Original Article
Electrocardiographic Findings Among Asymptomatic Adults With Haemophilia
Electrocardiographic Findings Among Asymptomatic Adults With Haemophilia
International Journal of Cardiovascular Sciences, vol. 37, e20220179, 2024
Sociedade Brasileira de Cardiologia
Received: 25 October 2022
Revised document received: 04 October 2023
Accepted: 08 November 2023
Funding
Funding source: Coordination for the Improvement of Higher Education Personnel Foundation
Contract number: PDSE-88881.362041/2019-1
Funding statement: HemoCardio Study received no funding nor grant to be developed/implemented. RMC received scholarship (PDSE-88881.362041/2019-1) from the Coordination for the Improvement of Higher Education Personnel (CAPES) Foundation, an agency under the Ministry of Education of Brazil, in order to conduct part of his doctoral research as a visiting student at Leiden Universitair Medisch Centrum in the Netherlands.
Abstract
Background: People with haemophilia (PwH) are living longer. Therefore, they can develop atherosclerotic cardiovascular disease (ASCVD). Electrocardiogram (ECG) alterations may be a sign of initial ASCVD before the occurrence of symptoms.
Objective: To describe the prevalence of resting ECG alterations among PwH adults asymptomatic for ASCVD.
Methods: PwH aged ≥ 30 years without previous ASCVD events were considered for the analysis. Resting ECG traces were analysed according to international reference values and the Brazilian Longitudinal Adult Health Study (ELSA-Brasil) results for asymptomatic Brazilian men. Based on the established normal values and using the QT index, we further described the altered ECGs as minor or major changes, according to the Minnesota Code. Differences between prevalences were evaluated by Pearson's χ2 test. Differences between medians were evaluated by the Mann-Whitney U test. A p-value < 0.05 was accepted as statistically significant.
Results: A total of 64 PwH were included in the study. Median age was 44 years (interquartile range 35-52). Most patients had haemophilia A (81%) and 47% were severe. The prevalence of obesity, systemic arterial hypertension (SAH), diabetes mellitus (DM), and dyslipidaemia were 16%, 56%, 14%, and 72%, respectively. All the PwH had sinus rhythm, except for one, who had an implanted pacemaker due to idiopathic third-degree atrioventricular block. Altered ECGs were found in 25% and 30% of PwH, according to established criteria and ELSA-Brasil criteria, respectively. Major changes were found in eight (13%) PwH according to the Minnesota Code, including two ECGs with ischaemia-like wall inactivity.
Conclusions: The prevalence of altered ECG varied from 25% to 30% among asymptomatic PwH.
Keywords: Haemophilia A+ Haemophilia B+ Atherosclerotic+ Atherosclerosis+ Electrocardiography.
Introduction
Haemophilia A and B are rare hereditary bleeding disorders caused by a reduction or absence of the activity of the clotting factors VIII (FVIII) and IX (FIX), respectively. 1 After the introduction of safer factors concentrated on treating or preventing bleeding, life expectancy of people with haemophilia (PwH) has increased. 2, 3 As PwH are ageing, atherosclerotic cardiovascular diseases (ASCVD) may increasingly occur. 4- 7 Many studies suggest ASCVD risk factors are prevalent among PwH. For example, systemic arterial hypertension (SAH) is of utmost relevance, since it is described in up to 60% of PwH aged 30 years or older. 7
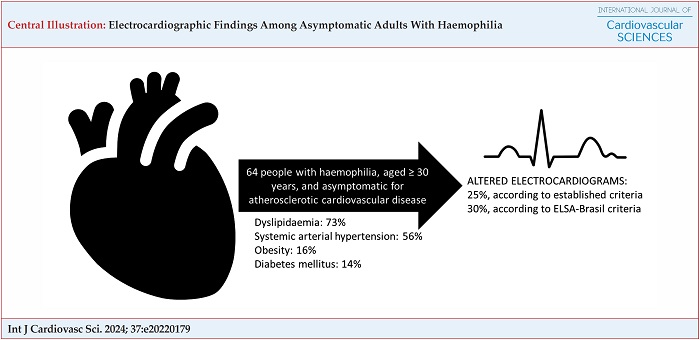
The HemoCardio Study included 64 PwH, aged 30 years and older, and asymptomatic for ASCVD. After resting ECG, we found that 25% were altered when evaluated according to the established criteria, and 30% were altered when evaluated according to the ELSA-Brasil criteria. ELSA-Brasil: Brazilian Longitudinal Adult Health Study.
Resting electrocardiography (ECG) alterations (e.g., ST-segment or T-wave abnormalities, bundle branch block, and left-axis deviation) are associated with an increased risk for subsequent ASCVD events in asymptomatic non-haemophilia adults. 8 The prevalence of major and minor abnormal ECG traces ranged from 8% to 9% and from 8% to 39%, respectively, when evaluated by the Minnesota Code, in populations of 993 up to 5,835 men aged 35 years or more. 8 However, the United States Preventive Services Task Force (USPSTF) recommended against routinely screening with resting ECG of asymptomatic adult population at low risk of ASCVD events. In addition, current evidence was considered insufficient to assess the balance of benefits and harms of screening asymptomatic non-haemophilia adults at intermediate- or high-risk of ASCVD events with resting ECG to prevent ASCVD events. 9 Besides that, there is substantial and consistent evidence that identifying and treating traditional, modifiable ASCVD risk factors such as obesity, SAH, and dyslipidaemia improve cardiovascular outcomes. 10 Nevertheless, they recommended ASCVD prevention by screening and treating risk factors. 9
A recent study reported a high prevalence of cardiac disease signs in advanced ECG performed in asymptomatic PwH. 11 However, there is a paucity of information about resting ECG alterations among PwH in the literature. The current analysis of the Haemophilia Cardiovascular Risk (HemoCardio) Study 7 aimed to describe the prevalence of alterations in resting ECG among asymptomatic PwH. In addition, other risk factors of ASCVD as obesity, SAH, and dyslipidaemia were also evaluated.
Methods
Study design and patients
This cross-sectional study was performed at the comprehensive haemophilia treatment centre of HEMOPE (Centre of Haematology and Haemotherapy of Pernambuco) in the city of Recife (PE), Brazil. 7 The inclusion criteria were male PwH (A or B) of any severity or inhibitor status and aged ≥ 30 years. There were approximately 240 PwH registered in this centre, of whom 120 were ≥ 30 years. All these 120 PwH registered at the outpatient clinic were invited to participate, while attending elective consultations between August 1, 2018 and July 31, 2019. PwH with previous history of ASCVD events were considered symptomatic, and therefore, excluded from this analysis. HEMOPE Committee on Ethics and Research approved the project, and all included patients signed the consent form. Standardized forms were used to collect data through medical file review, patient interviews, physical examinations, blood tests, and ECG trace reports.
Measurements and definitions
Haemophilia history
Clinical data were collected from medical files. We documented age, ethnicity, haemophilia type and severity, inhibitor titre in the previous year, replacement factor regimen (either exclusively episodic or prophylactic), 1 and human immunodeficiency virus (HIV) and hepatitis C virus (HCV) statuses. HIV and HCV statuses are routinely evaluated at HEMOPE, according to international recommendations. 12- 14 When the HemoCardio Study was held, emicizumab and extended half-life FVIII concentrates were not available in Brazil.
Mild, moderate, and severe haemophilia were defined as plasma factor activities between 5% and 40%, 1% and 5%, and below 1%, respectively. 1 PwH were considered positive inhibitors if they presented two consecutive titres ≥ 0.6 BU/mL, at least two weeks apart from each other. HIV infection was confirmed if, after two positive screening tests for HIV antibody enzyme-linked immunosorbent assay (ELISA, there was a positive result on a confirmatory test (Western blot). 14 Patients were considered to have a current or previous HCV infection if they had a reactive HCV-antibody test confirmed by a positive quantitative ribonucleic acid (RNA) for HCV. 13
Atherosclerotic cardiovascular risk factor profile
We collected information about previous history of ASCVD and medicaments in use. Previous history of ASCVD event was defined as a clinical evidence of coronary or cerebrovascular disease or peripheral artery obstruction; significant subclinical disease documented by any diagnostic method (e.g., echocardiography, coronary tomography or resonance, nuclear medicine, or coronarography); or arterial revascularization procedures. Body mass index (BMI) was calculated as weight/(height) 2. A patient was considered obese if his BMI was ≥ 30 kg/m 2. 15, 16 Systolic (SBP) and diastolic (DBP) blood pressures were measured three times and ≥ 5 min apart one from another, by only one researcher, with a mercury-calibrated sphygmomanometer. The patient rested in a seated position for at least 15 minutes. The mean of these three measurements was considered for the analyses. SAH was considered when the mean SBP was ≥ 140 mmHg and/or DBP was ≥ 90 mmHg, or when the patient was on medicament for SAH. 17
Twelve-hour fasting blood tests performed one year before until six months after the inclusion date were considered for this study. Blood glucose, triglycerides (TG), total cholesterol (Tc), low- (LDLc) and high-density lipid cholesterol (HDLc) tests were performed at the local laboratory. A patient was considered to have diabetes mellitus (DM) if fasting blood glucose levels were ≥ 7.0 mmol/L or if he was on treatment for DM. 18 Hypertriglyceridaemia was diagnosed when TG was ≥ 2.3 mmol/L, hypercholesterolaemia when Tc ≥ 5.2 mmol/L, and hypoHDLaemia when HDLc < 1.0 mmol/L. 19 A patient was considered dyslipidaemic if he had hypertriglyceridaemia, hypercholesterolaemia, or hypoHDLaemia. 20
Resting electrocardiography data
Twelve-lead resting ECG was performed using a Burdick Atria 6100 device (Cardiac Science Corporation, Bothell, USA), calibrated at 10 mm/mV and speed of 25 mm/s, on the same day the patient signed the consent form. The patient was placed in supine position for at least 5 minutes beforehand. If needed, the ECG was repeated until minimal or no background noise was evident. Traces were analysed by two researchers (RMC and CGPR), and upon disagreement, a third researcher (AMV) was consulted until a final decision was reached as to the final ECG trace characteristics. Heart rate (HR) and rhythm, premature beats, wave intervals, axis, strains, bundle branch blocks, and Q and ST/T segment alterations were evaluated. QT interval was corrected (QTc) using the Hodges formula: QTc = QT interval+1.75*(HR−60), both intervals in ms and HR in beats/min. 21, 22 Lead II — or lead V5 if lead II could not be read — was used. A tangent against the steepest part of the end of the T wave was drawn. The QT interval started at the beginning of the QRS interval and ended where the tangent crossed the baseline drawn over the PR segment. The upper limit of normality was defined as 440 ms. 23 We analysed the results according to both international reference values 23 and the values for asymptomatic males described by the Brazilian Longitudinal Adult Health Study (ELSA-Brasil), depicted in Table 1. 23, 24 The latter was used to assess further if the results obtained from the current study fitted the results of the non-haemophilia male ELSA-Brasil population. Briefly, the ELSA-Brasil is a population-based cohort study that evaluates the development and progression of chronic diseases, particularly ASCVD and DM. At baseline (2008–2010), the study enrolled 15,105 Brazilians aged between 35 and 74 years. 24 Pinto-Filho et al. 22 evaluated resting ECGs of 5,341 asymptomatic males from ELSA-Brasil. Participants with previous heart disease and ECGs with major alterations according to the Minnesota Code were excluded from the analysis. 22 The described normal intervals found in this ELSA-Brasil male population were used as another reference for the ECG evaluation in this study. 22
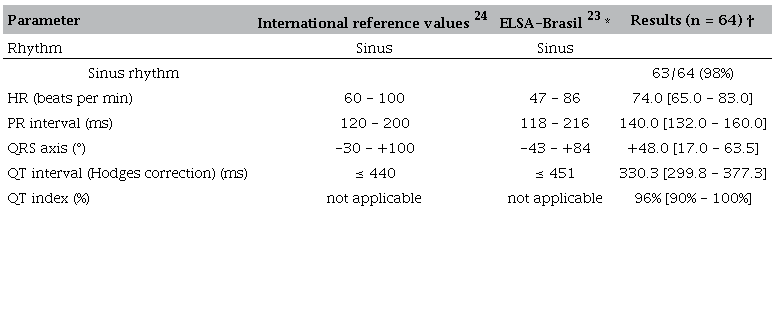
ELSA-Brasil, Brazilian Longitudinal Adult Health Study; PR: segment PR; QRS: complex QRS; QT: segment QT; HR: Heart rate.* Only male asymptomatic people aged 35 years or older were assessed. The values correspond to a CI between 2% and 98%.† Continuous variables are expressed as median (interquartile range). Prevalence values are expressed as absolute cases/total numbers (percentage).
We also classified the ECG trace in conformity with the Minnesota Code. 25 In general, we used the aforementioned international reference values, 23 except for the use of QT index (QTi) instead of QTc by the Hodges formula. QTi was calculated by the formula QTi = (QT interval/656)*(HR+100), considering QT interval in ms and HR in beats/min. QTi ≥ 112% was assumed as an abnormality. The ECG alterations were classified as major or minor, consistent with the Minnesota Code (Supplementary Table 1). 25 Individuals with combined minor and major alterations were classified as having major alterations. 25 Since a previous metanalysis showed that left-axis deviation (MC 2-1) in resting ECG was a clear risk factor for subsequent ASCVD, 8 we considered it as a major alteration, differently from the original Minnesota Code. 25 ECGs from participants without minor or major alterations were classified as normal.
Statistical analyses
Categorical variables are presented as absolute (n) and relative (%) frequencies. Differences between prevalence were assessed by Pearson's chi-square (χ 2) test without Yates’ continuity correction. The normality distribution of the continuous variables was evaluated by the Kolmogorov–Smirnov test. As a result of the small size of the population, the distributions were non-parametric. Consequently, continuous variables were expressed as medians and interquartile range (IQR). Differences between medians were evaluated by the Mann-Whitney U test. The Cohen's Kappa (κ) test (95% confidence interval [CI]) was used to evaluate the agreement between the different methods of ECG evaluation. Strength of agreement was classified as poor if κ ≤ 0.2, fair if 0.2 < κ ≤ 0.4, moderate if 0.4 < κ ≤ 0.6, good if 0.6 < κ ≤ 0.8, and very good if 0.8 < κ ≤ 1. For analytical purposes, the Minnesota Code, which stratifies the ECGs as normal, minor, and major changes, was adapted as a binary score: normal and altered (i.e., the sum of minor and major changes). A p-value < 0.05 was accepted as statistically significant. Data were analysed using Statistical Package for Social Sciences software, version 26 (SPSS®, IBM, Armonk, USA).
Results
Patient characteristics
From 82 PwH who were ≥ 30 years old, 11 (13%) were excluded because of previous ASCVD (n = 6) or missing data on previous ASCVD (n = 5) ( Figure 1). Of the 6 PwH with previous ASCVD, all had an atherosclerotic-related event, being ischemic cerebrovascular event, the most prevalent (50%). Another 7 (9%) PwH were excluded because of missing ECG ( Figure 1). Detailed haemophilia and ASCVD risk profile characteristics of the 64 (78%) included PwH are shown in Table 2. The median age at study entry was 43.5 years (IQR 35.0-51.8). Most patients had haemophilia A (81%) and 30 (47%) were severe. Among the severe PwH, 23 (77%) were under prophylaxis. All PwH were HIV negative, and 53% were current or previously HCV positive.
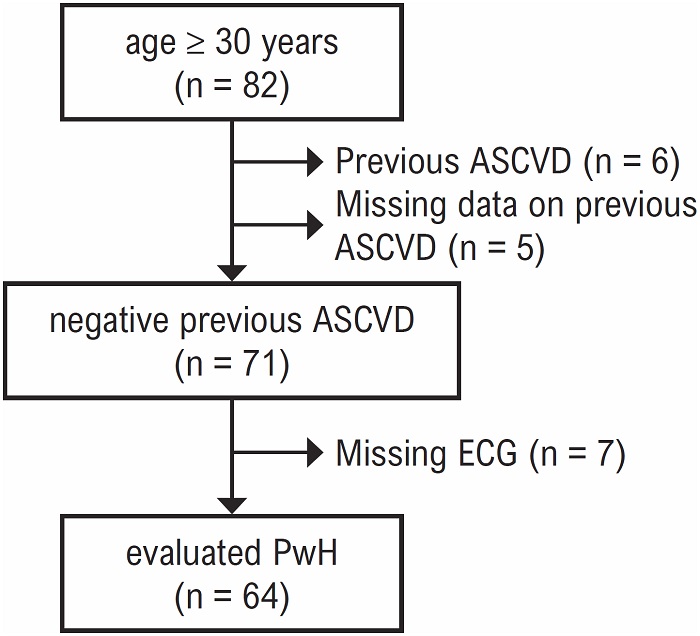
Figure 1
Patients included in the resting electrocardiography analysis of the HemoCardio Study.

Continuous variables are expressed as median (interquartile range). Prevalence values are expressed as absolute cases/total numbers (percentage). HCV: hepatitis C virus; HDLc: high-density lipid cholesterol; HIV: human immunodeficiency virus; LDLc: low-density lipid cholesterol; SAH: Systemic arterial hypertension; SBP: Systolic blood pressure; DBP: Diastolic blood pressure; DM: Diabetes mellitus. Results were calculated based on valid data. The number of missing for each variable is reported in the Supplementary Table 2.
Atherosclerotic cardiovascular risk profile
The ASCVD risk profile data are shown in Table 2 and the corresponding number of missing data of each variable are depicted in Supplementary Table 2. There were 10 (16%) obese PwH. Considering those patients taking anti-hypertensive medicaments (40%) and those who had untreated hypertension, 56% had SAH. DM was considered in 14% PwH. The prevalence of hypertriglyceridaemia, hypercholesterolaemia, and hypoHDLaemia were 21%, 43%, and 26%, respectively. Dyslipidaemia was found in 72% of PwH.
Resting electrocardiography alterations
The characteristics of resting electrocardiographs are depicted in Table 1. The prevalence of altered ECGs is depicted in Table 3. Altered ECGs were found in 25% and 30% of PwH, according to international reference values and ELSA-Brasil criteria, respectively. Observing the Minnesota Code, major changes were found in 8 PwH (12%), including two ECGs with ischaemia-like wall inactivity. There was a fair agreement between the ELSA-Brasil criteria and both the international reference values and the Minnesota Code, while the agreement between the international reference values and the Minnesota Code was very good ( Table 4). Finally, we compared the haemophilia history data and the ASCVD profiles between asymptomatic PwH with normal and altered resting ECGs, following the three evaluation systems. All results were similar between the two PwH populations, except for one in each system. The prevalence of severe haemophilia was higher among individuals with altered ECG than among individuals with normal ECG, when evaluated by the international reference values (p = 0.043; Table 5) and by the binary-adapted Minnesota Code (p = 0.047; Supplementary Table 3). The median (IQR) glycaemia level was higher among PwH with altered ECG than among individuals with normal ECG (p = 0.029), evaluated according to the ELSA-Brasil values (Supplementary Table 4).

Prevalence values are expressed as absolute cases/total numbers (percentage). ELSA-Brasil: Brazilian Longitudinal Adult Health Study; ECG: electrocardiogram.a We considered left-axis deviation (MC 2-1) as a major alteration, according to the results of a previous metanalysis. 8

Kappa coefficients are expressed as median and 95% CI. ELSA-Brasil, Brazilian Longitudinal Adult Health Study.* We considered left-axis deviation (MC 2-1) as a major alteration, according to the results of a previous metanalysis. 8
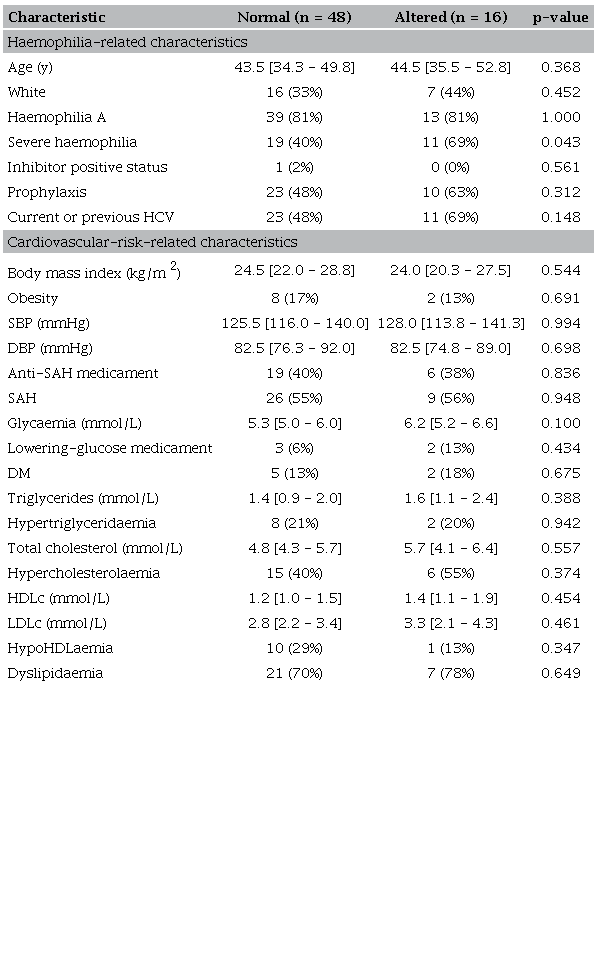
Continuous variables are expressed as median (interquartile range). Prevalence values are expressed as absolute cases/total numbers (percentage). Differences between medians were evaluated by Mann-Whitney U test. Differences between prevalence were evaluated by Pearson's χ 2 test. HCV: hepatitis C virus; HDLc: high-density lipid cholesterol; LDLc: low-density lipid cholesterol; SAH: systemic arterial hypertension; SBP: Systolic blood pressure; DBP: Diastolic blood pressure; DM: Diabetes mellitus. Results were calculated based on valid data. The number of missing for each variable is reported in the Supplementary Table 2.
Discussion
We evaluated ECG changes in a cohort of 64 PwH aged 30 years or older. We found 25% and 30% of PwH with altered resting ECG, in accordance with established or ELSA-Brasil criteria (Central Figure), respectively, and 12% of PwH with major changes on ECG, according to the Minnesota Code. The alterations included a one-third-degree atrioventricular block 26 and two ischaemia-like wall inactivities. Severe haemophilia was associated with altered ECG, according to international reference criteria and the Minnesota Code. Glycaemia levels, but not DM prevalence, were associated with altered ECG, following the ELSA-Brasil criteria. To the best of our knowledge, there are no published data on resting ECG changes among PwH in the literature.
A recent retrospective study evaluated advanced ECG traces of 29 PwH aged ≥ 40 years, in a clinical audit at a Swedish hospital. 11 The advanced analysis is a computer-based platform that determines ECG changes according to vectorcardiography and waveform complexity. 27 They excluded not only patients with previous diagnosed ASCVD, but also those with bundle branch block, sinus tachycardia, non-sinus rhythm and paced rhythm, and pre-excitation. They found that 85% of PwH had trace signs of cardiac disease, which suggests a high prevalence of subclinical heart disease. Instead of using an advanced technique, we manually evaluated the ECG because this was the most widespread, simple, and cheap method used in many clinics. Therefore, it was difficult to compare both studies because the applied methodologies were different. Besides that, we performed two different analyses, using international reference values 23 and values described in a cross-sectional multicentric study with asymptomatic Brazilian men. 22 We also performed a formal evaluation using the Minnesota Code, 25 adapted by considering left-axis deviation as a major change, since a previous metanalysis showed it as a risk factor for heart disease. 8 The prevalence of altered ECG among PwH without previous ASCVD event varied from 25% to 30%. This is similar to the aforementioned prevalence of abnormal ECG traces up to 40%, when evaluated by the Minnesota Code, among men without haemophilia aged 35 years or more. 8
Notwithstanding, the clinical value of this finding is still unknown because ECG abnormalities can have many causes, only some of which are attributable to ASCVD. 23 A recent systematic review summarized the evidence on screening asymptomatic adults of the general non-haemophilia population for ASCVD risk using ECG. 28 Based on cohort studies, the authors considered that the addition of resting ECG to traditional risk factors could help to reclassify the ASCVD risk mainly in intermediate-risk people (e.g., using the Framingham Risk Score). 7, 10, 28 In such cases, intermediate-risk people with an altered resting ECG could be reclassified into high-risk for ASCVD, for example. However, the USPSTF recommended against screening with resting ECG to prevent ASCVD events in asymptomatic adults at low risk of ASCVD events. Moreover, they concluded that current evidence was insufficient to assess the balance of benefits and harms of screening with resting ECG to prevent ASCVD events in asymptomatic adults at intermediate- or high-risk of ASCVD events. 9
Besides that, there is substantial and consistent evidence that identifying and treating traditional, modifiable ASCVD risk factors such as obesity, SAH, DM, and dyslipidaemia improve cardiovascular outcomes. 10 These risk factors are linked to the pathophysiology of ASCVD. 10 In this analysis of the HemoCardio Study, the prevalence of DM (14%) and obesity (16%) is considerable, but the prevalence of SAH (56%) and dyslipidaemia (72%) is even more impressive, especially after considering that we have evaluated relatively young PwH aged ≥ 30 years. Other authors published comparable results, showing that the prevalence of these ASCVD risk factors among PwH is non-negligible, and may be higher than the prevalence among non-haemophilia men. 4- 7 Interestingly, having severe haemophilia was associated with an altered ECG both according to the international reference values and the binary Minnesota Code, in our study. Until a few decades ago, one should consider intuitively that having severe haemophilia would be protective against ASCVD, 29 but recent data from the United States showed that PwH had a higher risk of ASCVD and the events might present earlier than in their non-haemophilia counterparts. 30 As the American report did not discriminate PwH among severities, 30 we could not infer our results based on theirs. In addition, since the cardiovascular risk profiles were similar between asymptomatic PwH with normal and altered ECGs, we could not find a metabolic reason for our finding.
There are formal recommendations to evaluate and treat these diseases in non-haemophilia people 15- 19 but no formal study has assessed the impact of treating these diseases in the haemophilia population. Although there is no evidence of the impact of such changes on outcomes and some medical institutions recommend against screening ECG, it does not abrogate us from our responsibilities to proactively manage ASCVD risk. Therefore, we suggest the same recommendations used among non-haemophilia individuals should be used to evaluate and treat ASCVD risk factors among PwH. 7
This study has several limitations. Firstly, the small number of PwH and the unicentric design of the study preclude the generalizability of the results. Besides, we included 68% (82/120) of PwH aged ≥ 30 years. Secondly, the study was not designed to evaluate ECG changes in a non-haemophilia contemporaneous asymptomatic population. This may limit the comparison between the populations, although our results clearly showed that the prevalence of ECG changes among asymptomatic PwH is not negligible. Thirdly, we could not describe the clinical impact of ECG changes because the study was not designed to follow patients. Finally, there were some missing values related to glycaemic and lipid profile results. Future research is under development and will involve more asymptomatic PwH and asymptomatic age-matched individuals without haemophilia to further elucidate the accuracy of our results and compare them with a non-haemophilia population.
Conclusion
We described a prevalence of altered resting ECG in 25% and 30% of asymptomatic PwH male adults aged ≥ 30 years, according to the international reference values and the ELSA-Brasil reported values, respectively. We also observed a remarkably high prevalence of SAH (56%) and dyslipidaemia (72%) among this population. Our group recommends evaluating and treating these patients according to guidelines formally directed to the general population. Finally, we reinforce that using ECG as a screening tool for ASCVD in asymptomatic PwH should also follow the guidelines for the general population.
References
Srivastava A, Santagostino E, Dougall A, Kitchen S, Sutherland M, Pipe SW, et al. WFH Guidelines for the Management of Hemophilia, 3rd edition. Haemophilia. 2020;26(Suppl 6):1-158. doi: 10.1111/hae.14046.
Lövdahl S, Henriksson KM, Baghaei F, Holmström M, Nilsson JÅ, Berntorp E, et al. Incidence, Mortality Rates and Causes of Deaths in Haemophilia Patients in Sweden. Haemophilia. 2013;19(3):362-9. doi: 10.1111/hae.12092.
Jardim LL, van der Bom JG, Caram-Deelder C, Gouw SC, Leal Cherchiglia M, Meireles Rezende S. Mortality of Patients with Haemophilia in Brazil: First Report. Haemophilia. 2019;25(3):e146-e152. doi: 10.1111/hae.13730.
van de Putte DEF, Fischer K, Makris M, Tait RC, Chowdary P, Collins PW, et al. Unfavourable Cardiovascular Disease Risk Profiles in a Cohort of Dutch and British Haemophilia Patients. Thromb Haemost. 2013;109(1):16-23. doi: 10.1160/TH12-05-0332.
Minuk L, Jackson S, Iorio A, Poon MC, Dilworth E, Brose K, et al. Cardiovascular Disease (CVD) in Canadians with Haemophilia: Age-Related CVD in Haemophilia Epidemiological Research (ARCHER study). Haemophilia. 2015;21(6):736-41. doi: 10.1111/hae.12768.
Lövdahl S, Henriksson KM, Baghaei F, Holmström M, Berntorp E, Astermark J. Hypertension and Cardiovascular Diseases in Swedish Persons with Haemophilia - A Longitudinal Registry Study. Thromb Res. 2019;181:106-11. doi: 10.1016/j.thromres.2019.07.017.
Camelo RM, Caram-Deelder C, Duarte BP, de Moura MCB, Costa NCM, Costa IM, et al. Cardiovascular Risk Factors Among Adult Patients with Haemophilia. Int J Hematol. 2021;113(6):884-92. doi: 10.1007/s12185-021-03104-y.
Chou R, Arora B, Dana T, Fu R, Walker M, Humphrey L. Screening Asymptomatic Adults with Resting or Exercise Electrocardiography: A Review of the Evidence for the U.S. Preventive Services Task Force. Ann Intern Med. 2011;155(6):375-85. doi: 10.7326/0003-4819-155-6-201109200-00006.
US Preventive Services Task Force; Curry SJ, Krist AH, Owens DK, Barry MJ, Caughey AB, et al. Screening for Cardiovascular Disease Risk with Electrocardiography: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(22):2308-14. doi: 10.1001/jama.2018.6848.
Alagona P Jr, Ahmad TA. Cardiovascular Disease Risk Assessment and Prevention: Current Guidelines and Limitations. Med Clin North Am. 2015;99(4):711-31. doi: 10.1016/j.mcna.2015.02.003.
Zong Y, Maanja M, Chaireti R, Schlegel TT, Ugander M, Antovic JP. Substantial Prevalence of Subclinical Cardiovascular Diseases in Patients with Hemophilia A Evaluated by Advanced Electrocardiography. J Electrocardiol. 2020;58:171-5. doi: 10.1016/j.jelectrocard.2019.12.008.
Branson BM, Handsfield HH, Lampe MA, Janssen RS, Taylor AW, Lyss SB, et al. Revised Recommendations for HIV Testing of Adults, Adolescents, and Pregnant Women in Health-Care Settings. MMWR Recomm Rep. 2006;55(RR-14):1-17; quiz CE1-4.
Getchell JP, Wroblewski KE, DeMaria A, Bean CL, Parker MM, Pandori M, et al. Testing for HCV infection: An update of guidance for clinicians and laboratorians. MMWR Morb Mortal Wkly Rep. 2013;62(18):362-5. doi: 10.18621/eurj.945588.
Centers for Disease Control. Revised Surveillance Case Definition for HIV Infection. MMWR Morb Mortal Wkly Rep. 2014;63(3):29-31.
Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, Donato KA, et al. 2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Obesity Society. J Am Coll Cardiol. 2014;63(25 Pt B):2985-3023. doi: 10.1016/j.jacc.2013.11.004.
Garvey WT, Mechanick JI, Brett EM, Garber AJ, Hurley DL, Jastreboff AM, et al. American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines for Medical Care of Patients with Obesity. Endocr Pract. 2016;22 Suppl 3:1-203. doi: 10.4158/EP161365.GL.
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Himmelfarb CD, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71(19):e127-e248. doi: 10.1016/j.jacc.2017.11.006.
Chamberlain JJ, Rhinehart AS, Shaefer CF Jr, Neuman A. Diagnosis and Management of Diabetes: Synopsis of the 2016 American Diabetes Association Standards of Medical Care in Diabetes. Ann Intern Med. 2016;164(8):542-52. doi: 10.7326/M15-3016.
Jellinger PS, Handelsman Y, Rosenblit PD, Bloomgarden ZT, Fonseca VA, Garber AJ, et al. American Association of Clinical Endocrinologists and American College of Endocrinology Guidelines for Management of Dyslipidemia and Prevention of Cardiovascular Disease. Endocr Pract. 2017;23(Suppl 2):1-87. doi: 10.4158/EP171764.APPGL.
Kopin L, Lowenstein C. Dyslipidemia. Ann Intern Med. 2017;167(11):ITC81-ITC96. doi: 10.7326/AITC201712050.
Vandenberk B, Vandael E, Robyns T, Vandenberghe J, Garweg C, Foulon V, et al. Which QT Correction Formulae to Use for QT Monitoring? J Am Heart Assoc. 2016;5(6):e003264. doi: 10.1161/JAHA.116.003264.
Pinto MM Filho, Brant LCC, Padilha-da-Silva JL, Foppa M, Lotufo PA, Mill JG, et al. Electrocardiographic Findings in Brazilian Adults without Heart Disease: ELSA-Brasil. Arq Bras Cardiol. 2017;109(5):416-24. doi: 10.5935/abc.20170146.
Punjabi NP, Sahdev N, Punjabi PP. Book Review: Goldberger's Clinical Electrocardiography. 9th ed. A Simplified Approach. 2017;32(8):709. doi: 10.1177/0267659117732938.
Schmidt MI, Duncan BB, Mill JG, Lotufo PA, Chor D, Barreto SM, et al. Cohort Profile: Longitudinal Study of Adult Health (ELSA-Brasil). Int J Epidemiol. 2015;44(1):68-75. doi: 10.1093/ije/dyu027.
Prineas RJ, Crow RS, Zhang ZM. The Minnesota Code Manual of Electrocardiographic Findings Including Measurement and Comparison with the Novacode Standards and Procedures for ECG Measurement in Epidemiologic and Clinical Trials. 2nd ed. Berlim: Springer International Publishing; 2010.
Camelo RM, Duarte BP, do Nascimento AM Jr, Vanderlei AM. Third-Degree Atrioventricular Block and Pacemaker Implantation in a Man with Severe Hemophilia A: A Case Report. J Arrhythm. 2021;37(2):460-1. doi: 10.1002/joa3.12513.
Schlegel TT, Kulecz WB, Feiveson AH, Greco EC, DePalma JL, Starc V, et al. Accuracy of Advanced versus Strictly Conventional 12-Lead ECG for Detection and Screening of Coronary Artery Disease, Left Ventricular Hypertrophy and Left Ventricular Systolic Dysfunction. BMC Cardiovasc Disord. 2010;10:28. doi: 10.1186/1471-2261-10-28.
Jonas DE, Reddy S, Middleton JC, Barclay C, Green J, Baker C, et al. Screening for Cardiovascular Disease Risk with Resting or Exercise Electrocardiography: Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2018;319(22):2315-28. doi: 10.1001/jama.2018.6897.
Darby SC, Sau WK, Spooner RJ, Giangrande PLF, Hill FGH, Hay CRM, et al. Mortality Rates, Life Expectancy, and Causes of Death in People with Hemophilia A or B in the United Kingdom who were not Infected with HIV. Blood. 2007;110(3):815-25. doi: 10.1182/blood-2006-10-050435.
Pocoski J, Ma A, Kessler CM, Boklage S, Humphries TJ. Cardiovascular Comorbidities are Increased in U.S. Patients with Haemophilia A: A Retrospective Database Analysis. Haemophilia. 2014;20(4):472-8. doi: 10.1111/hae.12339.
*Supplemental Materials
For additional information, please click here.
Notes
HemoCardio Study received no funding nor grant to be developed/implemented. RMC received scholarship (PDSE-88881.362041/2019-1) from the Coordination for the Improvement of Higher Education Personnel (CAPES) Foundation, an agency under the Ministry of Education of Brazil, in order to conduct part of his doctoral research as a visiting student at Leiden Universitair Medisch Centrum in the Netherlands.
This study is not associated with any thesis or dissertation work.
This study was approved by the Ethics Committee of the Fundação de Hematologia e Hemoterapia do Estado de Pernambuco under the protocol number 2.667.012 (CAAE 86067818.6.0000.5795. All the procedures in this study were in accordance with the 1975 Helsinki Declaration, updated in 2013. Informed consent was obtained from all participants included in the study.
Author notes
Mailing Address: Ricardo Mesquita Camelo Federal University of Minas Gerais, Medicine School. Rua Lorena, 1020, 101. Postal code: 30730-170. Padre Eustáquio, Belo Horizonte, MG – Brazil E-mail: rmcamelo@hotmail.com
Conflict of interest declaration
RMC received honoraria for participating as a speaker at scientific and educational meetings and travel support for scientific meetings by Novo Nordisk, Bayer, Takeda, and Hoffman-La Roche. NCMC received honoraria for participating as a speaker at scientific and educational meetings from Takeda and travel support for scientific meetings from Takeda and Novo Nordisk. IMC received honoraria for participating as a speaker at scientific and educational meetings from Takeda and travel support for scientific meetings from Takeda and Hoffman-La Roche. AMV received honoraria for participating as a speaker at scientific and educational meetings from Takeda and travel support for scientific meetings from Novo Nordisk, Takeda, Hoffman-La Roche, and BioMarin. TMRG received travel support for scientific meetings from Takeda. SG received an unrestricted research grant from Sobi. JvdB received reimbursement for educational activities from Bayer and unrestricted research funding from Novo Nordisk. CGPR, CCD, BPD, MCBM, and SMR declare they have no interests that might be perceived as posing a conflict of bias.
