ORIGINAL ARTICLE
Use of potentially inappropriate medications and medications of concern in older adults with multiple myeloma
Utilização de medicamentos potencialmente inapropriados e medicamentos que requerem cautela em idosos com mieloma múltiplo
Use of potentially inappropriate medications and medications of concern in older adults with multiple myeloma
Geriatrics, Gerontology and Aging, vol. 18, e0000044, 2024
Sociedade Brasileira de Geriatria e Gerontologia, SBGG
Received: 06 July 2023
Accepted: 11 August 2023
Abstract
Objectives: To analyze the use of potentially inappropriate medications (PIMs) and medications used in supportive therapy that require caution in older adults with cancer, in addition to determining associated factors the agreement between criteria sets used to identify PIMs.
Methods: This cross-sectional study included individuals with multiple myeloma aged ≥ 60 years who were undergoing outpatient treatment. PIMs were identified according to American Geriatric Society Beers 2019, PRISCUS 2.0, and Brazilian Consensus on Potentially Inappropriate Medicines criteria. Medications of concern were defined according to National Comprehensive Cancer Network criteria. Factors associated with the use of PIMs and medications of concern were identified using multiple logistic regression. The degree of agreement between the 3 criteria sets was measured using Cohen’s kappa coefficient.
Results: The frequency of PIM use was 52.29% according to American Geriatric Society Beers criteria, 62.74% according to Brazilian Consensus criteria, and 65.36% according to PRISCUS criteria, while 52.29% of the patients were using medications of concern. Agreement between American Geriatric Society Beers, PRISCUS, and Brazilian Consensus criteria was high, while it was excellent between Brazilian Consensus and PRISCUS criteria. In the final logistic regression model, polypharmacy was associated with PIM use according to each criteria set, as well as the use of medications of concern.
Conclusions: The frequency of PIMs and medications of concern was high. Agreement about PIM use between the American Geriatric Society Beers, Brazilian Consensus, and PRISCUS criteria was high or excellent. There was an independent association between polypharmacy and the use of PIMs and medications of concern by older patients with multiple myeloma.
Keywords: List of potentially inappropriate medications+ older adults+ multiple myeloma+ cancer.
Resumo
Objetivo: Analisar o uso de medicamentos potencialmente inapropriados (MPIs) e o uso de medicamentos usados em terapia de suporte que requerem cautela em idosos com câncer (MTSRCICs), determinando os fatores associados. Visou-se também determinar a concordância entre os critérios explícitos empregados na identificação de MPI.
Metodologia: Estudo transversal com indivíduos com mieloma múltiplo (MM), idade ≥ 60 anos em tratamento ambulatorial. Os MPI foram identificados de acordo com os critérios AGS Beers 2019, PRISCUS 2.0 e o Consenso Brasileiro de Medicamentos Potencialmente Inapropriados (CBMPI). Os MTSRCIC foram definidos de acordo com a National Comprehensive Cancer Network. Os fatores associados ao uso de MPI e MTSRCIC foram identificados por regressão logística múltipla. O grau de concordância entre os três critérios explícitos empregados no estudo foi mensurado pelo coeficiente kappa Cohen.
Resultados: As frequências de MPI foram 52,29% (AGS Beers 2019), 62,74% (CBMPI), 65,36% (PRISCUS 2.0) e 52,29% (MTSRCICs). As concordâncias entre AGS Beers 2019 com PRISCUS 2,0 e com CBMPI foram altas, enquanto a concordância entre CBMPI e PRISCUS 2.0 foi excelente. No modelo final de regressão logística polifarmácia foi associada positivamente ao uso de MPI por idosos para os três critérios explícitos utilizados, além de associado à utilização de MTSRCICs.
Conclusões: A frequência do uso de MPI e de MTSRCIC foi elevada. A concordância em relação ao uso de MPI entre os critérios AGS Beers 2019, CBMPI e PRISCUS 2.0 foi alta ou excelente. A polifarmácia apresentou associação independente e positiva com uso de MPIs e de MTSRCICs por pacientes idosos com MM.
Palavras-Chaves: lista de medicamentos potencialmente inapropriados, idoso, mieloma múltiplo, câncer.
INTRODUCTION
Multiple myeloma (MM), a malignant neoplasm of plasma cells and activated B lymphocytes, is characterized by the uncontrolled proliferation of these cells and their accumulation in bone marrow.1 The pathogenesis of MM is complex and highly heterogeneous. The progression of the disease has several stages, in which chromosomal translocations, aneuploidy, genetic mutations, and epigenetic aberrations are essential determinants. The main complications of this disease are determined according to bone marrow involvement and include bone destruction, hyperkalemia, renal dysfunction, immunological paralysis, leukopenia, thrombocytopenia, and venous thromboembolism.1, 2, 3
MM represents 1.00%–1.80% of all cancers and has the second highest incidence among hematological malignancies, in which only 10.00%–15.00% of patients meet or exceed expected survival compared to the corresponding general population.2, 3 According to data from the Brazil Oncology Panel, around 2800 cases of MM were diagnosed annually in both sexes between 2013 and 2019 more than 60.00% of which were in people > 60 years of age.4
Older people with cancer have an elevated risk of adverse events because multiple medications are often added to their pharmacotherapy plan to compensate for symptoms caused by cancer or the adverse effects of antineoplastic therapy. Adequate drug prescription regarding both selection and dosage schedule is thus of paramount importance among older patients, requiring a difficult balance between efficacy, safety, and tolerability.5
In this context, potentially inappropriate medications (PIMs) are those that older adults should avoid in most circumstances or in specific situations because (1) the risk-benefit ratio is unfavorable and (2) there are safe and more effective pharmacological and non-pharmacological options.6 PIMs are associated with preventable adverse drug events, such as constipation, falls, immobility, mental confusion, and hip fractures, in addition to an increased risk of hospitalization and mortality.7, 8
Attempts to detect inappropriate prescriptions have intensified in recent decades, including the development and validation of a series of criteria sets for PIMs, such as the American Geriatric Society (AGS) Beers, PRISCUS 2.0, and the Brazilian Consensus of Potentially Inappropriate Medications for Older Adults (BCPIM), which are important for clinical practice and should be included in the pharmacotherapeutic assessment of every older patient.6, 9
Aiming to promote the safe use of medications by cancer patients, the U.S. National Comprehensive Cancer Network (NCCN) has developed specific pharmacotherapy guidelines, as well as a list of medications commonly used for supportive care that are of concern (MCSCC) in older adults.10
The main objective of this study was to analyze the use of PIM and MCSCC among in older people with MM and determine associated factors. This study also aimed to determine the agreement between the criteria used to identify PIM.
METHODS
This cross-sectional study was conducted in 3 health services in a metropolitan region in southeastern Brazil:
- 1. An oncology unit in a public teaching hospital
- 2. A public oncology reference hospital
- 3. A private onco-hematology clinic.
The study is linked to a research project called “Adverse events, treatment adherence and quality of life in patients with MM”. Strengthening the Reporting of Observational Studies in Epidemiology guidelines11 were used to develop this study. The study population consisted of MM outpatients aged ≥ 60 years (according to the United Nations’ definition of older adults for developing countries) at the participating health care services.
The sample was recruited from patient care flow between April 2019 and March 2020. Older patients diagnosed with MM who were using ≥ 1 medications were included. Patients diagnosed with MM who did not complete the interview, refused to provide written consent, or were not taking any medications were excluded.
Data on the study variables was collected using a form prepared by the research team that consisted of the above-mentioned sociodemographic, clinical, and pharmacotherapeutic variables. The data were recorded in Questionnaire Development System 2.6.1.
Patients were recruited through scheduling information from the hematology services at participating institutions. On a single occasion, they were interviewed and the data collection instrument was applied. The data collected in the interview were confirmed through information on continuous-use medications in their medical records, establishing a temporal relationship with the interview. To avoid memory bias and due to the impossibility of verifying patient-reported information, only formally prescribed medications were considered, i.e., data on self-medication were disregarded.
The study’s dependent variables were medication related:
- 1. The use of at least 1 PIM. The criteria sets used to classify PIM were: AGS Beers 2019,6 BCPIM,8 and Potentially Inadequate Medications in the Elderly (PRISCUS 2.0).12 Only medications to be avoided regardless of the clinical condition or those with maximum dosage restrictions were considered PIMs.
- 2. The use of at least one medication of concern identified in the NCCN’s list of MCSCC in Older Patients.10
The independent variables were classified as sociodemographic, clinical, or pharmacotherapeutic:
-
Sociodemographic: age, sex, income (≤ or > 3 times the federal minimum salary), education (lower: none to incomplete high school; higher: completed high school or higher) and care type (public [Unified Health System] or private).
-
Clinical: history of hospitalization (yes or no), multimorbidity (≥ 2 diseases), myeloma stage according to the International Staging System, systemic arterial hypertension (yes or no), diabetes mellitus (yes or no ), chronic kidney disease (yes or no), and solid cancer (yes or no).
-
Pharmacotherapeutic: polypharmacy (yes or no), which was defined as the use of ≥ 5 medications, not including antineoplastics used to treat MM, in addition to reported adverse events (yes or no).
Data recorded in Questionnaire Development System were exported to IBM SPSS Statistics 25.0 for analysis. The results were described using frequency, proportions, and dispersion measures as applicable. Numerical variables were dichotomized with the median. Normality of distribution was determined using the Kolmogorov-Smirnov test.
For each set of PIM criteria and medications of concern criteria, the association between use and the independent variables was determined using Pearson’s χ2 test. Variables with p ≤ 0.20 in univariate analysis were included in multivariate analysis (logistic regression modeling). Those with p ≤ 0.05 at this stage were maintained in the final model. A logistic model was constructed for each set of criteria.
The degree of agreement between the 3 PIM criteria sets was measured using the Cohen kappa coefficient, being classified as follows: > 0.81 = excellent, 0.61-0.80 = high, 0.41-0.60 = moderate, and ≤ 0.40 = low.13
The present study was approved by the institutional research ethics committee, via Plataforma Brasil and was authorized by the involved health services. The study was conducted in accordance with Resolution 466/12, which regulates research involving human beings. All participants provided written informed consent prior to inclusion.
RESULTS
The characteristics of the 153 individuals who participated in the study are described in Table 1. The median age was 71 (IQR = 13, min. = 60, max. = 92) years and the majority were women (54.25%). The majority had an income > 3 times the federal minimum salary (53.60%), a high-school education level or higher (52.29%) and used private health care providers (56.86%).
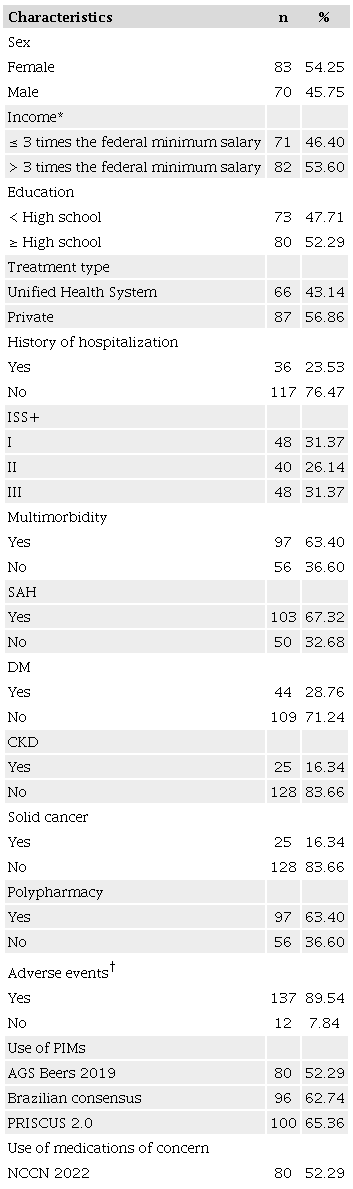
AGS: American Geriatric Society; Brazilian consensus: Brazilian Consensus on Potentially Inappropriate Medicines; DM: diabetes mellitus; CKD: chronic kidney disease; ISS: International Staging System; NCCN: National Comprehensive Cancer Network; PIMs: potentially inappropriate medications; SAH: systemic arterial hypertension. *Federal minimum salary = USD 275.00 according to exchange rates in January 2019; †Total varies according to the ignored information.
Regarding the patients’ clinical characteristics, 63.40% had multimorbidity, 67.32% were hypertensive, 28.76% were diabetic, 16.34% had chronic kidney disease, and 16.34% had a solid tumor. The median number of medications was 7 (IQR = 5, min. = 1, max. = 19), polypharmacy was identified in 63.40%, and 89.54% reported an adverse event due to their medication.
At least 1 PIM was used by 52.29% of the patients according to AGS Beers criteria, 62.74% of the patients according to BCPIM criteria, and 65.36%, according to PRISCUS 2.0 criteria. According to the NCCN criteria, 52.34% of the patients used at least 1 medication of concern.
Agreement between the AGS Beers and BCPIM criteria (kappa = 0.762) and between the AGS Beers and PRISCUS 2.0 criteria (kappa = 0.708) was high, while agreement between the BCPIM and PRISCUS 2.0 criteria (kappa = 0.830) was excellent. All 3 cases were statistically significant (p < 0.001).
According to the 3 criteria sets, the most commonly used PIMs were proton pump inhibitors, opioids, and benzodi-azepines, which together represented more than half of all PIMs. Other commonly identified PIMs were antidepressants, Zolpidem, and antipsychotics (Table 2). The most frequent medications of concern were corticosteroids, opioids, and selective serotonin reuptake inhibitors, which represented > 70% of all medications listed by the NCCN. As in the PIM criteria, benzodiazepines, zolpidem, and antipsychotics were common (Table 3).
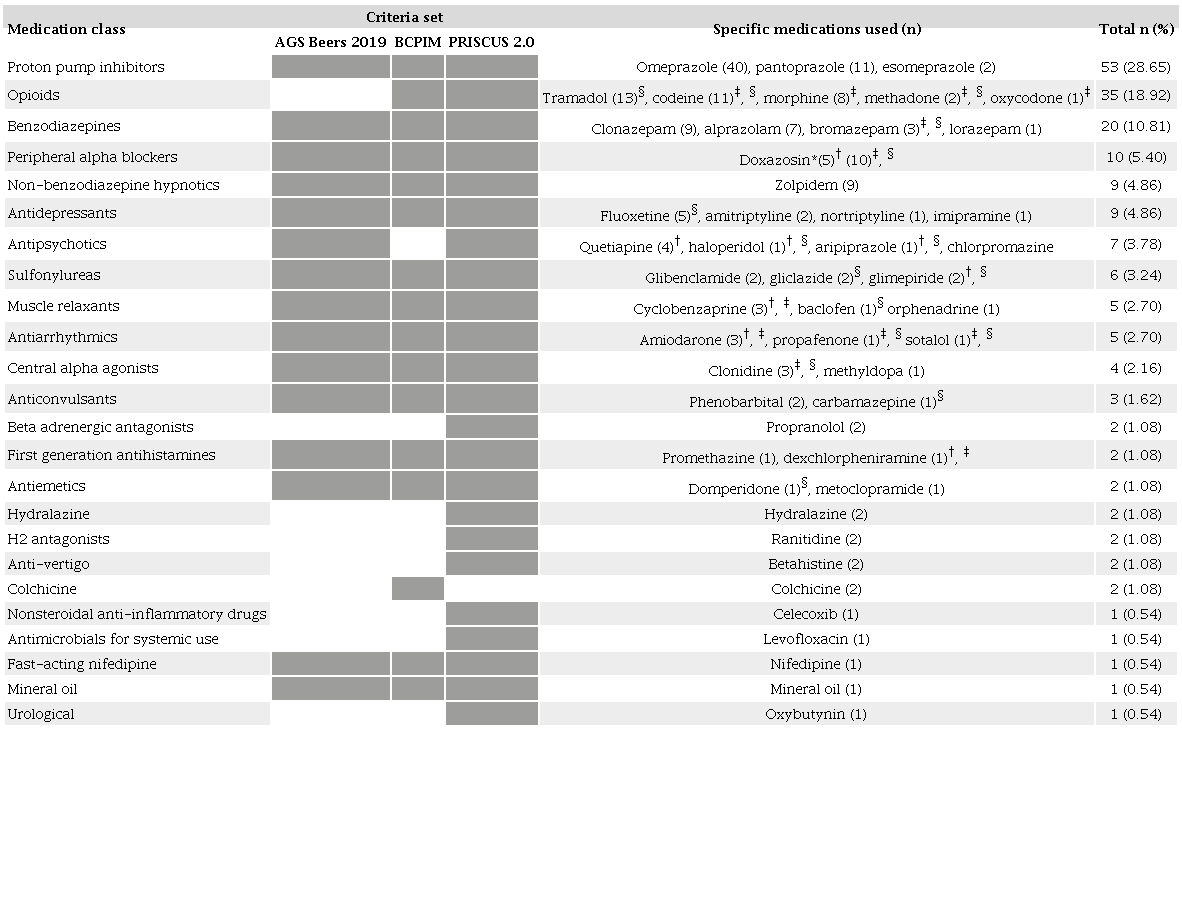
AGS: American Geriatrics Society; BCPIM: Brazilian Consensus on Potentially Inappropriate Medicines. *According to AGS Beers criteria, doxazosin is only a PIM if used as an antihypertensive. According to BCPIM and PRISCUS 2.0 criteria, doxazosin is a PIM regardless of the indication; †Drugs considered PIMs according to AGS Beers 2019 criteria; ‡Drugs considered PIMs according to BCPIM criteria; §Drugs considered PIMs according to PRISCUS 2.0 criteria. The dark gray represents lists in which the PIM criterion is used.
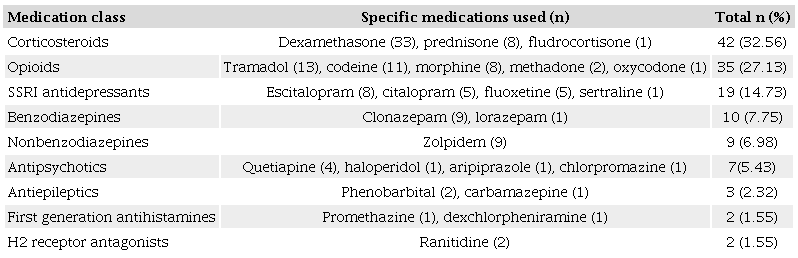
SSRI: Selective serotonin re-uptake inhibitor.
In univariate analysis, there was an association (p < 0.20) between PIM use and the independent variables (Table 4) for all 3 criteria sets, multimorbidity, SAH, and polypharmacy. A higher education level, income > 3 times the minimum salary, and use of private health care were only associated with PIM use according to AGS Beers criteria. A diagnosis of diabetes mellitus was only associated with PIM use according to PRISCUS 2.0 criteria.
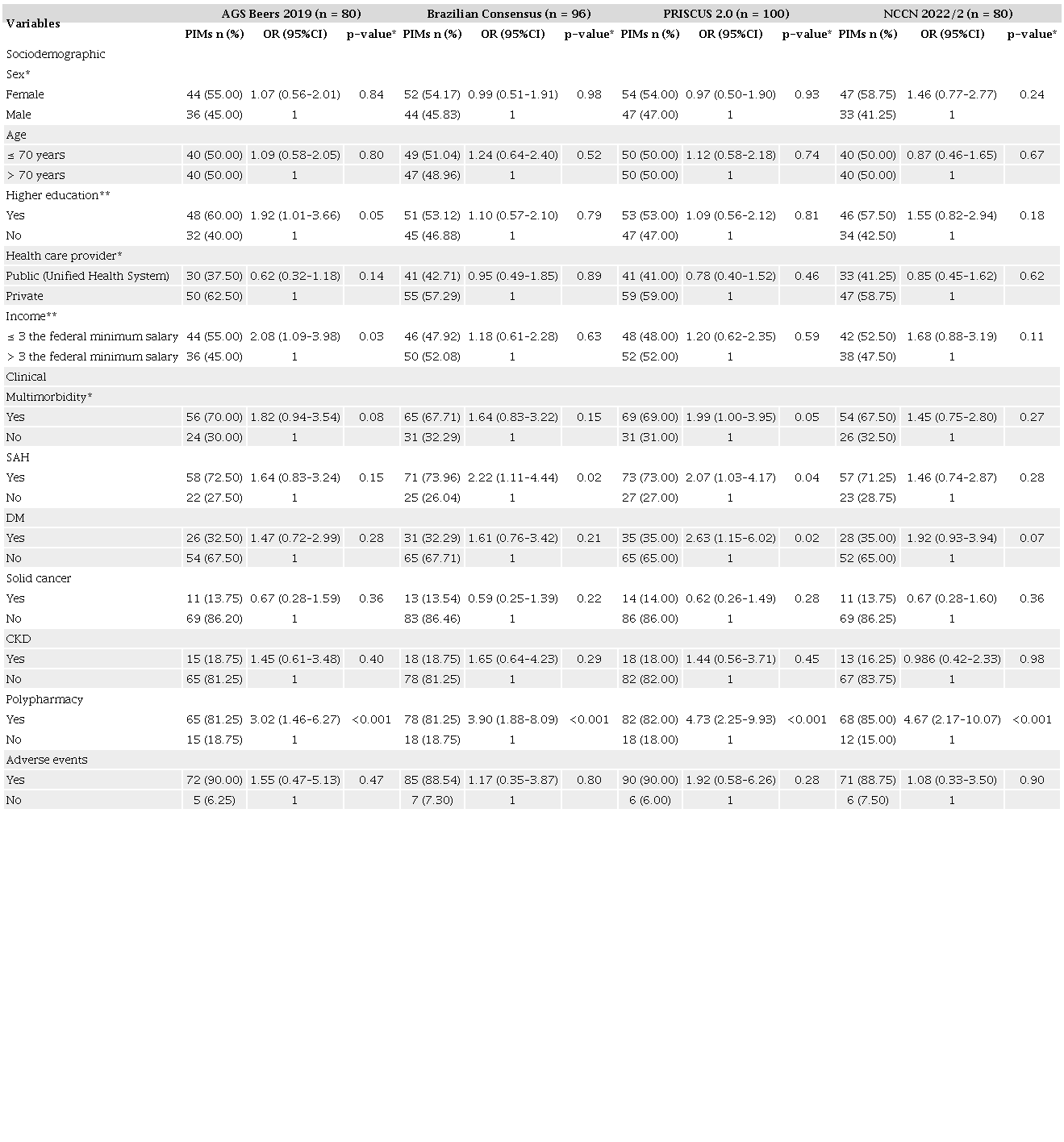
AGS: American Geriatric Society; CKD: chronic kidney disease; DM: diabetes mellìtus; NCCN: Medications of concern according to the National Comprehensive Cancer Network; OR: odds ratio; PIMs: potentially inappropriate medications; SAH: systemic arterial hypertension. *Pearson’s χ2 test.
Analysis of the association between medications of concern and the independent variables (shown in the final column of Table 4) indicated associations with a high education level, income > 3 times the minimum salary, diabetes mellitus, and polypharmacy.
In the final logistic regression model (Table 5), polypharmacy was associated with PIM use (according to all 3 criteria sets) and medications of concern. A high education level was also associated with PIM use according to AGS Beers criteria.

AGS: American Geriatric Society; BCPIM: Brazilian Consensus on Potentially Inappropriate Medications; NCCN: National Comprehensive Cancer Network; OR: odds ratio.
DISCUSSION
Although each criteria set was developed for a specific population, they are commonly adapted in other countries. Brazil has its own criteria, the BCPIM, which were developed for this country’s health system and the social and clinical characteristics of its patients. Although the Brazilian criteria were published in 2016, their high agreement with criteria published in 2019 and 2023 indicate that are still useful for decision-making about the safest pharmacotherapy for older patients. Opioid analgesics were the second most frequent PIM among patients in this study, which probably explains the greater agreement between BCPIM and PRISCUS 2.0, given that this class is included in both of these criteria sets but not in AGS Beers.
The 3 PIM criteria sets agreed about the risk of certain medication classes, including proton pump inhibitors, benzodiazepines, nonbenzodiazepines, tricyclic antidepressants, musculoskeletal relaxants, and first-generation antihistamines. However, other classes are only considered inappropriate in 1 or 2 criteria sets, as shown in Table 2. PRISCUS 2.0, the most recent criteria set, includes new drugs and/or classes not included in the others.
In addition to listing tricyclic antidepressants as a potential risk for older patients, PRISCUS 2.0 also classifies some selective serotonin reuptake inhibitors, such as fluoxetine and paroxetine, as PIMs. Other medications are only found in PRISCUS 2.0, such as fluoroquinolone-class antibacterials, cyclooxygenase-2 inhibitors, and anti-vertigo drugs. Gliclazide, a short-acting sulfonylurea, was classified as a PIM in PRISCUS 2.0, although it is a therapeutic alternative to long-acting sulfonylureas, which are considered inappropriate according to previous criteria sets.
Like PRISCUS 2.0, the most recent version of the AGS Beers criteria, published in May 2023, classified sulfonylurea as a potentially inappropriate short-acting drug. The panel of experts justified this decision because sulfonylureas are associated with a higher risk of cardiovascular events, mortality, and hypoglycemia. However, if sulfonylurea is required, short-acting substitutes, such as gliclazide or glipizide, are recommended. This decision recognizes that access to antidiabetic drugs from other classes may not be available in certain health care systems, in which short-acting sulfonylurea is a viable option. Importantly, AGS Beers emphasizes that PIMs cannot be absolutely contraindicated in practice; the decision must be based on the patient’s clinical context.14
The NCCN criteria for medications of concern are similar to those for PIMs in a number of aspects, since they also include opioid analgesics, benzodiazepines, nonbenzodiazepines, antipsychotics, antiepileptics, and first-generation antihistamines. However, the most frequently used medications of concern were corticosteroids, which are not included in the PIM criteria. This was expected, given that this class is a first-choice treatment for MM. Another major difference is that the NCCN designated the entire class of selective serotonin reuptake inhibitors as medications of concern, rather than certain representatives of that class.
Other studies using the 2019 AGS Beers criteria in older cancer patients have obtained similar results to the present study, i.e., > 50% frequency of ≥ 1 PIM.5, 15 Although Brazilian studies using the BCPIM criteria in older cancer patients are scarce, our results are similar to those of the general population.16, 17 To the best of our knowledge, no studies have used the PRISCUS 2.0 criteria to assess PIM use in older patients with MM, other forms of neoplasm, or the general older adult population.
The most frequent (28.65%) PIMs were proton pump inhibitors. Proton pump inhibitors have been classified as a PIM for older adults due to their association with bone loss and increased risk of fractures.6 The association between bone loss and lytic lesions caused by the pathophysiology of MM increases the need for caution regarding this class of medications. Furthermore, proton pump inhibitors are associated with a risk of several adverse effects, including Clostridioides difficile infection, community-acquired pneumonia, vitamin B12 deficiency, kidney disease, and dementia.18
Another widely used class of PIMs was opioid analgesics (18.92%). These are the basis of pain treatment for cancer patients, and although all opioids are considered effective for treating cancer pain, no specific well-designed studies have been conducted about their effects in older patients. The most common adverse effects of opioid analgesics include constipation, dry mouth, nausea, vomiting, drowsiness, confusion, respiratory depression, and withdrawal symptoms.19 In addition to adverse drug reactions, the physical dependence they cause is associated with a high risk of death.20
Another important observation was in relation to benzodiazepines (10.81%). Benzodiazepines are commonly used for anxiety and insomnia, although psychotherapy and alternative medications are preferred.21 Benzodiazepines have been associated with several adverse effects, including ataxia, dizziness, excessive sedation, anterograde amnesia, and dependence, and older patients may have more severe of adverse effects.22 Due to their slower reaction time and metabolism, older patients are at greater risk of falls and fractures, cognitive problems, and delirium from benzodiazepines.10 Nonbenzodiazepines, an alternative treatment for insomnia, were also identified in this study (4.86%). However, in older adults they cause adverse events similar to those of benzodiazepines, including delirium, falls, and fractures, in addition to that fact that they have a minimal effect on sleep patterns.6, 10
A high education level (≥ high school graduate) was positively associated with PIM use according to AGS Beers criteria. However, an association between high education level and PIM use in older cancer patients has not yet been reported in the literature. The results of other Brazilian population-based studies have shown that older adults with a high education level have better to access health care, which could lead to more prescribed medications.23, 24 This socioeconomic difference might explain the association we found between higher education level and PIM.
Polypharmacy has been associated with PIM use in all 3 criteria sets for the general older adult population and in specific criteria for cancer patients. Polypharmacy is an important public health problem, since it has been associated with several negative outcomes, such as worsening adherence to pharmacotherapy, more complex therapeutic regimens and increased health system costs, as well as a higher risk of adverse events, falls, fractures, frailty, hospitalization, and even mortality.7, 9, 15, 25, 26, 27, 28, 29
Patients with polypharmacy were 3-4 times more likely to use a PIM. In this study, polypharmacy was identified in 63.40% of the patients, which is extremely close to the results of Umut et al. (63.80%) in a study on the association between polypharmacy, PIMs, and the risk of falls and neuropathy in older patients with MM.27 Although studies assessing PIM use in older patients with MM are scarce, our results were similar to (albeit somewhat lower than) previous studies on polypharmacy among older patients with hematological malignancies: 76.8% in a study at the Dana-Farber Cancer Institute (Boston, MA, USA) and 75.00% in a study on older French adults with hematological malignancies.28, 29
Since polypharmacy and PIM use by older cancer patients make pharmacotherapy more complex and less safe, strategies are needed to optimize medication use in this population.9 The positive association between polypharmacy and fall risk among older people with MM can reduce quality of life and functionality.25 Deprescribing PIMs and other medications that are not necessary or safe for older adults has been an effective intervention.30
This study contributes to the promotion of appropriate drug use among older people with cancer. As far as we know, it is the first study to describe and compare the frequency of PIM use by older MM patients according to AGS Beers 2019, BCPIM, and PRISCUS 2.0 criteria and to assess agreement between these 3 criteria sets. It is also the first Brazilian study on PIMs to perform an additional analysis involving medications of concern in older cancer patients, since medications are commonly added to the pharmacotherapy regimen of cancer patients based on the appearance of symptoms caused either by the disease or the antineoplastic therapy.
However, certain study limitations must be pointed out. First, some PIM criteria were disregarded in this study, since they involve increased risk for older adults only for specific clinical conditions or due to dosage limits. Thus, this data was not collected. Second, since only prescribed medications were evaluated, PIM use may have been underestimated. Third, this observational study only included patients from a single city, which impedes extrapolation of the results to broader populations. Fourth, the 2023 update of the AGS Beers criteria was published after the study was developed, although it would probably not have resulted in significant changes in PIM frequency, since the changes do not involve drugs frequently used by patients with MM. Finally, although memory bias regarding medication use was likely, it was mitigated by cross-checking with medical records.
CONCLUSIONS
A high frequency of PIMs and medications of concern was used in this sample of older adults with MM. The AGS Beers, BCPIM, and PRISCUS 2.0 criteria had high agreement, while the BCPIM and PRISCUS 2.0 criteria had excellent agreement. There was an independent and positive association between polypharmacy and the use of PIMs and medications of concern.
REFERENCES
Bolli N, Martinelli G, Cerchione C. The molecular pathogenesis of multiple myeloma. Hematol Rep. 2020;12(3):9054. https://doi.org/10.4081/hr.2020.9054
Drummond PLM, Santos RMM, Reis AMM, Malta JS, Silveira LP, Costa IHF, et al. Real-world effectiveness and safety of multiple myeloma treatments based on thalidomide and bortezomib: a retrospective cohort study from 2009 to 2020 in a Brazilian metropolis. Cancer Epidemiol. 2023;85: 102377. https://doi.org/10.1016/j.canep.2023.102377
Dimopoulos MA, Moreau P, Terpos E, Mateos MV., Zweegman S, Cook G, et al. Multiple myeloma: EHA-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2021;32(3):309-22. https://doi.org/10.1016/j.annonc.2020.11.014
Brasil. Ministério da Saúde. DATASUS. Painel Oncologia – Brasil. Available from: http://tabnet.datasus.gov.br/cgi/dhdat.exePPAINEL_ONCO/PAINEL_ONCOLOGIABR.def. Accessed May 11, 2023.
Hshieh TT, DuMontier C, Jaung T, Bahl NE, Hawley CE, Mozessohn L, et al. Association of polypharmacy and potentially inappropriate medications with frailty among older adults with blood cancers. J Natl Compr Canc Netw. 2022;20(8):915-23.e5. https://doi.org/10.6004/jnccn.2022.703
By the 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 Updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674-94. https://doi.org/10.1111/jgs.15767
Almeida TA, Reis EA, Pinto IVL, Ceccato MGB, Silveira MR, Lima MG, et al. Factors associated with the use of potentially inappropriate medications by older adults in primary health care: an analysis comparing AGS Beers, EU(7)-PIM List, and Brazilian Consensus PIM criteria. Res Social Adm Pharm. 2019;15(4):370-7. https://doi.org/10.1016/j.sapharm.2018.06.002
Oliveira MG, Amorim WW, Oliveira CRB, Coqueiro HL, Gusmão LC, Passos LC. Consenso brasileiro de medicamentos potencialmente inapropriados para idosos. Geriatr Gerontol Aging. 2017;10(4):168-81. https://doi.org/10.5327/Z2447-211520161600054
Sharma M, Loh KP, Nightingale G, Mohile SG, Holmes HM. Polypharmacy and potentially inappropriate medication use in geriatric oncology. J Geriatr Oncol. 2016;7(5):346-53. https://doi.org/10.1016/jjgo.2016.07.010
Walter LC, Chair V, Beechinor R, Browner IS, Clifton K, Jay Cohen H, et al. NCCN Guidelines Version 2.2022 Older Adult Oncology NCCN Guidelines Panel Disclosures Continue [Internet]. 2022. Available from: https://www.nccn.org/guidelines/guidelines-detail?category=4&id=1452. Accessed Jan 22, 2024.
Malta M, Cardoso LO, Bastos FI, Magnanini MMF, Silva CMFP. STROBE initiative: guidelines on reporting observational studies. Rev Saude Publica. 2010;44(3):559-65. https://doi.org/10.1590/s0034-89102010000300021
Mann NK, Mathes T, Sönnichsen A, Pieper D, Klager E, Moussa M, et al. Potentially inadequate medications in the elderly: PRISCUS 2.0. Dtsch Arztebl Int. 2023;120(1-2):3-10. https://doi.org/10.3238/arztebl.m2022.0377
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33(1):159-74. PMID: 843571
By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;71(7):2052-81. https://doi.org/10.1111/jgs.18372
Ramsdale E, Mohamed M, Yu V, Otto E, Juba K, Awad H, et al. Polypharmacy, potentially inappropriate medications, and drug-drug interactions in vulnerable older adults with advanced cancer initiating cancer treatment. Oncologist. 2022;27(7):e580-8. https://doi.org/10.1093/oncolo/oyac053
Garcia TS, Rocha BS, Castro SMJ, Heineck I. Potentially inappropriate medications for older adults in a primary healthcare unit in southern Brazil. Int J Clin Pharm. 2020;42(3):911-22. https://doi.org/10.1007/s11096-020-01048-7
Farias AD, Lima KC, Oliveira YMC, Leal AAF, Martins RR, Freitas CHSM. Prescription of potentially inappropriate medications for the elderly: a study in Primary Health Care. Cien Saude Colet. 2021;26(5):1781-92. https://doi.org/10.1590/1413-81232021265.04532021
Maes ML, Fixen DR, Linnebur SA. Adverse effects of proton-pump inhibitor use in older adults: a review of the evidence. Ther Adv Drug Saf. 2017;8(9):273-97. https://doi.org/10.1177/2042098617715381
Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain — United States, 2016. MMWR Recomm Rep. 2016;65(1):1-49. https://doi.org/10.15585/mmwr.rr6501e1
Abrahamsson T, Berge J, Öjehagen A, Håkansson A. Benzodiazepine, z-drug and pregabalin prescriptions and mortality among patients in opioid maintenance treatment-A nation-wide register-based open cohort study. Drug Alcohol Depend. 2017;174:58-64. https://doi.org/10.1016/j.drugalcdep.2017.01.013
Maust DT, Kales HC, Wiechers IR, Blow FC, Olfson M. No end in sight: benzodiazepine use in older adults in the United States. Am Geriatr Soc. 2016;64(12):2546-53. https://doi.org/10.1111/jgs.14379
McIntosh B, Clark M, Spry C. Benzodiazepines in older adults: a review of clinical effectiveness, cost-effectiveness, and guidelines. Ottawa: Canadian Agency for Drugs and Technologies in Health; 2011. Available from: https://www.ncbi.nlm.nih.gov/books/NBK174561/. Accessed Dec 2, 2022.
Oliveira ECT, Louvison MCP, Teixeira DSC, Menezes TN, Rosa TER, Duarte YAO. Difficulties in accessing health services among the elderly in the city of São Paulo-Brazil. PLoS One. 2022;17(5):e0268519. https://doi.org/10.1371/journal.pone.0268519
Cruz PKR, Vieira MA, Carneiro JA, Costa FM, Caldeira AP. Difficulties of access to health services among non-institutionalized older adults: prevalence and associated factors. Rev Bras Geriatr Gerontol. 2020;23(6):e190113. https://doi.org/10.1590/1981-22562020023.190113
Machado TRL, Pádua CAM, Drummond PLM, Silveira LP, Malta JS, Santos RMM, et al. Use of fall risk-increasing drugs in older adults with multiple myeloma: a cross-sectional study. J Geriatr Oncol. 2022;13(4):493-8. https://doi.org/10.1016/j.jgo.2022.01.007
Tian F, Chen Z, Chen X, Zhao M. Increasing trends of polypharmacy and potentially inappropriate medication use in older lung cancer patients in China: a repeated cross-sectional study. Front Pharmacol. 2022;13:935764 https://doi.org/10.3389/fphar.2022.935764
Umit EG, Baysal M, Bas V, Asker I, Kirkizlar O, Demir AM. Polypharmacy and potentially inappropriate medication use in older patients with multiple myeloma, related to fall risk and autonomous neuropathy. J Oncol Pharm Pract. 2020;26(1):43-50. https://doi.org/10.1177/1078155219835303
Hshieh TT, Dumontier C, Jaung T, Bahl NE, Magnavita ES, Hawley CE, et al. Impact of polypharmacy and potentially inappropriate medications among older adults with blood cancers. Blood. 2021;138(S1):4089. https://doi.org/10.1182/blood-2021-148731
Leger DY, Moreau S, Signol N, Fargeas JB, Picat MA, Penot A, et al. Polypharmacy, potentially inappropriate medications and drug-drug interactions in geriatric patients with hematologic malignancy: observational single-center study of 122 patients. J Geriatr Oncol. 2018;9(1):60-7. https://doi.org/10.1016/j.jgo.2017.07.015
Kimura T, Fujita M, Shimizu M, Sumiyoshi K, Bansho S, Yamamoto K, et al. Effectiveness of pharmacist intervention for deprescribing potentially inappropriate medications: a prospective observational study. J Pharm Health Care Sci. 2022;8(1):12. https://doi.org/10.1186/s40780-022-00243-0
Notes
This study received no specific funding from agencies in the public, commercial, or non-profit sectors.
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Universidade Federal de Minas Gerais. Informed consent was obtained from all individual participants included in the study
Author notes
Correspondence data Adriano Max Moreira Reis – Av. Presidente Antônio Carlos, 6627 – Pampulha – CEP: 31270-901 – Belo Horizonte (MG), Brazil. E-mail: amreis@ufmg.br
Conflict of interest declaration
The authors declare no conflict of interest.
