ORIGINAL ARTICLE
Frailty does not affect prognostic markers in patients with acute coronary syndrome: results from a Brazilian university hospital
Fragilidade não afeta marcadores prognósticos em pacientes com síndrome coronariana aguda: resultados de um hospital universitário no Brasil
Frailty does not affect prognostic markers in patients with acute coronary syndrome: results from a Brazilian university hospital
Geriatrics, Gerontology and Aging, vol. 18, e0000061, 2024
Sociedade Brasileira de Geriatria e Gerontologia, SBGG
Received: 10 August 2023
Accepted: 24 December 2023
Abstract
Objective: To evaluate frailty and its relationship with prognostic markers in hospitalized patients with acute coronary syndrome.
Methods: This cross-sectional study with a prospective variable analysis (prognostic markers) involved adults of both sexes aged ≥ 50 years with acute coronary syndrome. Patients with ≥ 3 of the following criteria were considered frail: 1) unintentional weight loss; 2) exhaustion (assessed by self-reported fatigue); 3) low handgrip strength; 4) low physical activity level; and 5) low gait speed. The included prognostic markers were: metabolic changes (lipid and glycemic profile), changes in inflammatory status (C-reactive protein), thrombolysis in myocardial infarction risk score, troponin level, angioplasty or surgery, hospitalization in the intensive care unit, length of hospital stay, and hospital outcome.
Results: The sample consisted of 125 patients, whose mean age was 65.5 (SD, 8.7) years. The prevalence of frailty was 48.00%, which was higher in women (PR = 1.55; 95%CI 1.08– 2.22; p = 0.018) and patients with systemic arterial hypertension (PR = 2.18; 95%CI 1.01–5.24; p = 0.030). Frailty was not associated with age, cardiac diagnosis, or prognostic markers (p > 0.05).
Conclusions: Frailty was highly prevalent in patients with acute coronary syndrome, affecting almost half of the sample, particularly women and patients with hypertension, irrespective of age. However, despite its high prevalence, frailty was not associated with markers of metabolic change or poor prognosis.
Keywords: Frailty+ acute coronary syndrome+ prognosis.
INTRODUCTION
Frailty is a complex clinical syndrome associated with adverse health outcomes such as dependence, falls and injuries, acute illness, slow recovery from illnesses, hospitalization, long-term institutionalization, and increased mortality.1
The most widely accepted model for assessing frailty is that of Fried et al.,2 which considers individuals meeting ≥ 3 of the following criteria to be frail: unintentional weight loss, exhaustion, low handgrip strength, low physical activity level, and low gait speed.
Although frailty is more prevalent among individuals with advanced age, it is not limited to older adults. The World Health Organization has suggested that, without intervention, functional capacity begins declining in middle age.3 Previous evidence suggests the importance of assessing frailty prior to old age, since it may help identify contributing factors to pre-frailty and frailty, which could lead to early interventions.4
Frailty is increasingly recognized as a risk factor for adverse cardiovascular outcomes.5,6,7 The frequency of frailty among individuals with cardiovascular diseases can be up to 3 times higher than in those without the disease.8 Previous studies have investigated the impact of frailty on patients with acute coronary syndrome (ACS), suggesting a relationship with negative clinical outcomes.9,10 Therefore, frailty assessment may help refine risk stratification and aid in the decision-making process for patients with ACS.11
Although frailty is more frequently investigated in patients with heart failure or valve disease, it has not yet been included in the routine assessment of patients with ACS, and guidelines for its early identification using appropriate tools in the post-ACS period are still unclear and unsatisfactory.12 However, the association between frailty and ACS has crucial implications, since it has been associated with worse clinical outcomes, such as increased mortality risk, longer hospital stays, rehabilitation difficulties, re-hospitalization, and specific complications, such as delirium and functional dependence, as well as higher resource consumption and hospital expense.12,13 In this context, the aim of this study was to assess frailty and its association with prognostic markers in hospitalized patients with ACS.
METHODS
This cross-sectional study prospectively analyzed prognostic markers in adults aged ≥ 50 years who had been diagnosed with ACS and were admitted to a university hospital in northeastern Brazil between April 2022 and October 2022.
The exclusion criteria were clinical conditions that precluded handgrip strength or gait speed testing (eg, patients who were bedridden, amputees, disoriented, and/or had severe clinical conditions). Patients with heart failure, dialysis-dependent nephropathy, chronic obstructive pulmonary disease, HIV, and cancer were also excluded.
The sample size was calculated based on an alpha error of 5% and a beta error of 20%. The estimated correlation between weight loss percentage (one of the criteria for frailty) and length of hospital stay was 0.5 (p), which was obtained from a pilot study of the first 30 patients enrolled in the study, with a variability of 0.18 (d2). Thus, a minimum sample of 121 patients was required. This number was increased by 10% for potential losses, resulting in a total of 134 participants.
This study was approved by the research ethics committee of the University of Pernambuco’s Emergency Cardiology Unit (decision 29975420.9.0000.5192). All eligible patients were informed of the study’s objectives and methodology, and those who agreed to participate provided written informed consent. Data were collected within 72 hours of admission to the ward.
Based on the criteria of Fried et al.,2 frailty syndrome was diagnosed when ≥ 3 of the following were present:
- 1. unintentional weight loss
- 2. self-reported exhaustion or fatigue
- 3. low handgrip strength
- 4. low physical activity level
- 5. low gait speed.
To measure weight loss, participants were asked to report unintentional weight loss of ≥ 4.5 kg or ≥ 5% of their body weight in the past year. The weight loss percentage was then calculated using the formula: (normal weight – current weight) x 100/normal weight.2
Fatigue was assessed using 2 items from the Center for Epidemiological Studies Depression Scale: “I felt that everything I did was an effort” and “I could not ‘get going’.” Patients who answered “Most or all the time” to at least 1 of these items were scored as weak.14
The participants’ muscle strength was determined through a handgrip strength test15 with a Jamar digital hand dynamometer (Sammons Preston Rolyan, Bolingbrook, IL, USA). The dynamometer’s grip was individually adjusted to each individual’s hand size, and the dominant arm was used for the test. The participants were instructed to exert as much force as possible with their hand while sitting with their hips and knees flexed at 90º, their shoulders in a neutral position, their elbow flexed at 90º, and their wrist in a neutral position. We followed the technique recommended by the American Society of Hand Therapists (1992), ie, the arm was kept immobile, allowing only flexion of the interphalangeal and metacarpophalangeal joints. The test was performed 3 times, with a 15-second interval between each attempt. The highest value was recorded in kg/F.2. The patients were verbally encouraged during the test to make a maximum effort.
Based on previous studies, the cut-off points for low muscle strength in individuals aged ≥ 60 years were < 27 kg/f for men and < 16 kg/f for women.15 For individuals aged between 50 and 60 years, muscle strength was considered low when the values were below the tenth percentile of the reference standard for the age group and sex.16
Physical performance was measured using the gait speed test according to International Academy on Nutrition and Aging parameters. The test was conducted on a flat 4-meter course, and patients were instructed to walk at their normal pace. The time taken to complete the course was recorded. The assessment was performed twice, and the shortest time was used as in the analysis. Gait speed < 0.8 meters/second (m/s) was considered low.15
Several prognostic markers were considered: troponin levels, the thrombolysis in myocardial infarction score (which assesses the risk of post-infarction complications), the need for coronary angioplasty or myocardial revascularization surgery, hospitalization complications (such as admission to the intensive care unit), length of hospital stay (in days), and hospitalization outcome (discharge or death).
The following sociodemographic variables were collected from the patients’ electronic medical records: age (years), sex, race (Black, White, or multiracial), and years of education. Clinical variables included cardiac diagnosis, the presence of comorbidities (such as systemic arterial hypertension or diabetes mellitus), and the presence of metabolic syndrome.
According to NCEP-ATP III guidelines, metabolic syndrome was diagnosed when ≥ 3 of the following parameters were altered: waist circumference, triglycerides, HDL-cholesterol, blood pressure, and fasting glucose. The following values were considered altered: triglycerides ≥ 50 mg/dL, blood pressure ≥ 130/85 mmHg, blood glucose ≥ 100 mg/ dL, HDL-cholesterol < 40 mg/dL for men and < 50 mg/dL for women, and waist circumference ≥ 102 cm for men and ≥ 88 cm for women.
The biochemical variables included C-reactive protein, lipid profile, and fasting glucose. The reference values were total cholesterol < 190 mg/dL and low-density-lipoprotein cholesterol < 70 mg/dL, according to the Brazilian Guidelines for Dyslipidemia and Atherosclerosis Prevention (2017). High C-reactive protein was defined as ≥ 5.0 mg/ dL, the cut-off used by the institution’s laboratory.
The anthropometric variables included body mass index (BMI) and calf circumference. To calculate BMI, weight in kilograms (kg) was divided by height in m2 (weight/ height2), with the result expressed in kg/m2. BMI was classified according to World Health Organization criteria (1997) for adults aged 50–59 years: malnutrition (BMI < 18.5 kg/m2), eutrophy (BMI between 18.5–24.9 kg/m2), overweight (BMI between ≥ 25 kg/m2).
For analytical purposes, overweight and obesity were grouped together, with BMI ≥ 25 kg/m2 considered overweight. Lipschitz’s BMI classification criteria for older adults were used, with malnutrition considered < 22 kg/ m2, eutrophy considered 22-27 kg/m2, and overweight considered ≥ 27 kg/m2.17 Calf circumference was considered low when the area of greatest volume was ≤ 34 cm for men and ≤ 33 cm for women.18
The data were analyzed in IBM SPSS 13.0 IBM SPSS Statistics (IBM, Armonk, NY, USA). The normality of continuous variables was assessed using the Kolmogorov-Smirnov test and was reported as mean or median depending on the distribution pattern.
The association between frailty and prognostic variables was assessed using either Pearson’s chi-square test or Fisher’s exact test. To compare means and medians between frail and non-frail groups, Student’s t-test or the Mann-Whitney U test was used, respectively. The significance level for all tests was set at p < 0.05.
RESULTS
A total of 134 eligible patients were enrolled, of whom 9 were excluded from the analysis due to data inconsistencies or missing information, resulting in a final sample of 125 patients.
The mean age of the patients was 65.5 (SD, 8.7) years; most were men (59.20%), older adults (76.80%), multiracial (74.40%), and had a low education level (66.07%). Regarding clinical variables, 38.40% had acute myocardial infarction with ST-segment elevation, 86.40% had systemic arterial hypertension, and 45.60% had diabetes mellitus. The mortality rate was 2.40% (Table 1).
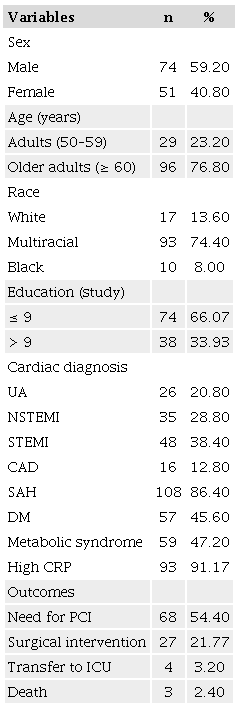
CAD: coronary artery disease; CRP: C-reactive protein; DM: diabetes mellitus; ICU: intensive care unit; NSTEMI: non-ST segment elevation myocardial infarction; PCI: percutaneous coronary intervention; SAH: systemic arterial hypertension; STEMI: ST-segment elevation myocardial infarction; UA: unstable angina.
A total of 48.00% were considered frail, with most common criteria being physical inactivity (74.40%) and low gait speed (70.40%). An additional 30.40% of the patients met 2 criteria for frailty and were considered pre-frail. Regarding BMI, 55.20% were overweight and 11.20% were underweight; 39.20% had a low calf circumference (Table 2).
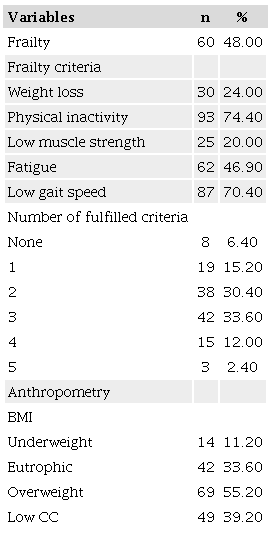
BMI: body mass index; CC: calf circumference.
Frailty was more prevalent in women (prevalence ratio = 1.55; 95% CI 1.08–2.22; p = 0.018) and in patients with systemic arterial hypertension (prevalence ratio = 2.18; 95% CI 1.01–5.24; p = 0.030). However, frailty was not associated with age, cardiac diagnosis, or prognostic markers (p > 0.05) (Table 3).
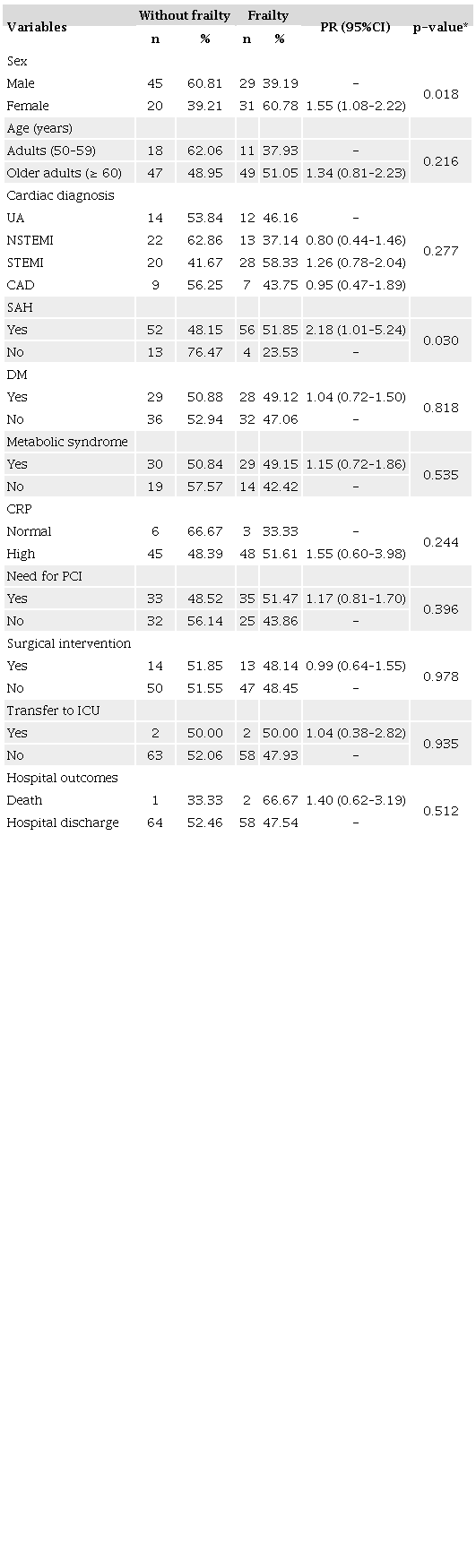
* Pearson chi-square test; CAD: coronary artery disease; CRP: C-reactive protein; DM: diabetes mellitus; ICU: intensive care unit; NSTEMI: non-ST segment elevation myocardial infarction; PCI: percutaneous coronary intervention; PR: Prevalence Ratio; SAH: systemic arterial hypertension; STEMI: ST-segment elevation myocardial infarction; UA: unstable angina.
When cardiometabolic parameters were compared between frail and non-frail patients, no significant differences were found for lipid profile or glycemia (p > 0.05). Length of hospital stay and the thrombolysis in myocardial infarction risk score did not differ significantly between the groups (p > 0.05), although non-frail patients had higher troponin levels (p < 0.001) (Table 4).
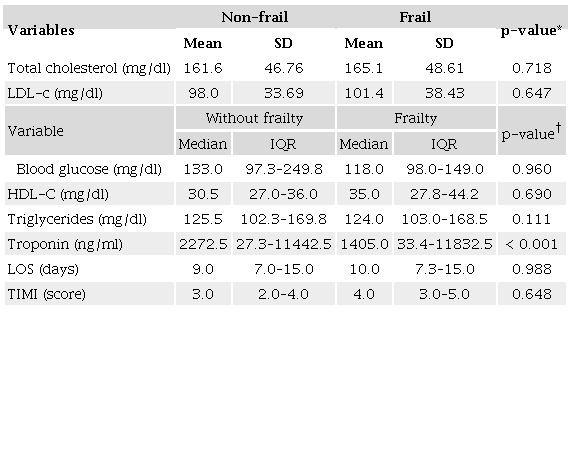
* Student’s t-test; † Mann-Whitney U Test; HDL-C: high density lipid cholesterol; LDL-C: low density lipid cholesterol; LOS: length of hospital stay; IQR: interquartile range; TIMI: thrombolysis in myocardial infarction
DISCUSSION
We found that frailty was not associated with poor prognosis in patients with ACS (mean age was 65.5 [SD, 8.7] years) admitted to a Brazilian university hospital. However, the high prevalence of pre-frailty (30.40%) and frailty (48.00%) is consistent with previous studies, in which almost half of the participants hospitalized with ACS were considered frail.19,20 Many studies worldwide have reported a high prevalence of frailty in these patients, ranging from 30.00 to 49.00%.19,20,21,22,23 Frailty may impact the clinical course of coronary patients due to its pathophysiological link with inflammation.12
We found no significant association between frailty and metabolic markers, such as diabetes mellitus, metabolic syndrome, HDL, triglycerides, and blood glucose, or with cardiac prognosis, in contrast with previous studies, which found frailty to be a risk factor for short-term adverse outcomes and mortality in patients with ACS (≤ 6 months).19,20
We point out that our investigation was conducted in a university hospital setting and involved a multidisciplinary team, including physicians, nutritional support, psychological assistance, and cardiac rehabilitation. This comprehensive approach could have contributed to holistic management and, thus, better overall outcomes, mitigating the impact of frailty. Moreover, the low mortality rate could have limited certain analyses and observations. Prospective investigations are needed to assess the prognostic role of frailty in the medium and long term, beyond hospitalization. It is important to note that medication use, such as lipid-lowering agents, glycemic control therapy, and medication for secondary ACS prevention, was not evaluated in this study.
Previous studies have indicated frailty’s potential as an independent marker for unfavorable outcomes, with few investigations showing contrary results. There may be publication bias in this context (ie, a greater likelihood that studies finding a positive association would be published). Moreover, some unexplored variables have been identified as important in the study of frailty and adverse outcomes, such as activities of daily living and quality of life.24,25 Another plausible explanation for the lack of an association between frailty and negative outcomes is that frail patients less frequently receive invasive treatment.25,26 Lower exposure to invasive procedures may reduce the risk of complications among these patients.
Although the relationship between frailty and adverse outcomes in ACS has not yet been fully elucidated, it is important to consider that frailty entails the loss of biological reserves, which could lead to the failure of homeostatic mechanisms in response to stress events, potentially increasing the risk of adverse outcomes compared to non-frail individuals.27
However, despite our findings, frailty should be systematically evaluated in patients with ACS for several reasons. First, because frailty is a reversible condition that can be improved through external interventions, it must be screened early to ensure that appropriate treatment measures are implemented. Second, because frailty can provide additional valuable prognostic insights to existing cardiovascular risk models, it can help clinical staff better define care pathways for their patients and improve outcomes.28
It is also important to note that the prognosis of patients with ACS is not solely determined by their “biological age”, and frailty can serve as a valuable predictor of adverse outcomes, optimizing treatment of comorbid conditions and facilitating early recognition of complications. Therefore, frailty assessment should help identify patients with ACS who have a high burden of comorbidities, which may outweigh the benefits of invasive treatments.29
There is no evidence in the literature to explain the higher troponin levels we observed in non-frail patients. This result may be a chance finding or it could have been influenced by confounding factors not accounted for in this study. Further research is needed to investigate this association and clarify its underlying mechanisms.
This study has some limitations that should be acknowledged. The main limitation is its relatively small sample selected from a single center, which limits the generalizability of the results. Additionally, due to its cross-sectional design, cause-and-effect relationships cannot be established. Nevertheless, we believe that these limitations do not compromise the validity of our findings, which can provide useful insight for future research in this area.
CONCLUSION
Frailty was highly prevalent in patients with ACS, affecting almost half of the patients, particularly women and patients with systemic arterial hypertension, regardless of age. However, frailty was not associated with metabolic markers or poor prognosis. Although the relationship between frailty and adverse outcomes in patients with ACS has not been fully elucidated, further research is needed to determine whether this condition can provide additional prognostic value beyond traditional risk factors.
REFERENCES
Lourenço RA, Moreira VG, Mello RGB, Santos IS, Lin SM, Pinto ALF, et al. Brazilian consensus on frailty in older people: concepts, epidemiology and evaluation instruments. Geriatr Gerontol Aging. 2018;12(2):121-35. https://doi.org/10.5327/Z2447-211520181800023
Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56(3):M146-56. https://doi.org/10.1093/gerona/56.3.m146
Word Health Organization. Global status report on noncommunicable disease 2014. Geneva: WHO; 2014.
Gordon SJ, Baker N, Kidd M, Maeder A, Grimmer KA. Pre-frailty factors in community-dwelling 40-75 year olds: opportunities for successful ageing. BMC Geriatr. 2020;20(1):96. https://doi.org/10.1186/s12877-020-1490-7
Theou O, O’Connell MDL, King-Kallimanis BL, O’Halloran AM, Rockwood K, Kenny RA. Measuring frailty using self-report and test-based health measures. Age Ageing. 2015;44(3):471-7. https://doi.org/10.1093/ageing/afv010
Hanlon P, Nicholl BI, Jani BD, Lee D, McQueenie R, Mair FS. Frailty and pre-frailty in middle-aged and older adults and its association with multimorbidity and mortality: a prospective analysis of 493 737 UK Biobank participants. Lancet Public Health. 2018;3(7):e323-32. https://doi.org/10.1016/S2468-2667(18)30091-4
Palmer KT, D’Angelo S, Harris EC, Linaker C, Gale CR, Evandrou M, et al. Frailty, prefrailty and employment outcomes in Health and Employment After Fifty (HEAF) study. Occup Environ Med. 2017;74(7):476-82. https://doi.org/10.1136/oemed-2016-104103
Frisoli Junior A, Ingham SJM, Martin FG, Borges J, Paes AT, Gonçalves Junior I, et al. Frailty is an independent predictor of early death in elderly outpatients with cardiovascular disease in the sarcos study. Rev Soc Cardiol Estado São Paulo Supl. 2018;28(3):331-5. http://dx.doi.org/10.29381/0103-8559/20182803331-5
Bottura C, Arcêncio L, Chagas HMA, Evora PRB, Rodrigues AJ. Frailty among non-elderly patients undergoing cardiac surgery. Arq Bras Cardiol. 2020;115(4):604-10. https://doi.org/10.36660/abc.20190082
Tonet E, Pavasini R, Biscaglia S, Campo G. Frailty in patients admitted to hospital for acute coronary syndrome: when, how and why? J Geriatr Cardiol. 2019;16(2):129-37. https://doi.org/10.11909/j.issn.1671-5411.2019.02.005
Moraes AAI, Chammas AZL, Melo Neto J, Mendes LCM, Aguiar YS, Ramos RF. Bleeding in non-ST-segment elevation acute coronary syndrome. Rev Bras Ter Intensiva. 2012;24(3):284-93. PMID: 23917831
Giallauria F, Di Lorenzo A, Venturini E, Pacileo M, D’Andrea A, Garofalo U, et al. Frailty in acute and chronic coronary syndrome patients entering cardiac rehabilitation. J Clin Med. 2021;10(8):1696. https://doi.org/10.3390/jcm10081696
Vicent L, Martínez-Sellés M. Frailty and acute coronary syndrome: does gender matter? J Geriatric Cardiol. 2019;16(2):138-44. https://doi.org/10.11909/j.issn.1671-5411.2019.02.007
Batistoni SST, Neri AL, Cupertino APFB. Validity of the Center for Epidemiological Studies Depression Scale among Brazilian elderly. Rev Saude Publica. 2007;41(4):598-605. https://doi.org/10.1590/s0034-89102007000400014
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16-31. https://doi.org/10.1093/ageing/afy169
Amaral CA, Amaral TLM, Monteiro GTR, Vasconcellos MTL, Portela MC. Hand grip strength: reference values for adults and elderly people of Rio Branco, Acre, Brazil. PLoS One. 2019;14(1):e0211452. https://doi.org/10.1371/journal.pone.0211452
Lipschitz DA. Screening for nutritional status in the elderly. Prim Care. 1994;21(1):55-67. PMID: 8197257.
Barbosa-Silva TG, Bielemann RM, Gonzalez MC, Menezes AMB. Prevalence of sarcopenia among community-dwelling elderly of a medium-sized South American city: results of the COMO VAI? study. J Cachexia Sarcopenia Muscle. 2016;7(2):136-43. https://doi.org/10.1002/jcsm.12049
Van Nguyen T, Le D, Tran KD, Bui KX, Nguyen TN. Frailty in older patients with acute coronary syndrome in Vietnam. Clin Interv Aging. 2019;14:2213-22. https://doi.org/10.2147/CIA.S234597
Kang L, Zhang SY, Zhu WL, Pang HY, Zhang L, Zhu ML, et al. Is frailty associated with short-term outcomes for elderly patients with acute coronary syndrome? J Geriatr Cardiol. 2015;12(6):662-7. https://doi.org/10.11909/j.issn.1671-5411.2015.06.010
Alonso Salinas GL, Sanmartin Fernandez M, Pascual Izco M, Asenjo RM, Recio-Mayoral A, Salvador Ramos L, et al. Frailty is a short-term prognostic marker in acute coronary syndrome of elderly patients. Eur Heart J Acute Cardiovasc Care. 2016;5(5):434-40. https://doi.org/10.1177/2048872616644909
Alonso Salinas GL, Sanmartin M, Pascual Izco M, Rincon LM, Martin-Acuna A, Pueyo P P, et al. The role of frailty in acute coronary syndromes in the elderly. Gerontology. 2018;64(5):422-29. https://doi.org/10.1159/000488390
Llaó I, Ariza-Solé A, Sanchis J, Alegre O, López-Palop R, Formiga F, et al. Invasive strategy and frailty in very elderly patients with acute coronary syndromes. EuroIntervention 2018;14(3):e336-e342. https://doi.org/10.4244/EIJ-D-18-00099
Pereira JPC, Diniz AS, Lemos MCC, Ramiro CPSP, Cabral PC. Frailty but not low muscle quality nor sarcopenia is independently associated with mortality among previously hospitalized older adults: a prospective study. Geriatr Gerontol Int. 2023;23(10):736-43. https://doi.org/10.1111/ggi.14660
Dou Q, Wang W, Wang H, Ma Y, Hai S, Lin X, et al. Prognostic value of frailty in elderly patients with acute coronary syndrome: a systematic review and meta-analysis. BMC Geriatr. 2019:19(1):222. https://doi.org/10.1186/s12877-019-1242-8
Kwok CS, Lundberg G, Al-Faleh H, Sirker A, Van Spall HGC, Michos ED, et al. Relation of frailty to outcomes in patients with acute coronary syndromes. Am J Cardiol. 2019;127(7):1002-11. https://doi.org/10.1016/j.amjcard.2019.07.003
Bebb O, Smith FG, Clegg A, Hall M, Gale C P. Frailty and acute coronary syndrome: a structured literature review. Eur Heart J Acute Cardiovasc Care. 2018;7(2):166-75. https://doi.org/10.1177/2048872617700873
Lyu H, Wang C, Jiang H, Wang P, Cui J. Prevalence and determinants of frailty in older adult patients with chronic coronary syndrome: a cross-sectional study. BMC Geriatr. 2021;21(1):519. https://doi.org/10.1186/s12877-021-02426-0
Tonet E, Campana R, Caglioni S, Gibiino F, Fiorio A, Chiaranda G, et al. Tools for the Assessment of the Malnutrition Status and Possible Interventions in Elderly with Cardiovascular Diseases. J Clin Med. 2021;10(7):1508. https://doi.org/10.3390/jcm10071508
Notes
This study was approved by the research ethics committee of the University of Pernambuco’s Emergency Cardiology Unit. All patients provided written informed consent.
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
Author notes
Correspondence data Claudia Porto Sabino Pinho – Rua dos Palmares, s/n – Santo Amaro – CEP: 50100-060 – Recife (PE), Brazil. E-mail: claudiasabinopinho@hotmail.com
Conflict of interest declaration
The authors declare no conflicts of interest.
