Article
The importance of quality management system to certify radioprotection professionals in the medical field
The importance of quality management system to certify radioprotection professionals in the medical field
Brazilian Journal of Radiation Sciences, vol. 11, no. 1, e2098, 2023
Sociedade Brasileira de Proteção Radiológica
Received: 02 September 2022
Accepted: 28 December 2022
Published: 03 March 2023
Abstract: Certifying professionals in radioprotection (RP), according to the requirements and qualifications established in current Brazilian legislation, ensures that procedures and practices in radiology, radiotherapy and nuclear medicine be performed based on the fundamental principles of radioprotection. However, in none of the regulations and resolutions of the country involving the use of ionizing radiation, the qualification in RP for interventional physicians, radiologists, radiotherapists, nuclear physicians, dentists, nurses and auxiliaries, specialists in medical physics, radiopharmaceuticals, technologists and technicians are clearly defined. It is necessary to adoption a certification scheme through the ABNT NBR ISO/IEC 17024:2013 standard, which will benefit the professionals themselves as well as the patients increasing the safety in RP in the country. This is to the fact that there is no harmony in the regulation of these professionals with the responsible bodies, nor uniformity in practices through international recommendations. This paper proposes the implementation of a quality management system to certify these professionals according to the principles of radiation protection through a survey of needs regarding to the certification of people and the evaluation of systems implemented in other countries.
Keywords: Accreditation, standard ISO/IEC 17024, radiation protection in the health area.
1. INTRODUCTION
Certifying professionals in radiation protection (RP), according to the requirements and qualifications defined in current Brazilian legislation, means ensuring that procedures in the areas of radiology, radiotherapy and nuclear medicine are performed in accordance with the fundamental principles of radiation protection. However, certifying professionals in this field requires the adoption of procedures and requirements of a certifying body of persons that is accredited by the General Accreditation Coordination (CGCRE) with the implementation of a Quality Management System (QMS), according to the Brazilian Association of Technical Standards, Brazilian Standards, and ABNT NBR ISO/IEC 17024 [1].
This system will benefit the professionals themselves of radiology, radiotherapy and nuclear medicine services, as well as patients and their families. The necessary harmonization with regulatory bodies will not only increase radiological safety and security in the country as well as improve different practices to attend international recommendations. The qualification and competence of the respective health professionals through training programs become the support of RP in these practices [2]. Brazil, as a Member State of the United Nation (UN), adopts some of the recommendations of the International Atomic Energy Agency (IAEA) in its national regulations, of which it is a signatory, becoming a consistent and reliable means of fulfilling its obligations [3]. The responsibilities of the federal government with radiation protection and safety include ensuring that measures are in place for: specialized technical services, covering dosimetry, calibration and testing laboratories, radiation individual and area monitoring and radiological assessments. An important part of its actions is the provision of training, specialization and continuous training tools and courses in the most diverse applications of ionizing radiation. In this context, the government is expected to ensure that associations of technical standards and relevant professional bodies interact cooperatively with the radiological protection regulatory body and the competent health authority to ensure the dissemination of knowledge and adequate training of the professionals [4].
It is known, for example, that the regulatory body of the nuclear area is responsible for the authorization of professionals working in these facilities and it is still up to it to answer for the final destination of radioactive waste generated in the country [5]. It is also known that the radiation facilities of the medical field should be authorized by the local health entity so that the requirements of the safe use of ionizing radiation are met. Obviously, for this, it is essential to coordinate and collaborate between the health authority - in this case the National Health Surveillance Agency (ANVISA) - and the regulatory body in RP and nuclear safety, National Nuclear Energy Commission (CNEN). It is certain that by the standards CNEN NN 6.01/1999 [6], NN 3.01/2014 and its regulatory positions [7], NN 3.05/2013 [8], NN 6.10/2017 [9] and NN 7.01/2020 [10], the regulatory body CNEN provides the respective radioactive facilities of nuclear medicine and radiotherapy with the necessary guidance for the safe use of radiation. ANVISA, together with state, municipal and Federal District health surveillance (VISA), establish technical policies and regulations for the safe use of ionizing radiation in the medical field through the Collegiate Board of Directors Resolution, Ordinance RDC no. 20/2006 [11], Resolution no. 38/2008 [12] and RDC no. 611/2022 [13] which reinforce theoretically the radiation protection policies in this area. This establishes the sanitary requirements for all medical services, addressing also organization and operation conditions that should be attend in dental, veterinary, diagnostic and interventional radiology services, regulating the control of medical, occupational and public exposures and requiring the adoption of a quality assurance program. However, in terms of radiological protection, CNEN standards are referenced by ANVISA resolutions, reinforcing the radiological protection requirements.
None of the country's standards, regulations or resolutions, however, is even clearly defined the qualification in RP for interventional physicians, radiologists, radiotherapists, nuclear physicians, dentists, nurses and auxiliaries, specialists in medical physics, radiopharmaceuticals, technologists and technicians to perform their duties with the use of ionizing radiation in health services. In this way, there are international recommendations regarding IRPA certification and description of responsibilities by the IAEA. Consequently, a general methodology is developed here to implement a Quality Management System (QMS) according to the ABNT/NBR ISO/IEC 17024:2013 Standard for the certification of professionals in radiation protection of the health area, aiming at accreditation with the competent body in the country. The specific objectives are: a) to conduct a survey of the needs of the country in relation to the certification of health professionals; b) to evaluate the systems implemented in other countries according to International Organization for Standardization, ABNT NBR ISO/IEC 17024; c) to develop a certification system to be applied to health professionals who use ionizing radiation for the various functions and d) to propose a management system model for a certifying body of people in radiation protection that reaches these professionals.
2. MATERIALS AND METHODS
2.1. Certification in Brazil
Following the example of other countries, the General Accreditation Coordination of INMETRO (CGCRE) is the only accreditation body in Brazil. In this, only one accreditation body is recognized per country and there is a clear and objective separation between accreditation and certification activities [14]. The Brazilian Standard that contains the principles for a body that certifies people, according to specific requirements - including the development and maintenance of a certification scheme for people in any area of activity - is the ABNT NBR ISO/IEC 17024:2013 standard. It is therefore necessary that the scope of certification for a given function aligns with legal regulatory requirements. The certification body shall consider the requirements or guidelines derived from the regulatory body, especially if the process requires regulatory approval. If the process is voluntary, the same rule must be followed. There are four main components for the certification body to define requirements: knowledge, skills, competence and experience. In the requirements of knowledge and skills, the first aspect considered is the level of education. All certification schemes have detailed competency requirements, ensuring that a certified candidate is able to act effectively and independently in relevant practical situations. The review of experience requirements within the processes shows the existence of an interval of two to six years, marked by time and specificity of the experience [15].
2.2. Certification schemes
Each certification process defines the evaluation methods. This is perhaps the most challenging aspect of certification; and there is a wide range of approaches in the processes. Some define what the candidate should present, whether there is a need to participate in a theoretical and/or practical examination, or interview. The following is a summary of two important certification schemes, among several applied at the international level.
USA. The certification of professionals in RP is carried out through the American Board of Health Physics (ABHP). In addition to the ABHP, there is the American Board of Medical Physicists (ABMP) that certifies medical physicists in their subareas, as well as the American Board of Radiology (ABR), which certifies medical physicians and radiologists. Another institution is the National Registry of Radiation Protection Technologists, which certifies technicians and technologists in RP. The requirements for certification are: schooling, knowledge assessment, competence and experience with renewal of the certification process after 4 years. Other international schemes can be found in the reference [15].
Brazil.In medical applicationsthis is actually performed by National Nuclear Energy Commission, CNEN, which certifies radiation protection officers (RPO) in the areas of nuclear medicine and radiotherapy. Certification requirements are: schooling, knowledge, competence and proven experience. The evaluation consists of two theoretical or practical tests, one of general knowledge and the other of specific knowledge. Renewal of the certification takes place every 5 years [10].
2.3. Regulation of certification schemes – OPC
The Brazilian regulation of certification schemes for professionals in a given area occurred with the publication of ISO/IEC 17024 Standard in 2003 aiming to harmonize the certification process of people worldwide. This first edition was based on European Standard EN 45013:1989 [16, 17]. The requirements of this standard are "ensuring that certification bodies that develop certification schemes act consistently, comparably and reliably". The ABNT NBR ISO/IEC 17024 standard contains the necessary requirements for a certifying body of persons to certify the competence of a professional in accordance with the items contained in the specific regulations of his profession. This includes the development and maintenance of a people certification scheme, as well as the implementation of a management system, to administer the certification process [14].
2.4. Accreditation in Brazil
It is granted by an authorised body called the accreditation of the technical competence of the Conformity Assessment Body (OAC) for products, processes, services and management or personnel systems. As CGCRE is the official accreditation body in Brazil, it is up to the organizations accredited by it to conduct compliance certification activities [14]. A certifying body of persons, when accredited by CGCRE, has recognized the competence to evaluate a candidate for certification, based on the scope previously defined in the accreditation process [14, 15]. The criteria established for the accreditation of a Body of Certification of Persons (OPC) are set out in ABNT NBR ISO/IEC 17024 and in INMETRO document NIT-DICOR-004. CGCRE maintains an up-to-date database with the accreditation status of each OPC in the country [18].
2.5. Methodology
The methodology adopted in this study consisted of: a) analysis of regulations that addresses the requirements of radiation protection in the medical field in order to understand the national panorama of certification, still conducting a research of models of certification schemes applied in other countries; b) understanding the requirements of ABNT NBR ISO/IEC 17024 Standard [14]; c) development of a management proposal to be adopted to implement a system according to ABNT NBR ISO/IEC 17024 standard. The results and discussions presented led to the elaboration of a methodology to implement the ABNT NBR ISO/IEC 17024 standard in a structured, able and capable class institution or association to adopt the certification program in radiation protection of professionals in the medical field [19].
3. RESULTS AND DISCUSSION
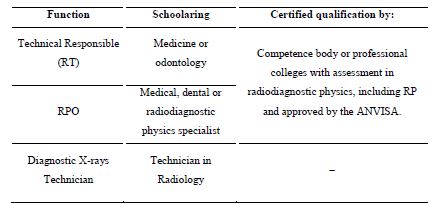
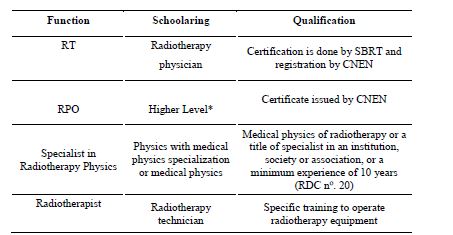
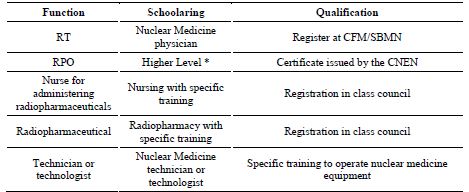
The summary of the analysis performed on the qualifications and professional requirements demanded by RP regulations in the field of nuclear medicine, radiotherapy and diagnostic or interventional radiology, covering RDC no. 20, RDC no.38, RDC 611/2022, are shown in Tables 1, 2 and 3, respectively [11,12,13]. In time, with the recent edition of RDC no. 611, Ordinance no. 453 and RDC no. 330 were revoked [13]. These previous resolutions and ordinances that have already been revoked did not adequately address, and therefore did not solve the issue addressed in this text, that is, the certification of professionals. From these it can be seen that national regulations have requirements that call for certification in RP for professionals involved in the medical field. However, there are still roles that do not fulfill such a requirement or specify the need for qualification. So even if for some roles there is the qualification specified in the regulation, there is no certification body for this professional, resulting in legislation that cannot be fulfilled by the regulatory body. However, this recent RDC no. 611 remains silent on the training and certified qualification that makes up the professional requirement.
Note: The newly published RDC 611 replaced, but did not change, the qualification requirements contained in the former Ordinances 453 and 330.
* Fields of knowledge: engineering, exact and earth sciences, biological, health, agrarian or radiological.
* Fields of knowledge: engineering, exact and earth sciences, biological, health, agrarian or radiological.
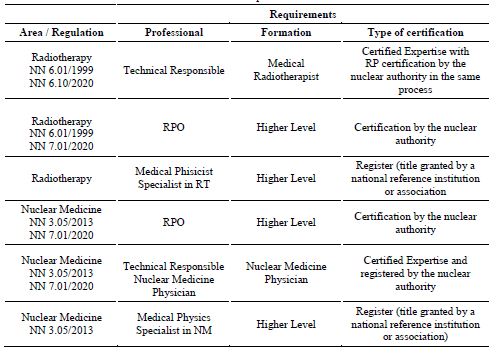
Table 4 shows the nuclear safety standards that regulate radiological protection in the medical field, and it is possible to evaluate the requirements for professional certifications. However, it can be observed that the radiology field does not have a RPO certification process established by this regulatory body. Currently in Brazil, there is no institution which offers this certification in RP for physics professionals in this field, and for dentists, despite the qualification requirements being defined in the recent published RDC no. 611. In addition, in the aforementioned Ordinance, there were no requirements for qualification for the function of diagnostic X-rays technician nor for technologists; much less a certification requirement for such professionals, as there is required in RDC no. 611. The certification of the Technical Responsible (RT), radiologist, is performed by a class collegiate, the Brazilian College of Radiology, CBR [13].
The standards CNEN NN 6.01 and NN 6.10, and the RDC no. 20 for professionals in the radiotherapy sector define well the attributions of these professionals in the area of treatment planning and quality control of equipment [6, 9, 11]. However, they do not mention certification requirements for professionals in this area, nor for professionals responsible for equipment maintenance. CNEN NN 6.02 mentions that a technical report issued by a professional responsible for the shielding calculation project is required to grant authorization for the construction of a radioactive facility, whether in radiology, nuclear medicine or radiotherapy [20]. However, the basic qualification requirements needed to certify this professional are not described.
Currently these projects, because they play an important function in RP, are being conducted by physicists, engineers and architects [21]. The CNEN NN 6.01 mentions also the professional for self-shielded cell irradiators, which can be blood irradiator operators in hospitals and gamma cell in a research laboratory [6]. However, the number of professionals working in this area is also not registered in any database and their certification by qualification is not established, much less specified in regulations. For professionals working in nuclear medicine, the qualification requirements for the technical manager and RPO are defined in the RDC nº 38 and NN 3.05 [7, 12].
These regulations also cite the professional with the qualification of medical physics specialist in a nuclear medicine (NM) service, but do not specifically refer to the medical physicist in NM so certified. The IAEA General Safety Requirements, GSR Part 3 [22] cites the role of the medical physicist, their qualifications and their responsibilities, and among these are the optimization of radioprotection in medical exposures and assessment of doses delivered to the patient [2]. However, in CNEN NN 3.05 [8], the responsibility for dose planning in radionuclide therapy is not assigned to the medical physicist, unlike what occurs in other countries and in the IAEA recommendation [4,22]. Still in the field of NM, the RDC no. 38 mentions professionals who are extremely important in the preparation and administration of radiopharmaceuticals to be used in treatment or diagnosis - nurses and radiopharmacists - who work in radiopharmaceutical production sites by cyclotron and also in centralized or industrial radiopharmacies [12].
These functions, however, do not even have a certification requirement in RP established in Brazilian standards or regulations in force, or even a certification body recognized in Brazil. Differently, the IAEA Safety Standards Series SSG 46 describes that their qualification is acquired by additional specialized training or post-graduate courses, and cites the need for evaluation of education, training, qualification and competence of these professionals in NM services [4]. In compliance with standard CNEN NN-7.01 [10], the nuclear authority performs the certification of RPO in nuclear medicine and radiotherapy, requiring a graduate level qualification in the knowledge areas: exact and earth sciences, biological sciences, engineering, health sciences, agricultural sciences, or radiological sciences, in a certification process of people that are not accredited by CGCRE.
The results of the evaluation of the RP regulation in Brazil have shown that there is urgency in the implementation of a certification process for professionals in radiation protection in the medical field, encompassing all the functions mentioned in the regulations, in order to obtain greater safety for patients, workers and family members or caregivers of the patients, with a view on the optimization of the procedures performed. As currently in Brazil, it is emphasized, there is no OCP in the RP area accredited by CGCRE, the certification of these professionals has been performed by class councils or private institutions, without control of the regulatory body or the country's accrediting body, except for the certification of RPO in nuclear medicine and radiotherapy. Therefore, it is urgent to act on the regulations of certification requirements for some professions, seeking greater clarification regarding the approval of certificates or registrations required, as well as uniformity in the requirements of the functions cited in more than one standard or regulation.
Analysis of certification models for RP professionals around the world - Existing certification schemes differ in scope of application, knowledge requirements, skills, competencies, experience, and assessment methods. It was evident, however, that each model must meet both regulatory and professional expectations in each country for the demonstration of radioprotection knowledge. It was noted that in some countries certification is not carried out by the regulatory body in RP, but by non-governmental non-profit entities, sometimes directly linked to class societies or the IRPA (International Radiation Protection Association). In none of the certification schemes analyzed was there direct application or accreditation to ABNT NBR ISO/IEC 17024 [1], although in some schemes the requirements necessary for its implementation are met.
The RP certification process carried out by the Brazilian regulatory body is based on the differentiated certification method by field of application, serving the area of nuclear medicine and radiotherapy. In the certification of RPO in these specialties, the institution itself plans and executes the process. But in the certification of medical specialist in nuclear medicine and radiotherapy, the authority participates in the process of other institutions, not being the final certifying body, recognizing the certificate issued by this process for the accreditation of the professional. The current scheme of certification of RPO of CNEN has some basic requirements for compliance with ABNT NBR ISO/IEC 17024 [1]. For full implementation of the standard, however, there is a need for reformulation and adequacy of this process. There is also the need to increase the scope of the certification process so that all the functions mentioned in the regulations and legislation in force in radioprotection is contemplated.
Management system of a certifying body for RP professionals - According to ABNT NBR ISO 9000:2015 [22] a quality management system (QMS) "comprises activities by which the organization identifies its objectives and determines the processes and resources necessary to achieve the desired results." By implementing a QMS according to ABNT NBR ISO/IEC17024 [1], an organization will be able to act as a certifying body for professionals in RP. The implementation of a QMS is based on the seven principles of quality management: customer focus, leadership, people engagement, process approach, improvement, evidence-based decision making, and relationship management. [22]. The first step in implementing a quality standard is defining the scope of the QMS. In this case, the scope in question will be the certification of people in radioprotection in the medical field. Thus, the competence of the medical professionals will be attested by the certificate obtained, increasing the safety in the use of ionizing radiation in medicine. It is important to highlight that when developing a management system for a RP personnel certification body in the medical field according to ABNT NBR ISO/IEC 17024, all the functions existing in radiology, radiotherapy, and nuclear medicine services must be included, even if the certification requirement for some of these professions is not currently included in the Brazilian regulations. Thus, the detailed scope of the RP personnel certification body is described in Table 5.
The certification body is responsible for deciding on the entire certification process, from granting and maintaining certification to its cancellation, and may not delegate this function. After the scope definition, the legal aspects of the organization and the management of impartiality and confidentiality must be defined. The certification body must establish, document, implement, and maintain a management system to demonstrate and support the implementation of ABNT NBR ISO/IEC 17024. Top management involved in the certification process must establish and document the policies and objectives of certification. It must appoint a member to have responsibility and authority for ensuring that the QMS is implemented and maintained; it must report on the performance of the QMS and any improvement or adverse situations in the certification process. Detailed information about the structuring, control of records and documents necessary to implement this certification body is included are obtained in a previous paper [19]. It is also shown how to create a Certification Committee, responsible for final decisions concerning certification, as well as the results of appeals and complaints, emphasizing that the members of this Committee should be experienced professionals and have knowledge about the certification process in the various areas of scope, to verify that all requirements have been met. The certification body must have a scheme for monitoring the performance of the examiners and the reliability of the judgments made by them.
The activities of the certification body must be carried out in adequate facilities, with equipment, examination centers, and resources capable of carrying out certification. Records must make it possible to identify the status of a person's certification and provide information to make its validity available to the public.
The certification application process must be documented and records must be maintained by the certification body, which must review the application to confirm that the application meets the requirements defined in the certification scheme. The certification body must have documented the method and mechanisms for evaluating candidates in a structured and planned way, which may be conducted by written, oral or practical examination, developed according to the requirements defined in the certification scheme. The decisions related to all stages of certification (from granting to renewal of the certification including suspension) are the responsibility of the certification body, through the Certification Committee, and the outsourcing of this stage is prohibited. The workforce involved in this certification decision stage cannot be part of the evaluation process (examination) and candidate training. The certificate must be signed or authorized by the responsible member defined in the body's structure, and be drawn up in such a way as to reduce possible forgeries. For suspension, cancellation, or reduction of the scope of certification, the body must document a policy and procedure containing the subsequent actions to be taken in these cases. For renewal of the certification the documented procedure must describe the process that asserts the maintenance of the renewal of the certification candidate's competence in accordance with the requirements defined in the scheme.
The timeframe defined for renewal of the certification must also be documented and in accordance with the requirements of the certification scheme, regulatory requirements, labor market, technological advances in the field, inherent risks of incompetence in the work, and the frequency and content of supervision activities defined. In order to verify the effectiveness of the implementation of all the requirements of ABNT NBR ISO/IEC 17024, the certification body must have procedures for internal audits. Audits must be conducted by competent personnel, knowledgeable in certification and auditing, and independent of the work being audited. At least once every 12 months, the top management of the body must conduct a critical review to review its QMS, including compliance with its policies and objectives, and analyze internal and external audits, comments from candidates, impartiality and confidentiality of the process, preventive and corrective actions taken, follow-up on actions from previous critical reviews, verification of opportunities for improvement and changes affecting the QMS, resource requirements, and results of appeals and complaints [19].
Certification schemes - The following must be defined and documented in the scheme: criteria, methods and mechanisms for application and assessment for initial certification and renewal of the certification, methods and criteria for supervision, criteria for suspension and withdrawal of certification, and criteria for changing the scope or level of certification. The body shall document and demonstrate that the development and review of the certification scheme includes: the involvement of qualified experts in each assessment area, the use of an appropriate framework, the definition of prerequisiteswith the competence requirements of the profession, and integration of the assessment mechanisms with the competence requirements [19]. It is important to note that for each function described in Table 5, it is necessary to define a certification scheme, with its scope, requirements, criteria, and assessment methodology.
Management System Documents - A management system is based on a set of documents, and at the top of this hierarchy is the Quality Manual (QM). This is the document that guides the entire implementation of the QMS according to ABNT NBR ISO/IEC 17024. It will contain the objective of the document, the scope of the QM, information about the entity that represents the certification body, such as location, history, work objectives, organizational structure, description of functions, and the hierarchy of documents that are part of the QMS. It should also contain the statement that the responsibility for the decision on certification lies with the body through the Certification Committee [19]. It also cites the structure and responsibility of the Certification Committee in the process. A computerized system to control documents and records is very useful in a QMS; as well as, for the certification body, an electronic system for question bank, exam preparation and evaluation of candidates' results is very interesting and practical. The exams, depending on the requirements of the category being assessed, can be conducted remotely and virtually. Currently there are some digital platforms that do this data management in a practical and reliable way.
4. CONCLUSION
As the certification model currently applied in Brazil is incomplete and needs improvements so that it covers all the professionals occupationally exposed to ionizing radiation in the medical field, it is necessary to adjust the current process conducted by the national regulatory authority. This process should cover all professionals who need PR certification, such as those working in radiology, radiotherapy and nuclear medicine, besides improving the current process for certification of radiological protection officers (RPO).
Through the analysis of legislation and radiation protection standards, the importance of certification for professionals in the medical field, covering radiology, radiotherapy, and nuclear medicine, was demonstrated. To increase the safe use of ionizing radiation, it is important that all professionals mentioned in the regulations are certified in RP, thus requiring four, eight, and six certification schemes in radiology, radiotherapy and nuclear medicine, respectively. The study of certification schemes around the world has shown that there are different certification models that can be used as references.
A management system according to ABNT NBR ISO/IEC 17024 is essential for a personnel certification body to conduct the process with appropriate excellence and quality. Thus, it was revealed here the urgency of training a certification body for RP professionals in the medical field, provided it is accredited by CGCRE, since the institutions that currently perform this task precariously and are not accredited by the only accrediting body in the country.
The requirement of certification in RP for medical professionals, the accreditation by CGCRE of a certifying body of persons in RP with a quality management system according to ABNT NBR ISO/IEC 17024, would make Brazil a safe country in the use of ionizing radiation in medical practices as in radiology, radiotherapy and nuclear medicine, following the international guidelines. The implementation of this certification process in radiation protection in the medical field in the national regulatory agency benefits both the professionals themselves and the patients in medical practices that involve the use of ionizing radiation in diagnostics and/or therapy procedures.
REFERENCES
ABNT NBR ISO/IEC 17024 – International Organization for Standardization, Conformity assessment — General requirements for bodies operating certification of persons, ISO, 2013.
IAEA - International Atomic Energy Agency, Radiation Protection and Safety of Radiation Sources: International Basic Safety Standards - General Safety Requirements Part 3 Nº GSR Part 3, Vienna, 2014.
IAEA - International Atomic Energy Agency, Governmental, Legal and Regulatory Framework for Safety - General Safety Requirements NºGSR Part 1 (Rev. 1), Vienna, 2016.
IAEA - International Atomic Energy Agency, Radiation Protection and Safety in Medical Uses of Ionizing Radiation, IAEA Safety Standards Series no. SSG-46, Vienna, 2018.
BRASIL, Lei no. 7.781, de 27 de junho de 1989.
CNEN NN 6.01 - Requisitos para os registros de pessoas físicas para o preparo, uso e manuseio de fontes radioativas, Resolução CNEN 005/99, março de 1999.
CNEN NN 3.01, Diretrizes básicas de proteção radiológica, Resolução CNEN 164/14, março 2014.
CNEN NN 3.05, Requisitos de segurança e proteção radiológica para serviços de medicina nuclear, Resolução CNEN 159/13, dezembro 2013.
CNEN NN 3.10, Requisitos de segurança e proteção radiológica para serviços de radioterapia, Resolução CNEN 267/20, outubro 2020.
CNEN NN 7.01 Certificação da qualificação de supervisores de proteção radiológica, Resolução CNEN 259/20, março de 2020.
ANVISA- Agência Nacional de Vigilância Sanitária – Resolução RDC no. 20, de 02 de fevereiro de 2006.
ANVISA - Agência Nacional de Vigilância Sanitária – Resolução no. 38, de 04 de junho de 2008.
ANVISA - Agência Nacional de Vigilância Sanitária – Resolução RDC no. 611 de 9 de março de 2022.
http://www.inmetro.gov.br/qualidade/index.asp - consulta feita em 05-03-2019.
IRPA- International Professional Association, Guidance on Certification of a Radiation Protection Expert, Paris, 2016.
MORAES, C.S.V.; NETO, C. S.L. Educação, Formação Profissional e Certificação de Conhecimentos: Considerações sobre uma política pública de certificação profissional, Educação & Sociedade, Campinas, vol. 26, n. 93, p. 1435-1469, set./dez. 2005.
https://www.legislation.gov.uk/uksi/2017/1075/contents/made, The Ionising Radiations Regulations 2017, no.1075, UK Statutory Instruments – consulta feita em 10 de outubro de 2019.
INMETRO, NIT-DICOR-004, Critérios para a acreditação de organismo de certificação de pessoas, INMETRO – Revisão 30, maio de 2019.
LUZ, C.P.V.C. Proposta de um sistema de gestão para certificar profissionais em radioproteção na área médica, Rio de Janeiro: Tese de Mestrado. IRD, 2019.
CNEN NN 6.02 – Licenciamento de Instalações Radiativas, CNEN, Brasil, 2014.
LUZ, C. P. V.; SÁ, L. V.; DELGADO, J. U., Radiological protection and certification for medical professionals in Brazil - Brazilian Journal of Radiation Sciences, 2018.
IAEA - International Atomic Energy Agency, Radiation Protection and Safety of Radiation Sources: International Basic Safety Standards- General Safety Requirements NºGSR Part 3 Vienna, 2014.
ABNT NBR ISO 9000:2015 - Associação Brasileira de Normas Técnicas - Sistemas de gestão da qualidade – Fundamentos e vocabulário, Brasil, Terceira edição, 2015.
Author notes
*camille.luz@ird.gov.br
Conflict of interest declaration

![Scope of the certification body for people in RP [19].](../t5.jpg)