Abstract
Background: Several hemodynamic and respiratory variables measured during cardiopulmonary exercise testing (CPX) have been shown to predict survival. One such measure is the cardiorespiratory optimal point (COP) that reflects the best possible circulation-respiration interaction, but there are still limited data on its relationship with adverse outcomes.
Objective: To assess the association between COP and cardiovascular mortality in men aged 46 to 70 years.
Methods: A sample of 2201 men who had anthropometric, clinical, and COP data obtained during cycling CPX between 1995 and 2022 was extracted from the CLINIMEX Exercise cohort. COP was identified as the minimal minute-to-minute VE/VO2 during CPX. Vital data were censored on October 31, 2022 for ICD-10-identified cardiovascular deaths. Cox proportional hazard models were used to estimate hazard ratios (HRs) with 95% confidence intervals (95% CIs).
Results: The mean ± standard deviation age was 57 ± 6 years and the median COP value was 24 (interquartile range = 21.2 to 27.4). During a mean follow-up of 4688 ± 2416 days, 129 (5.6%) patients died from cardiovascular causes. The death rates for low (< 28), high (28 to 30), and very high (> 30) categories of COP were 3.2%, 9.6%, and 18.7%, respectively. Following adjustment for age, history of myocardial infarction, diagnosis of coronary artery disease, and diabetes mellitus, the HR (95% CI) for cardiovascular mortality comparing very high versus low COP was 2.76 (1.87 to 4.07; p < 0.001).
Conclusions: Our data indicate that, for a general population-based sample of men, COP > 30 represents a considerably higher risk for cardiovascular death. Information on COP could assist cardiovascular risk assessment in men.
Keywords: Exercise testing, cardiopulmonary exercise testing, physical fitness, cardiovascular diseases.
Original Article
High Cardiorespiratory Optimal Point Values Are Related to Cardiovascular Mortality in Men Aged 46 to 70 Years: a Prospective Cohort Study
Received: 18 June 2023
Revised document received: 26 June 2023
Accepted: 05 July 2023
Robust evidence has been accumulated over the last decades indicating that high levels of cardiorespiratory (aerobic) fitness are inversely and independently associated with mortality risk.1–5 In this context, a Scientific Statement from the American Heart Association published in 20166 proposed that cardiorespiratory fitness should be considered as a clinical vital sign. More recently, Laukkanen et al.7 published an updated meta-analysis with data from 37 unique studies comprising over 2 million individuals and found a 50% reduction in risk for all-cause mortality when comparing higher and lower cardiorespiratory fitness tertiles.
Cardiorespiratory fitness can be estimated by different methods, using either formulas based on exercise variables such as heart rate, cycling workload, or treadmill speed/incline achieved during exercise testing 8,9 or population-specific questionnaires,10 such as the CLINIMEX Aerobic Fitness Questionnaire.11 However, whenever possible, directly measured cardiorespiratory fitness obtained from a maximal cardiopulmonary exercise test (CPX) is preferable, controlling for inter-individual variability in exercise mechanical efficiency and capacity. Using metabolic analyzers, expired gases are collected and O2 and CO2 fractions analyzed, allowing the determination of ventilation (VE), VO2, and VCO2 at designated time intervals. Combining these respiratory variables, it is possible to calculate ratios, such as the ventilatory equivalent for O2 (VE/VO2). During an incremental CPX, there is a clear U-shaped pattern when plotting VE/VO2 and a minimal value can be easily identified either visually or when a minute-to-minute listing of VE/VO2 values is available. In 2012,12 the CLINIMEX research group initially termed this minimal VE/VO2 in a given minute of an incremental CPX as the cardiorespiratory optimal point (COP) and characterized its sex- and age-related reference values for healthy adults. Since this first report, several other studies13–21 have used COP as a CPX variable with additional information on aerobic fitness and of clinical relevance. Notwithstanding, more information is still needed to confirm its role as a valid CPX measure for survival.
The aims of the current study were: 1) to assess the association between COP and cardiovascular mortality in men aged 46 to 70 years; and 2) to verify whether the pattern of death rates according to COP were similar at each 5-year interval in the 46 to 70 year age range.
Exercise Medicine Clinic (CLINIMEX) is a privately-owned medical enterprise founded at the beginning of 1994, located in Copacabana, Rio de Janeiro, Brazil. Its primary aim is to provide medical services through comprehensive evaluation protocols and advanced medically-supervised exercise programs. The CLINIMEX Exercise open cohort was established in 1994 incorporating all individuals evaluated since. Between January 1994 and February 2023, after excluding those formally classified as athletes,22 a total of 9055 individuals voluntarily underwent a medical-functional evaluation to assess their health and levels of physical fitness and to obtain advice regarding exercise and/or sports practice. The sample was largely white, and most of the individuals belonged to the country's upper socioeconomic and education strata. In general, individuals were referred by their attending physicians, although some were self-referred for the evaluations. Only data from their initial (baseline) evaluation were utilized for this study in those who underwent multiple evaluations.
For this analysis, a pre-defined sex and age range that included data from men aged 46 to 70 years were retrospectively reviewed (N = 2792). A final sample of 2201 individuals was obtained (78.9% of the initial sex- and age-selected sample) after applying the following inclusion criteria: a) having voluntarily agreed to be evaluated and given permission to have their data used for research purposes; b) absence of locomotor or musculoskeletal complaints or clinical restrictions to perform an CPX; c) having undergone a maximal CPX using a leg cycling ergometer; and d) having data available for censored vital status, including height (measured to the nearest 0.1 cm), weight (measured to the nearest 0.1 kg, without shoes and while wearing minimal clothing), BMI (kg/m2), and COP. In addition, information regarding relevant clinical variables were reviewed by the evaluating physician and included as a yes/no classification for: previous medical diagnosis of coronary artery disease; hypertension; dyslipidemia; obesity; diabetes mellitus; and history of myocardial infarction or coronary revascularization (either bypass graft or percutaneous interventions) (see dataset flowchart in Supplementary Figure 1).
All evaluations were carried out by only five well-trained exercise and sport physicians in a standard exercise lab facility, adequately prepared in terms of personnel and equipment to deal with medical emergencies.23 Details regarding the identification and definition of COP are available in previously published studies.12–20 In brief, during an individualized ramp-protocol cycling CPX (Cateye EC 1600, Japan, from 1994 to 2006 and Inbramed CG-04, Brazil from 2007 to 2023) designed to last between 8 and 12 minutes, the expired gases were collected by the use of a Prevent pneumograph (MedGraphics, United States) coupled to a mouthpiece, with concomitant use of a nose clip. The expired gases were measured and analyzed with a TEEM100 system (Aerosport, United States) from 1995 to 1999 and VO2000 metabolic analyzer (MedGraphics, United States) from 2000 to 2023. The systems were calibrated with a 2L-syringe and with gases of known concentrations before the first assessment of the day. Pulmonary VE and O2 and CO2 partial fractions were expressed every 10 seconds and averaged for each minute of CPX using a spreadsheet. The COP was obtained by identifying the minimal VE/VO2 ratio at a given minute of CPX, thus being a non-dimensional value. Since COP is obtained at a relatively low submaximal exercise intensity, 12–14, 16, 21, 24 individuals who did not achieve formal criteria for a maximal CPX were not excluded from the study.
Vital status (alive or dead) was last censored on October 31, 2022, and date of death were obtained from the official records of the Health Secretary of Rio de Janeiro State. ICD-10 coding was used to identify the deaths with cardiovascular diseases as the main cause. The duration of follow-up was expressed in days, calculated as the time between the evaluation and last censored dates. All individuals read and signed a consent form allowing the evaluation to be undertaken and giving permission to use their de-identified results for research purposes. Both the evaluation and research protocols were formally registered with relevant governmental bodies and approved by an external research ethics committee.
Baseline characteristics were presented as mean ± standard deviation for continuous variables and count (percentage) for categorical variables. Depending on the type of variable analyzed, chi-square or one-way ANOVA were used to compare groups. Death rate (%) was separately computed for each decile of COP, enabling the identification of the most appropriate data grouping for survival analysis (see Supplementary Table 1), resulting in three numerically convenient round values of COP for the grouping: low (< 28), high (28 to 30) and very high (> 30). Kaplan–Meier curves were constructed, and log-rank (Mantel–Cox) tests were used to analyze survival times according to COP groups. Cox proportional hazard regression models were then used to estimate hazard ratios (HRs) with 95% confidence intervals (CI) for the association between COP and cardiovascular mortality risk, after confirming no major departure from the assumptions of the proportionality of hazards using Schoenfeld residuals. Data from other variables that were significantly associated with cardiovascular mortality were used as covariates to assess the independence of the association. HRs were calculated and progressively adjusted using the following four models: unadjusted (model 1); age (model 2); age, presence or absence of coronary artery disease, and history of myocardial infarction (model 3); age, coronary artery disease, history of myocardial infarction, and diabetes mellitus (model 4). Selection of covariates for adjustment was based on their previously established role as risk factors for cardiovascular mortality, according to evidence from previously published studies, or their potential as confounders based on known associations with cardiovascular mortality using the available data.
Additionally, a two-way ANOVA (COP groups and age groups) and Tukey's multiple comparisons were conducted to assess the differences in death rates (%) between three COP groups within five distinct 5-year age intervals: 46 to 50, 51 to 55, 56 to 60, 61 to 65, and 66 to 70 years. All statistical analyses were conducted using Prism 9.5.1. (Graphpad, United States), using a two-tailed p < 0.05 as a criterion for statistical significance. This written report adhered to STROBE recommendations for cohort studies.
Table 1 presents results for the main variables of the study displayed in five columns – all subjects, three groups of COP results and p-values for the corresponding one-way ANOVA. The mean ± standard deviation age was 57.0 ± 6.0 years, with a modest (2.9 years) but significant increase from the lowest to highest COP group (p < 0.001). Among COP groups, there were small differences for height and weight, 2 cm and 2.5 kg, respectively (p < 0.001), with the low COP group (< 28) having the tallest and heaviest individuals. When comparing BMI, these small differences in height and weight cancelled out, and the values were remarkably similar, with a maximal difference of 0.2 kg/m2 among the COP groups (p = 0.600). Follow-up time was 4688 ± 2419 days for all individuals. For this sample of men aged 46 to 70 years, percentiles 1, 5, 95, and 99 for COP results were 16.1, 18.2, 34, and 40.6, respectively, with a median value of 24.0. When comparing survivors with those who died from cardiovascular causes, a lower COP was found in survivors, with a difference in median of 5.2. In survivors, the median (interquartile range) was 23.8 (21.1 to 27.0), and, in those who died from cardiovascular causes, it was 29.0 (24.5 to 34.1) (p < 0.001), as shown in Figure 1.
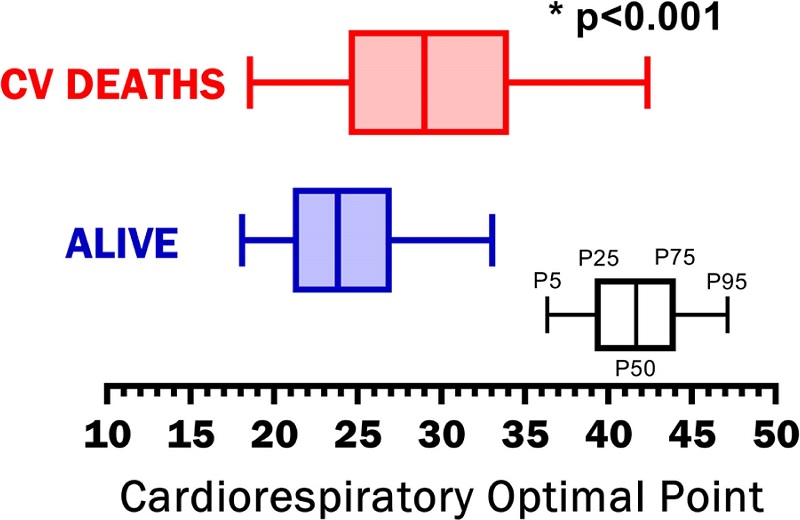
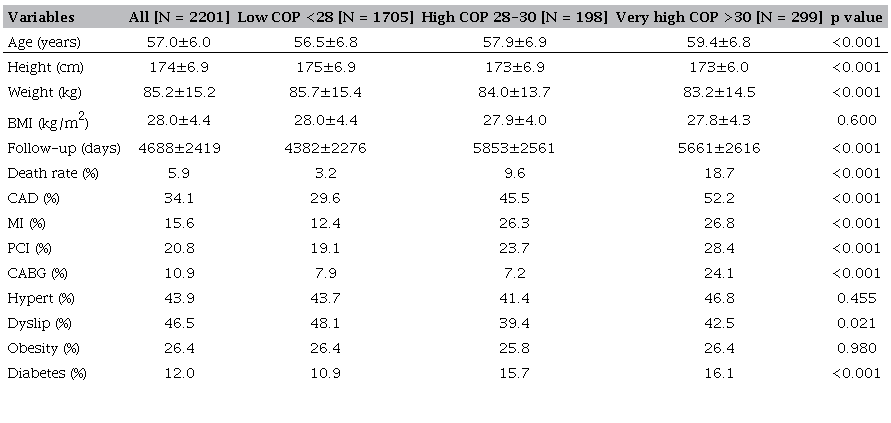
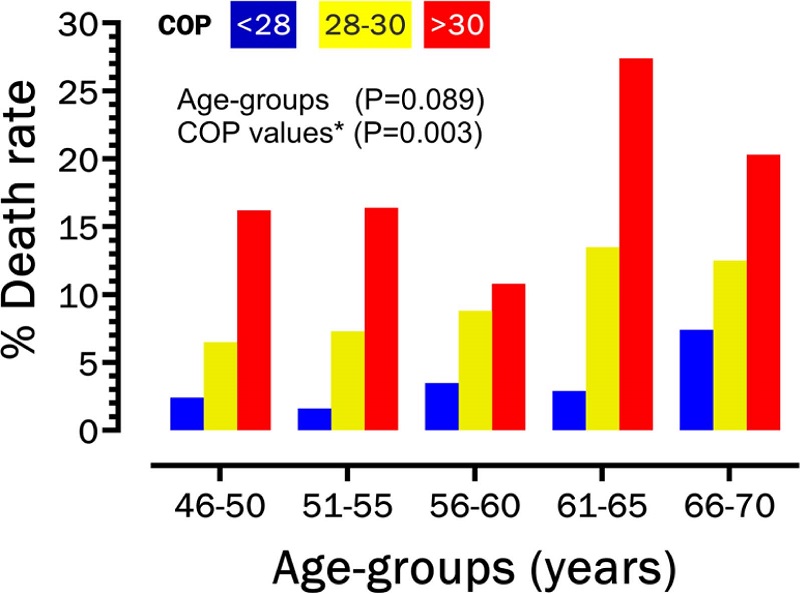
Annual death rate (%) was 5.6% for the entire sample, and it increased nearly 6-fold from the low (3.2%) to very high (18.7%) COP groups (p < 0.001). Figure 2 presents the death rate (%) for low, high, and very high COP groups at each 5-year interval. While the relative death rate (%) systematically increased from low to high COP values in a similar pattern for all five 5-year age intervals (p = 0.089), the absolute death rate (%) tended to progressively increase for all three COP groups with age (p = 0.003). It is also noteworthy that the death rates for older (66 to 70 years) individuals with COP < 28 was similar (7.4%) to those observed in the high COP group (7.3%) at 51 to 55 years. This was less than half that observed among younger (46 to 50 years) individuals (16.2%) with very high COP values, that is, > 30. For the 24 individuals with COP > 40, 8 (33.3%) died from cardiovascular causes during the follow-up.
Data from eight clinical variables were analyzed and further compared among the COP groups. The proportion of subjects classified (yes/no) as hypertensive was 43.9%, and 26.4% were classified as obese. However, no differences were observed among COP groups for either hypertension (p = 0.455) or obesity (p = 0.980). On the other hand, the proportions for all other five clinical variables differed among COP groups, with a trend for increasing percentages in the high and/or very high COP groups (p < 0.001). Most (86.3%) individuals had at least one of these clinical conditions studied: 34.1% had a diagnosis of coronary artery disease, 25.7% had undergone a revascularization procedure, and 15.6% had history of myocardial infarction.
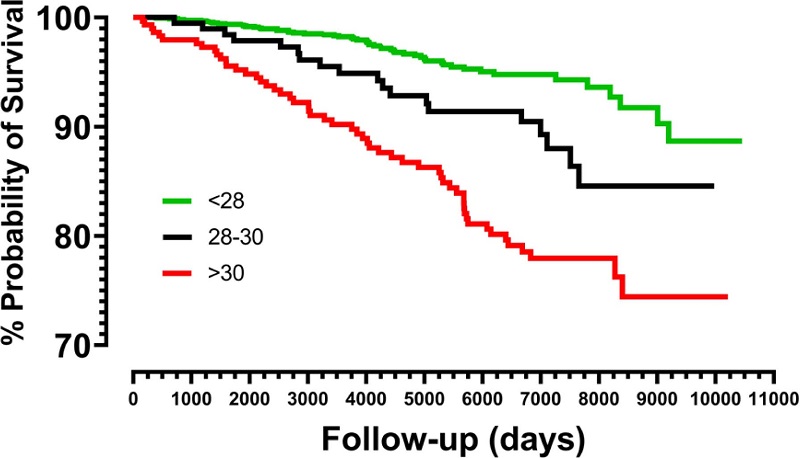
Figure 3 presents Kaplan–Meier curves and cardiovascular mortality for the three COP groups. Log-rank tests indicated that survival curves were different (x2 = 61.01; p < 0.001) with a linear trend for increasing cardiovascular mortality with progressively higher COP values (x2 = 59.91; p < 0.001).
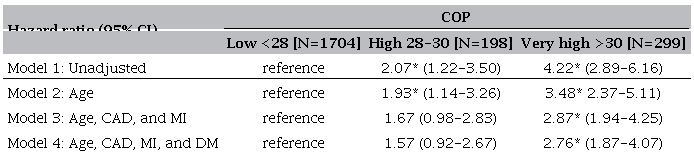
Results of Cox hazard regression modelling are presented in Table 2. The corresponding adjusted HRs (95% CIs) for very high COP when using low COP as a reference were as follows: in model 1 (unadjusted), 4.22 (2.89 to 6.17; p < 0.001); in model 2 (age-adjusted), 3.48 (2.37 to 5.21; p < 0.001); in model 3 (adjusted for age, coronary artery disease, and myocardial infarction), 2.88 (1.95 to 4.26; p < 0.001); and in model 4 (adjusted for age, coronary artery disease, myocardial infarction, and diabetes mellitus), 2.76 (1.87 to 4.08; p < 0.001). HRs for very high and high COP groups were significantly different between the low COP reference group for all models, except for model 4, in which the difference between HR for the high COP group had borderline significance (1.57 [0.90 to 2.62; p = 0.096]).
It is known that physiological variables related to aerobic performance quantified during a CPX, particularly maximum oxygen uptake and COP, are strong predictors of survival in healthy and unhealthy middle-aged and older individuals.6, 7, 15, 18, 25. Since its proposal in 2012,12 COP has been assessed in several clinical and physiological studies, by our research group and others. COP has also been the topic of several editorials26–28 and has been included as a variable to be considered in at least two review papers about CPX.29,30 The current results corroborate previous research findings obtained in distinct cohorts from different countries, including Brazil,20 Finland,17,18 Portugal,31 the United States,15 the Netherlands,14 Belgium,32 and Germany.33 All these studies have supported the clinical relevance of COP and its prognostic role for survival in adult men and women, while adding new information regarding mortality caused by cardiovascular disease in men in a specific age range.
Our data indicate that, for a general population-based sample of men aged 46 to 70 years, COP significantly differs between survivors and those who have had a cardiovascular death. COP > 30 represents a considerable cut-off point for a higher risk of death from cardiovascular cause. Death rate was 18.7% when the COP was > 30 and only 3.2% for those with COP < 28. When controlled for age, history of myocardial infarction, presence of coronary artery disease, and diabetes mellitus, the HR for COP > 30 was 2.76 times higher than for the reference group (COP < 28). COP between 28 and 30 exhibited a borderline increased risk. Notably, the results showed that these risk estimates applied for each 5-year interval in the age range from 46 to 70 years.
To provide clinically relevant information, epidemiological studies should provide an adequate combination of event rates, follow-up period, sample size, and main characteristics of the sample. The minimal required sample size is inversely related to the absolute number of events during the follow-up. The follow-up period should also consider the characteristics of the events being censored as well as age and the typical life expectancy for the age of the individuals included in the sample. These aspects were considered when selecting the sex and the age range of the individuals for this current study. Since previous data have indicated that age-matched healthy men and women differ in terms of COP,12 we selected only individuals of one sex (male). While data were available for younger and older individuals in the cohort, the death rates were too low or too high, respectively, during the available follow-up period to make meaningful and clinically relevant conclusions regarding the association between COP and cardiovascular mortality.
The CLINIMEX Exercise cohort started in 1994. Since then, it has been collecting medical and functional data among individuals of both sexes aged from 6 to 98 years who have voluntarily participated in the evaluation protocol. Notwithstanding, there are some characteristics of this cohort that deserve specific comments. The individuals evaluated in the cohort were primarily white, and they pertained to high educational and economic strata. Thus, they are not generally representative of the Brazilian population. Detailed information about the cohort is available in the supplemental materials of a previously published study.34
In spite of the fact that most of the participants had a diagnosis of coronary artery disease or risk factors at their baseline evaluation, the annualized death rate was about half of that expected for the general Brazilian population for a similar mean age of 57 years.35 This may be due to the demographic characteristics of this sample, as well as the possibility that these individuals had adhered to prescribed optimal medical treatment for their clinical conditions, including the chance to participate in a long-term medically-supervised exercise program.
COP has a very clear and logical physiological background, representing the minimal amount of ventilation required to consume one liter of oxygen and, very likely, reflecting the best possible interaction between circulation (heart and vessels) and respiration (lungs and cells). A previous study has shown that COP is equal or superior to several other CPX-related variables in terms of stability of measurement.36 Given that it occurs at a submaximal exercise intensity, it does not require a maximal CPX, a potentially relevant advantage for several clinical conditions, as shown, for example, in patients experiencing panic disorders.21 In addition, unlike several other commonly used CPX variables such as VO2 max, VO2 peak, anaerobic or ventilatory thresholds, oxygen uptake efficiency slope, the VE/VCO2 slope, and others, the determination of COP by the examiner is extremely easy to accomplish, and it is a practically observer-free error variable. COP has been safely determined in different populations, including patients with heart failure14, 31, 37 and also in individuals over 80 years of age,16 performing submaximal and maximal CPX. Indeed, it is possible for anyone retrieving data from a CPX performed in the past with access to the minute-to-minute listing of VE/VO2 results to precisely identify the COP.
The clinical relevance of COP has been assessed in healthy adult men,15 in healthy middle-aged men,17,18 in healthy and unhealthy middle-aged and older men and women,20 in a small sample of adults with congenital heart disease,33 and in male and female patients with heart failure.14, 31 In a previous study with the CLINIMEX Exercise cohort,20 COP was found to be a strong predictor of all-cause mortality in a broad age range (41 to 85 years) among healthy and unhealthy individuals who were followed for a mean of 6 years. In the United States, Peterman et al.,15 found a significant association between COP and long-term mortality in 3160 apparently healthy individuals (46% women) between 18 and 85 years of age. Laukkanen et al.,17, 18 reported a significant role of COP in predicting all-cause and cardiovascular mortality and sudden cardiac death in a large sample of healthy Finnish men aged 42 to 61 years. Two different European cohorts of patients with heart failure were studied by Reis et al.31 and Kensen et al.,14 and high values for COP were similarly associated with poor survival. In a study conducted at a hospital in Lisbon,31 COP had the highest area under curve for 442 patients with heart failure for cardiac death and urgent heart transplantation over a 12-month period, and COP significantly added to the prognostic power when compared with other CPX ventilatory variables. More recently, Wenhart et al.,33 showed that high COP values provided an objective feature to differentiate lesion severity in adults with congenital heart disease.
Our study confirms the available data from different populations and reinforces COP > 30 as associated with poor survival in men.14, 37 Preliminary evidence from the CLINIMEX Exercise cohort and from a Dutch cohort have suggested that attending exercise-based cardiac rehabilitation programs may reduce very high COP values.14, 38 In this sense, normalizing a very high COP by regular exercise training could be one of the pathophysiological mechanisms leading to better survival in patients enrolled in cardiac rehabilitation programs.39
This study has several strengths, including its planning, design, data collection, and analysis. Data for this study came from a long-term open cohort that has been utilized in several other epidemiological studies.20, 40 This dataset has been carefully cleaned and organized over several years by the principal investigator and close collaborators, making the available information accurate and valid.34 The quality of censoring was very high, being based on official sources reporting ICD-10 classified death certificates and complemented, when appropriate, by the patient's electronic records and by information obtained from attending physicians, relatives, and close friends. Over the 29-year study period, all CPX were conducted in a single center/exercise lab, using a cycle ramp protocol and metabolic analyzers for expired gases from the same manufacturer. All the pertinent medical information and CPX were conducted by only five specialized physicians, following strict protocols, and all data were entered into specifically-designed digital forms (Quattro Pro, Corel, Canada).
In addition, potential confounders were adequately controlled. Moreover, since differences in COP by mode of exercise (treadmill or cycle) have not been formally investigated, we selected only CPXs carried out using a leg cycle ergometer.
Due to the previously detailed demographic and clinical characteristics of the sample extracted from the CLINIMEX Exercise cohort, considerable caution should be applied in generalizing these results for other populations. Since this was a clinical study using a convenience sample of individuals referred to the CPX in our clinic for several distinct reasons, mixing healthy and unhealthy individuals, it was not possible to control for previous or regular use of medications. It has not been formally studied whether there is an influence of specific medications known to have cardiovascular and/or respiratory effects on COP. This should ideally be addressed in studies using repeated CPX with individuals on and off selected medications under well controlled conditions.
No maximal CPX data, such as maximum oxygen uptake or maximum heart rate, were reported, since both submaximal and maximal CPX tests were used to analyze the COP. In this context, no direct or add-on comparisons were made between COP and other hemodynamic and respiratory CPX variables. The feasibility and the validity of obtaining COP in a submaximal CPX was first shown in our study among patients with panic disorders21 and confirmed by data from a Portuguese study of patients with heart failure.31 Nevertheless, it remains to be properly addressed in future studies whether combining COP and VO2 max results would improve risk stratification more than each of these variables separately.
COP is a ventilatory variable obtained at a relatively low level of exercise intensity when ventilation and oxygen uptake data are available (no VCO2 data are needed). COP is also simple to obtain in various populations, including unhealthy middle-aged individuals. This study confirms that very high COP (> 30) is strongly associated with cardiovascular mortality. In addition, in an objective and practical perspective, it is noteworthy that the death rate was very low (2% to 3%) during a mean follow-up of > 12 years for individuals with COP < 28, in the age range of 46 to 70 years. When the COP is available, it may help practitioners to offer more personalized advice, especially for patients with very high COP values (> 30) who may benefit from a more specific exercise prescription and/or referral to a medically-supervised exercise program in clinical practice.
For Supplementary Figure, please click here.
For Supplementary Table, please click here.
To all individuals who were voluntarily tested and who have agreed to have their de-identified data used for scientific purposes. The authors also thank Drs. Claudia Lucia Castro and João Felipe Franca for their help in collecting and collaborating for organization of the data for this study. We thank the Health Secretary of Rio de Janeiro State for providing us access to their vital data records.
There were no external funding sources for this study.
This study is not associated with any thesis or dissertation work.
This study was approved by the Ethics Committee of the CONEP/Brazil CAAE: 40122320.8.0000.9433 under the protocol numbe 5.721.476. All the procedures in this study were in accordance with the 1975 Helsinki Declaration, updated in 2013. Informed consent was obtained from all participants included in the study.
No potential conflict of interest relevant to this article was reported.
Mailing Address: Claudio Gil Araújo, Exercise Medicine Clinic, CLINIMEX. Rua Siqueira Campos, 93-101. Postal code: 22031-071. Rio de Janeiro, RJ – Brazil. E-mail: cgaraujo@iis.com.br




