complexity that meets the health needs of that geographic area), and one medium-long stay hospital, caring for patients with chronic processes (or with a degree of functional dependency for daily activities, that cannot be provided at home, and require a prolonged period of hospitalization) with nursing teams of between 50-70 people made up of nurses (who are the ones who directly care for the patient vascular access) and auxiliary nursing care technicians.
Participants: The studied nursing units were traditional hospitalization units (with people over 14 years of age), excluding short-stay units, critical care units, and day hospitals. It was also considered as an exclusion criterion that the nursing unit in the study period had been completely moved to another location, which also entails changes in the typology of patients cared for and team professionals, a situation that occurred occasionally in the period of the study, which includes the COVID-19 pandemic; In addition, those units that were only opened for reasons of workload were excluded. A total of 25 hospitalization units from 5 Health Organizations were studied: OSI Araba (7 units), OSI Bilbao Basurto (5 units), OSI Goierri Alto Urola (6 units), OSI Ezkerraldea-Enkarterri-Cruces (2 units), and the Górliz Hospital for medium-long stay (5 units).
Variables: To meet the objectives of the study, the outcome variables that were evaluated and extracted from the Medical History were the following:
• The number of vascular access devices used: total, peripheral venous, and alternative catheters. Alternative venous access devices to the short peripheral catheter are the PICC, the central venous catheter, the subcutaneous venous reservoir, and the midlines, all of which are more suitable for intravenous therapies lasting more than seven days or with pH or irritating substances.
• The percentage of complications associated with vascular access devices (DAV). The following events were complications: phlebitis, induration, hematoma, infiltration, extravasation, thrombosis, infection or suspected infection, bacteremia, pneumothorax, and dermal lesions.
• Regarding the most commonly used and short peripheral catheter, the number and percentage of devices and phlebitis were analyzed as the most common complication.
• Other descriptive variables were also measured, such as the number of patients seen per month, number of stays, and type of unit.
Procedure: The selected recommendations were chosen based on their approach to the problems identified in the centers, the following being chosen: 1.0 (choice of the peripheral insertion site); 2.0 (infection prevention); 3.0 (aseptic technique); 5.0 (dressings); 6.0 (valuation and fixation); 10.0 (minimization of central venous access); 12.0 (registration) of the guide revised in 2008 (9). After the guide was updated in 2012 (6), the recommendations chosen by the centers were 2.1 (education of insertion processes); 3.1 (formation of intravenous therapy teams); 5.1 (catheter check); 6.1 (use of ultrasound).
The guide implementation leaders were trained at the RNAO good practices institute by the Host, and subsequently, in each center and unit, after the recruitment of promoters, the leaders carried out the training of the GBP of DAV to the promoters of good practices. The monitoring of the implementation process was coordinated by the project leaders in each institution, receiving support and monitoring from the Host.
The study began in May 2018, when the first center began implementing the guide, and ended in December 2022. The evaluation takes into account the different phases of the implementation process: the pre-implementation period (pre-three months), the early implementation phase after recruitment and training (post-six months), the consolidation phase (post-12 months), and the sustainability phase (post-48 months). Outcomes were measured at those cohort points for each BPSO candidate or designated institution. As the institutions joined the BPSO program in different calls, the analysis periods were different and corresponded to the following schedule(Table 1).
The data was obtained and anonymized for the entire study period, exploiting the results through dashboards designed with Oracle Business Intelligence, specifically built to monitor the implementation of good practices and RNAO guidelines in Osakidetza. These dashboards report on monitoring indicators and export anonymized information directly from medical records. The clinical history and care registration systems are standardized for the entire Osakidetza-Basque Health Service, and the configuration criteria for automated queries for data extraction are homogeneous and have been configured uniformly for all implementation sites.
Table 1. Periods Evaluated.
| Table 1. Periods Evaluated | |||||
| Institution | Year BPSO Beginning | Pre-3 months | Post 6 months | Post 12 months | Post 48 months |
| OSI Araba (Araba) | 2018 | oct-17 | May-18 | nov-18 | nov-21 |
| OSI Goierri Alto Urola (GAU) | 2019 | Oct-18 | may-19 | nov-19 | nov-22 |
| OSI Ezkerraldea Enkarterri (EEC) | 2019 | Oct-18 | may-19 | nov-19 | nov-22 |
| OSI Bilbao Basurto (BB) | 2022 | oct-21 | may-22 | nov-22 | — |
| Hospital Górliz (Górliz) | 2022 | oct-21 | may-22 | nov-22 | — |
| *The table shows the periods evaluated according to institution and year of BPSO initiation. | |||||
| Source: elaborated by authors. | |||||
The data was subsequently exported to Excel to clean the databases. The statistical analysis was descriptive; the Chi- square test was used to evaluate the comparisons. Data was analyzed using IBM SPSS Statistics v23 computer software.
Results
Twenty-five hospitalization units were evaluated at four assessment points (pre-implementation (3 months prior) and post-implementation (6, 12, and 48 months) across the five participating organizations. The OSI Araba, OSI GAU, OSI EEC, and OSI BB centers are acute patient hospitalization units equipped with intravenous therapy teams that disseminate and monitor vascular access blood pressure parameters within the organizations. They also perform PICC and midline catheterizations using ultrasound guidance and act as expert consultants for complex cases. The Górliz hospital is a medium to long-term care center with a dedicated intravenous therapy team. During the study period, 7,074 patients who were carriers of a vascular device were monitored in the centers and units implementing the RNAO Vascular Access guideline (Table 2).OSI Araba contributed 41.46% of patients to the sample, followed by OSI GAU with 21.91%.
Table 2.Sample Distribution.
| Table 2. Sample Distribution. | ||||||
| Number of nursing units | Number of patients Pre 3 months | Number of patients Post 6 months | Number of patients Post 12 months | Number of patients Post 48 months | Total patients | |
| Górliz | 5 | 104 | 91 | 97 | 292 | |
| Araba | 7 | 745 | 802 | 727 | 659 | 2933 |
| BB | 5 | 396 | 386 | 388 | 1,17 | |
| EEC | 2 | 320 | 281 | 281 | 247 | 1,129 |
| GAU | 6 | 422 | 437 | 430 | 261 | 1,55 |
| Total | 25 | 1,987 | 1,997 | 1,923 | 1,167 | 7,074 |
| *The table indicates the number of nursing units and patients included in the evaluation analysis in each period and the organization of services. | ||||||
| Source: elaborated by the authors. Data extracted from the Oracle Business Intelligence tool. | ||||||
*The table indicates the number of nursing units and patients included in the evaluation analysis in each period and the organization of services.
* The table indicates the number of nursing units and patients included in the evaluation analysis in each period and the organization of servicesThe data presented below were obtained through the Oracle Business Intelligence tool for exporting results from the care assessment and planning system. This program allows registering, per patient, the type and number of vascular access devices (DAV) used, including short peripheral venous catheter, central venous catheter, peripheral venous access catheter, tunneled central catheter, midline catheter, and subcutaneous venous reservoir. It also records their characteristics (type, number of lumens, gauge, etc.), along with the placement and removal dates, reasons for removal (completion of treatment, change of gauge or device, discomfort, accidental removal, leakage at insertion site), and complications (phlebitis, infiltration, extravasation, hematoma, skin lesions, infection, bacteremia, suspected infection, and thrombosis).
The total number of devices placed during the entire analysis period was 12,363, with 3,390 corresponding to the pre-implementation period, 3,476 to the post-implementation period at six months, 3,370 to the post-implementation period at 12 months, and 2,127 to the post-implementation period at 48 months (Table 3). The short peripheral venous catheter was the most commonly used type of DAV in all analysis periods and institutions, with its utilization ranging from 95.69% to 80.71% of the total inserted catheters, according to implantation centers and evolution in implementation (Table 3). The use of alternative catheters to the short peripheral route, not indicated for irritating or long-term intravenous therapies, increased from 6.81% pre-implementation to 10.58% post-48 months (Figure 3). From the post- implementation period of six months to the sustainability period post-48 months, the proportion of alternative catheters to the peripheral route increased from 7.10% to 23.66%. The comparison of periods using the chi-square test showed significant differences in catheter usage when contrasting the usage in the 3-month pre-implementation period with the 48-month post-implementation period with p<0.0001, as well as when comparing the post-implementation periods at 6 and 48 months (p<0.0001).
Table 3. Use of catheters
| Table 3. Use of catheters | ||||||||||||||||
| 3 months Pre intervention | 6 months Post intervention | 12 months Post intervention | 48 months Post intervention | |||||||||||||
| Nº DAV | Nº DAV alt | Nº CVP | % CVP | Nº DAV | Nº DAV alt | Nº CVP | % CVP | Nº DAV | Nº DAV alt | Nº CVP | % CVP | Nº DAV | Nº DAV alt | Nº CVP | % CVP | |
| Górliz | 186 | 19 | 167 | 89.78% | 160 | 26 | 134 | 83.75% | 163 | 27 | 136 | 83.44% | ||||
| Araba | 1,326 | 92 | 1,234 | 93.06% | 1,365 | 75 | 1,29 | 94.51% | 1,263 | 69 | 1,194 | 94.54% | 1,279 | 63 | 1,216 | 95.07% |
| BB | 670 | 32 | 638 | 95.22% | 696 | 38 | 658 | 94.54% | 682 | 24 | 656 | 96.19% | ||||
| EEC | 581 | 61 | 520 | 89.50% | 566 | 39 | 527 | 93.11% | 521 | 47 | 474 | 90.98% | 477 | 91 | 385 | 80.71% |
| GAU | 627 | 27 | 600 | 95.69% | 689 | 53 | 646 | 93.76% | 741 | 39 | 702 | 94.74% | 371 | 71 | 300 | 80.86% |
| Total | 3,39 | 231 | 3,159 | 93.19% | 3,476 | 231 | 3,255 | 93.64% | 3,37 | 206 | 3,162 | 93.83% | 2,127 | 225 | 1,901 | 89,37% |
| *The table shows the total number of vascular access devices (No. DAVs), number of peripheral venous catheters (No. CVP), percentage of peripheral venous catheters (% CVP), number of alternative vascular access devices (No. alt DAVs) (PICC, midlines or other type of DAVs) in each period and organization of services. | ||||||||||||||||
| Source: elaborated by the authors. Data extracted from the Oracle Business Intelligence tool. | ||||||||||||||||
*The table shows the total number of vascular access devices (No. DAVs), number of peripheral venous catheters (No. CVP), percentage of peripheral venous catheters (% CVP), number of alternative vascular access devices (No. alt DAVs) (PICC, midlines or other type of DAVs) in each period and organization of services.
elaborated by the authors. Data extracted from the Oracle Business Intelligence tool.* The table shows the total number of vascular access devices No DAVs number of peripheral venous catheters No CVP percentage of peripheral venous catheters % CVP number of alternative vascular access devices No alt DAVs PICC midlines or other type of DAVs in each period and organization of services
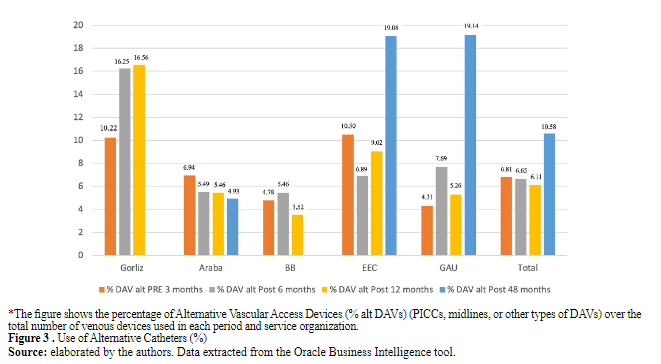
Figure 3 . Use of Alternative Catheters (%)
Figure 3. Use of Alternative Catheters (%)
*The figure shows the percentage of Alternative Vascular Access Devices (% alt DAVs) (PICCs, midlines, or other types of DAVs) over the total number of venous devices used in each period and service organization.
elaborated by the authors. Data extracted from the Oracle Business Intelligence tool.Additionally, organizations implementing good vascular access care practices aimed to remove unnecessary catheters as early as possible, thus avoiding complications and transitioning to oral therapy in cases where continued treatment was necessary. Therefore, it was observed that the percentage of short peripheral venous catheters removed in less than 48 hours increased from 53.25% to 57.54% from pre-implementation to 48 months post- implementation, with this difference being statistically significant (p< 0.001) (Table 4).
Table 4.Peripheral venous catheters withdrawn early (<48h)
| Table 4. Peripheral venous catheters withdrawn early (<48h) | ||||||||
| Pre 3 months | Post 6 months | Post 12 months | Post 48 months | |||||
| Average CVP length | %<=2 days | Average CVP length | %<=2 days | Average CVP length | %<=2 days | Average CVP length | %<=2 days | |
| Gorliz | 3.34 | 52.69 | 3.35 | 50.87 | 3.46 | 53.41 | ||
| Araba | 2.82 | 56.36 | 2.80 | 57.62 | 2.88 | 56.04 | 3.13 | 51.41 |
| BB | 2.95 | 43.60 | 3.17 | 49.82 | 2.87 | 56.28 | ||
| EEC | 3.47 | 48.55 | 3.25 | 54.56 | 3.46 | 49.22 | 2.43 | 65.76 |
| GAU | 2.51 | 65.06 | 2.74 | 60.72 | 2.70 | 60.56 | 2.82 | 55.47 |
| Total | 3.02 | 53.25 | 3.06 | 54.72 | 3.07 | 55.10 | 2.79 | 57.54 |
| *The table indicates the mean length of short peripheral catheters (SVC) and the percentage of SVC removed less than 2 days after placement in each period and service organization (%<=2 days). | ||||||||
| Source: elaborated by the authors. Data extracted from the Oracle Business Intelligence tool | ||||||||
*The table indicates the mean length of short peripheral catheters (SVC) and the percentage of SVC removed less than 2 days after placement in each period and service organization (%<=2 days).
elaborated by the authors. Data extracted from the Oracle Business Intelligence tool* The table indicates the mean length of short peripheral catheters SVC and the percentage of SVC removed less than 2 days after placement in each period and service organization %2 daysThe percentage of complications among the total vascular access devices can be observed in Figure 4. This result varies between 12% and 26% among organizations, depending on the complexity of patients and treatments in different hospitalization units. As observed, it also varies across different implementation periods. The statistical tests conducted to compare the results across different implementation phases Pre-Post 6 (p=0.014), Pre-Post 12 (p<0.001), and Pre-Post 48 (p<0.001) yielded significant results. However, the data obtained reflect some instability and indicate the need to continue strengthening this objective.
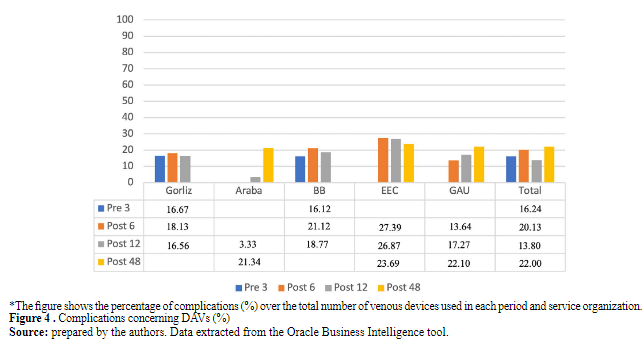
Figure 4 . Complications concerning DAVs (%)
Figure 4 . Complications concerning DAVs (%)
*The figure shows the percentage of complications (%) over the total number of venous devices used in each period and service organization
prepared by the authors. Data extracted from the Oracle Business Intelligence tool.The percentage of phlebitis concerning short peripheral catheters can be observed in Figure 5. This complication appeared in 9.88% of these devices and varied from 9.31% to 10.28% during the study period. The comparisons for this result in all compared periods showed a p>0.05, indicating no statistical significance.
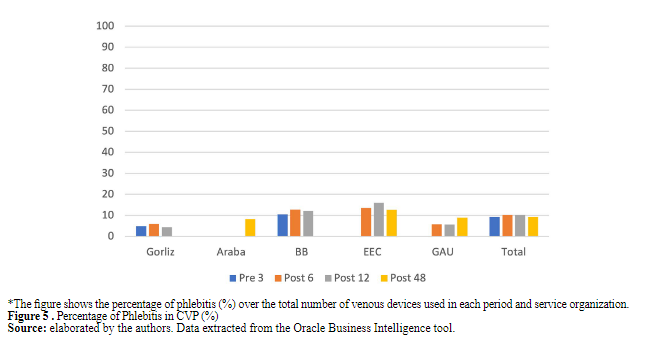
Figure 5 . Percentage of Phlebitis in CVP (%)
Figure 5. Percentage of Phlebitis in CVP (%)
*The figure shows the percentage of phlebitis (%) over the total number of venous devices used in each period and service organization.
elaborated by the authors. Data extracted from the Oracle Business Intelligence toolDiscussion
This study evaluates the results reached in hospitalization units that implant the BBPP guide of vascular access in Osakidetza in the BPSO program frame. The implantation methodology has mobilized and accelerated the incorporation of multiple changes despite carrying out the implantation in a big part of the pandemic term 2020- 2021. The effect of the implantation stays and consolidates in time, and the results suggest that 48 months after the implantation, the organizations still favorably evolve their results.
The results obtained after implanting recommendation 1.0 of the guide reviewed in 2008 (9)and 3.1, 6.1 of the 2021 guide (6) have given place to the extension and consolidation of intravenous therapy equipment in 5 institutions of Osakidetza, with an impact over the usage of alternative catheters and eco-guided technique, which nowadays is used as usual practice in the insertion of central catheters. The usage of alternative catheters for CVP increases by taking into account the performance according to the device algorithm of selection recommended by GPC; however, it has not been found in the literature a standard of good practice that works as a comparison element, except the numerous sources that estimate the usage of central catheters is 3.3% approximately of hospitalized patients (18), to which it would be necessary to add the recent usage of the midline. Regarding the early movement of the CVP backed up by the recommendation of the GPC with an evidence level of IA and the Flebitis Zero project (19,20), in which the Osakidetza organizations participate, is another indicator studied with low frequency. Therefore, it should take importance, in front of the traditional average duration of the catheter, orienting the indicators to the reduction of total days of catheterizing.
After the implantation of recommendations 1.0, 2.0, 3.0, 5.0, 6.0, and 10.0 of the 2008 guide (9) and 2.1 and 5.1 of the 2021 guide (6) observed the impact on the frequency of complications. Regarding the complication rates, the results are favorable about the ones other studies contribute. Organizations estimate complication percentages superior of 30% in the function of the catheter type (21) and assisted population, also showing a favorable result in Osakidetza organizations concerning the 12.9% of phlebitis detected in EPINE (4,20) that has only been surpassed by one organization in a concrete post- implantation term. However, it should be kept working to decrease the complications and phlebitis rates of CVP to reach the standard of 5% defined by the Infusion Nursing Society (8,20). Nevertheless, in interpreting the results, it is convenient to consider that achieving a great result in short catheter proportion is more difficult without phlebitis because many are being substituted with alternative catheters.
The results also suggest that guide implantation of the vascular access guide precipitates early time changes of good practice indicators, like the adequation in the selection of the device, vascular access, and early withdrawal, than those of health outcomes such as complications from these devices. This could be caused, in part, because complications are due to multiple causal effects besides a secure clinical practice, like the typology and condition of assisted patients and complex therapeutic necessities. Long- term following through the monitoring system built, which includes types of DAV, days of each catheter, withdrawing motives, and complications, will allow the evaluation of the impact outside the pandemic for the COVID-19 term. During the whole process, monitoring results and periodic feedback to clinicians helps to consolidate the work and establish strategies and new proposals that improve attention, being conscious that, to date, the results have not been too vigorous, but with the conviction that it will have an impact on daily life.
The guide’s implementation in institutions has supposed a dynamization of the evaluation systems and feedback, reflection, and the start-up of multiple strategies that improve vascular access care. During the implantation, groups of leaders and enhancers constituted in each institution to implant the guide has standardized and diffuse protocols and procedures, had formed professionals and actualized their knowledge, and had achieved the introduction of new materials like extensions with clamps, anti-reflux caps, or use of prefilled syringes to improve care. In some institutions, kits of vascular access are used with the material to properly channel and set the DAV, standardizing practices and avoiding variability. Also, the guides helped to consolidate intravenous therapy equipment, contributing this equipment to promote good practice recommendations in a determinative way.
Likewise, the effect of the study term over the reached results is worth mentioning. The years of implantation have coincided with the pandemic. During that time, there has been a tremendous professional replacement in the units and a high existential demand that doesn’t allow us to infer these results to other periods or contexts.
Parallely, the clinic history and register systems of the vascular access devices have adapted and improved valorization, following, and care continuity in patients carriers of vascular access devices, optimizing the coherence between Osakidetza clinic history records and the recommendations. This adequation made in the clinic history during the implantation period has been an essential strategy to transmit and maintain the good practices of vascular access care. However, this has provoked some occasions where it has not been possible to count all unit information in the results of this evaluation study. The evaluation methodology in implantation has to contribute not only to the results information but also to the same implantation process and its effectiveness, and this constitutes a limitation in this study and, at the same time, is a challenge for future studies, which they must contemplate in the methodology. The stability and maintenance of the information systems allow a better control of possible biases. Still, the question is how they should be combined with the constant actualization of knowledge and implantation processes.
In the limitations of this study, it is found that the implantation period has coincided with the pandemic; this could have influenced the results and the changes realized in the records for adequations recognized in the clinic history and its impact on the indicators. Also, the study’s retrospective design can entail information and selection biases, which were tried to control by analyzing the totality of patients in the study units during those periods. This type of study cannot demonstrate causality, but it can give a glimpse of the implantation effects of the unit guide. Future studies could be done to verify these results.
The event rate per day and patient evaluation weren’t monitorable during the significant part of the guide implantation process; that’s why that variable has yet to be evaluated. Starting in 2022, it has been possible to develop the evaluation of this data that will be reported in future studies.
Conclusions
In this study, the results evaluated were applying the clinical practice guide about vascular access in the Host Regional Euskadi organizations. This study analyzed the impact in 5 Osakidetza institutions with 20 units for acute patient hospitalization and 5 units for medium to long-term hospitalization. The implementation results show an increase in the use of alternative catheters to CVP and a decrease in the days of CVP use in relation to the reinforcement of recommendations for selecting DAV according to access difficulty, duration, and physicochemical characteristics of therapy, as well as the early removal of unnecessary catheters. The progress and strengthening of these good practices have been observed across the participating BPSO centers; However, the results have yet to show conclusive outcomes during the study period regarding the reduction of complication rates associated with vascular access devices. As mentioned, the implementation period coincided with the pandemic, which could have influenced these results. However, the construction of the results exploitation dashboard and the adjustments made following the implementation strategies will allow the progress of these indicators to be tracked sustainably over time. The path initiated, and the work being carried out by professionals and implementation teams may affect the reduction of complications in the near future. We believe that evaluation and research will allow us to verify the longer-term impact on health outcomes such as decreased phlebitis or complications.
Evaluating the implementation results makes it possible to visualize the changes and establish new objectives to improve the quality of care offered.
It is necessary to promote excellent quality care based on evidence; implementing the RNAO vascular access guidelines is another step towards achieving these results. The evaluation of the impact of the processes of incorporating evidence and the creation and follow-up of indicators that assess the implementation strategies and their effect on patient health is a necessary area that should continue to be promoted and researched to improve the care offered.
Acknowledgments
The authors thank the program’s leaders, promoters, and collaborators in Osakidetza and the Nursing and Management Departments for supporting and facilitating the implementation. They would also like to acknowledge the efforts of the leaders of the implementation program at the national level, other regional hosts, and the RNAO for sharing their knowledge and experience and for the support and encouragement received.
Conflicts of interest
The authors declare that there is no conflict of interest. The implementation of the Good Practice Guidelines through the Centers Committed to Excellence in Care® Program is partially funded by the Spanish Center for Evidence-Based Care: Joanna Briggs Institute Center of Excellence.
Funding
We thank Osakidetza and the Subdirectorate of Nursing for the resources provided for the development of this project. No funding was received for the writing of this manuscript.
Ethical responsibilities
Protection of people: This study is considered risk-free due to its nature.
Confidentiality of data: The authors declare they have followed their work center’s protocols on the publication of patient data. They worked with anonymized data.
References
1. Rodríguez-Calero MA, Blanco-Mavillard I, Morales-Asencio JM, Fernández-Fernández I, Castro-Sánchez E, De Pedro-Gómez JE. Defining risk factors associated with difficult peripheral venous Cannulation: A systematic review and meta-analysis. Heart & Lung [Internet]. 2020;49(3):273-86. doi: https://doi.org/10.1016/j.hrtlng.2020.01.009
2. Takahashi T, Murayama R, Abe-Doi M, Miyahara-Kaneko M, Kanno C, Nakamura M, et al. Preventing peripheral intravenous catheter failure by reducing mechanical irritation. Sci Rep [Internet]. 2020;10:1550. doi: https://doi.org/10.1038/s41598-019-56873-2
3. Royal College of Nursing. Standards for infusion therapy [Internet]. Reino Unido;2022. Avaliable from: https://www.rcn.org.uk/clinical-topics/Infection-prevention-and-control-advice/Standards-for-infusion-therapy
4. Sociedad Española de Medicina Preventiva, Salud Pública e Higiene. Estudio EPINE-EPPS nº 31: 2021. Informe España. Prevalencia de infecciones (relacionadas con la asistencia sanitaria y comunitarias) y uso de antimicrobianos en hospitales de agudos. [Internet] EPINE;2021. Avaliable from: https://epine.es/api/documento-publico/2021%20EPINE%20Informe%20Espa%C3%B1a%2027122021.pdf/reports-esp
5. González-María E, Moreno-Casbas MT, Albornos-Muñoz L, Grinspun D. Implantación de Guías de buenas prácticas en España a través del Programa de Centros Comprometidos con la Excelencia en Cuidados.. Enferm Clin [Internet]. 2020;30(3):136-144. doi: https://doi.org/10.1016/j.enfcli.2019.09.018
6. Registered Nurses’ Association of Ontario (RNAO). Guía de buenas prácticas: Acceso vascular. Segunda Edición [Internet]. Toronto:RNAO;2021. Avaliable from: https://rnao.ca/bpg/language/acceso-vascular-segunda-edici%C3%B3n
7. Bujalance-Hoyos J, Arenas-González F, González-Escobosa AC, Torres-Pérez L, Viñas-Vera C, Agudo-Guerrero E. Implantación de Buenas Prácticas en el Cuidado y Mantenimiento de los Dispositivos de Accesos Vasculares: estrategia y primeros resultados. Enfermería Docente [Internet]. 2019;111:10-22. Avaliable from: https://ciberindex.com/index.php/ed/article/view/11110ed
8. Gorski LA, Hadaway L, Hagle ME, Broadhurst D, Clare S, Kleidon T, et al. Infusion therapy standards of practice. J Infus Nurs [Internet]. 2021;44(S1):S1-S224. doi: https://doi.org/10.1097/nan.0000000000000396
9. Registered Nurses’ Association of Ontario (RNAO). Care and maintenance to reduce vascular access complications. [Internet]. Toronto:RNAO;2005. Avaliable from: https://rnao.ca/sites/rnao-ca/files/2014_CuidadoAccesoVascular_spp_022014.pdf
10. Registered Nurses’ Association of Ontario (RNAO). Assessment and Device Selection for Vascular Access Care and maintenance to reduce vascular access complications. [Internet]. Toronto:RNAO;2004. Avaliable from: https://rnao.ca/sites/rnao-ca/files/Vascular_Access_UPDATED.pdf
11. Chaves F, Garnacho-Montero J, Del Pozo JL, Bouza E, Capdevila JA, De Cueto M, et al. Diagnosis and treatment of catheter-related bloodstream infection: Clinical guidelines of the Spanish Society of Infectious Diseases and Clinical Microbiology and (SEIMC) and the Spanish Society of Spanish Society of Intensive and Critical Care Medicine and Coronary Units (SEMICYUC). Med Intensiva [Internet]. 2018;42(1):5-36. doi: https://doi.org/10.1016/j.medin.2017.09.012
12. Dartnell J, Hemming M, Collier J, Ollenschlaeger G. Putting evidence into context: some advice for guideline writers. Evid Based Nurs [Internet]. 2008;11(1):6–8. doi: https://doi.org/10.1136/ebn.11.1.6
13. Lynch EA, Mudge A, Knowles S, Kitson AL, Hunter SC, Harvey G. “There is nothing so practical as a good theory”: a pragmatic guide for selecting theoretical approaches for implementation projects. BMC Health Serv Res [Internet]. 2018;18:857. doi: https://doi.org/10.1186/s12913-018-3671-z
14. Albornos-Muñoz L, González-María E, Moreno-Casbas T. Implantación de guías de buenas prácticas en España. Programa de centros comprometidos con la excelencia en cuidados. MedUNAB [Internet]. 2015;17(3):163-169. doi: https://doi.org/10.29375/01237047.2383
15. Registered Nurses’ Association of Ontario. (RNAO). Herramienta de implantación de buenas prácticas. Segunda Edición [Internet] Toronto:RNAO;2012. Avaliable from: https://rnao.ca/sites/rnao-ca/files/Toolkit_en15.pdf
16. Registered Nurses’ Association of Ontario (RNAO). Social Movement Action Framework [Internet] Toronto:RNAO;2020. Avaliable from: https://rnao.ca/leading-change-toolkit
17. NHS England and NHS Improvement. Quality, Service Improvement and Redesign ((QSIR) Tools: Sustainability Model [Internet]. United Kingdom;AQUA:2023. Avaliable from: https://aqua.nhs.uk/qsir-tools/
18. Parra-Flores M, Souza-Gallardo LM, García-Correa GA, Centellas-Hinojosa S. Incidencia de infección asociada a catéter venoso central y factores de riesgo relacionados en pacientes con nutrición parenteral total en un hospital de tercer nivel. Cir Cir [Internet]. 2017;85(2):104-108. doi: https://doi.org/10.1016/j.circir.2016.05.013
19. Martínez-Ortega C, Suarez-Mier B, Del Río-Pisabarro C, Cantero M, Llinas M. Prevención de complicaciones relacionadas con accesos vasculares de inserción periférica. Programa Flebitis Zero. Línea estratégica III: Prevención. Plan Nacional de resistencia Antibióticos. Agencia Española de Medicamentos y Productos Sanitarios (AEMPS) [Internet]. 2019. Avaliable from: https://www.resistenciaantibioticos.es/sites/default/files/documentos/programa_flebitis_zero.pdf
20. Flebitis Zero. [Internet]. España;2023. Avaliable from: http://flebitiszero.com/app/
21. Liu C, Chen L, Kong D, Lyu F, Luan L, Yang L. Incidence, risk factors and medical cost of peripheral intravenous catheter-related complications in hospitalised adult patients. J Vasc Access [Internet]. 2022;23(1):57-66. doi: https://doi.org/10.1177/1129729820978124
Notas de autor
María Ángeles Cidoncha Moreno. Instituto de Investigación Sanitaria Bioaraba, Prevención, Promoción y Cuidados en Salud. Osakidetza, Dirección General, Subdirección de Enfermería. Álava Street 45, 01006, Vitoria-Gasteiz. Álava, Spain. Email. mariaangeles.cidonchamoreno@osakidetza.eus
Información adicional
Author Contributions: MACM and LGE. Were responsible for the study design and data collection. MACM, LGE, and IMC. Discussed and analyzed the data and drafted the manuscript.
How to reference: Cidoncha-Moreno MA, Garate-Echenique L, Moro-Casuso I. Evaluation of the Implantation of the Good Practices Guideline for Vascular Access in the Euskadi Regional Host of Osakidetza-Basque Health Service: A Descriptive Studio of the Process. MedUNAB [Internet]. 2023;26(2):187-199. doi: https://doi.org/10.29375/01237047.4631
Enlace alternativo
https://revistas.unab.edu.co/index.php/medunab/article/view/4631 (html)
https://revistas.unab.edu.co/index.php/medunab/article/view/4631/3879 (pdf)
https://revistas.unab.edu.co/index.php/medunab/article/view/4631/3880 (pdf)
https://revistas.unab.edu.co/index.php/medunab/article/view/4631/3898 (xml)
