Reportes verduras
Potential of colored sweet potato genotypes as source of bioactive compounds
Potencial de los genotipos de camote coloreado como fuente de compuestos bioactivos
Potential of colored sweet potato genotypes as source of bioactive compounds
Revista Iberoamericana de Tecnología Postcosecha, vol. 21, no. 2, 2020
Asociación Iberoamericana de Tecnología Postcosecha, S.C.
Received: 27 October 2020
Accepted: 03 December 2020
Published: 31 December 2020
Abstract: The aim of this work was to evaluate the bioactive composition and antioxidant capacity of fleshes, peels and fleshes + peels of six genotypes of raw colored sweet potatoes, in addition to determining the physical-chemical attributes of the samples of raw flesh + peel and after thermal treatment. Orange-fleshed sweet potatoes 'BRS Amélia', 'CPNH 1365' and 'CPNH 1358' and purple-fleshed sweet potatoes 'JN RX1', JN RX2 and 'JN RX7' were analyzed for the content of total phenolic compounds, total flavonoids, antioxidant capacity, total carotenoids (orange-fleshed genotypes) and total monomeric anthocyanins (purple-fleshed genotypes). Sweet potatoes were boiled in water for 12 minutes, separated in peel and flesh and analyzed after powdering in low temperature. Peel + flesh of orange-fleshed ‘CNPH 1365’ showed the highest levels of phenolic compounds (286.44 mg/100g) and total flavonoids (141.77 mg/100g). Peel + flesh of ‘CPNH 1358’ showed high levels of total carotenoids (11.21 mg/100g). Peel + flesh of purple sweet potato ‘JN RX1’ contains higher levels of total phenols (324.78 mg/100g) and ‘JN RX2’ showed the highest levels of flavonoids (160.17 mg/100g), as well as total anthocyanins (17.63 mg/100g). The 'JN RX7' peels also showed high anthocyanins content (15.64 mg/100g). After cooking, orange-fleshed sweet potatoes showed decreasing of titratable acidity levels and increasing of soluble solids, which contributed to higher ratio values. The thermal processing induced an increase in the ratio in purple-fleshed sweet, with the exception of ‘JN RX7’. Our results show that colored sweet potatoes are potential sources of phytochemicals, demonstrating that the use of this tuber, especially the peels, in the food and pharmaceutical industries should be expanded.
Keywords: Ipomoea batatas, carotenoids, phenolic compounds, antioxidants, boiling.
Resumen: El objetivo de este trabajo fue evaluar la composición bioactiva y capacidad antioxidante de pulpas, cáscaras y pulpas + cáscaras de seis genotipos de camote coloreado crudo, además de determinar los atributos físico-químicos de las muestras de pulpa + cáscara crudas y después del tratamiento térmico. Se analizaron los camotes anaranjados 'BRS Amélia', 'CPNH 1365' y 'CPNH 1358' y los camotes morados 'JN RX1', JN RX2 y 'JN RX7' para determinar el contenido de compuestos fenólicos totales, flavonoides totales, capacidad antioxidante, carotenoides totales (genotipos de pulpa naranja) y antocianinas monoméricas totales (genotipos de pulpa púrpura). Los camotes se hirvieron en agua durante 12 minutos, se separaron en cáscaras y pulpa y se analizaron después de pulverizarlas a baja temperatura. La cáscara + pulpa del "CNPH 1365" de pulpa anaranjada mostró los niveles más altos de compuestos fenólicos (286.44 mg /100g) y flavonoides totales (141.77 mg /100g). La cáscara + pulpa de "CPNH 1358" mostró niveles altos de carotenoides totales (11.21 mg /100g). La cáscara + pulpa del camote morado 'JN RX1' contiene niveles más altos de fenoles totales (324.78 mg/100g) y 'JN RX2' mostró los niveles más altos de flavonoides (160.17 mg/100g), así como antocianinas totales (17.63 mg/100g). Las cáscaras 'JN RX7' también mostraron un alto contenido de antocianinas (15.64 mg/100g). Después de la cocción, las batatas de pulpa anaranjada mostraron una disminución de los niveles de acidez titulable y un aumento de los sólidos solubles, lo que contribuyó a valores de proporción más altos. El procesamiento térmico indujo un aumento en lo ratio de camotes morados, con la excepción de "JN RX7". Nuestros resultados muestran que los camotes coloreadas son fuentes potenciales de fitoquímicos, demostrando que el uso de este tubérculo, especialmente las cáscaras, en las industrias alimentaria y farmacéutica debe expandirse.
Palabras clave: Ipomoea batatas, carotenoides, compuestos fenólicos, antioxidantes, cocción.
INTRODUCTION
Phenolic compounds and carotenoids are formed through reactions of plant secondary metabolism and are important to protect the cells and tissues against damage induced by biotic and abiotic stress (Borges et al., 2017). In the edible parts of plants (fruits, tubers, rhizomes, etc.), phenolic compounds contribute, in addition to protection, in the characteristics of texture, flavor, aroma, astringency and color (Maoka, 2020). The presence of these secondary metabolites becoming an interesting biomass for the exploitation of phenolic compounds and carotenoids, molecules considered free radical scavengers. Some authors report, for example, that these bioactive compounds, appear in higher levels around the cortex and or close to the peels (Islam et al., 2016; Jung et al., 2011).
In human nutrition, these phytochemicals are being researched due to their antioxidant properties with numerous health benefits and may present antibacterial, antitumor, antiviral, antimutagenic activity, as well as act as cardioprotective agents (Sagar et al., 2018). Studies also report that the consumption of food with significant amounts of these antioxidants may reduce the risk of developing degenerative diseases, such as cancer, and contributes to the delay of cellular aging (Lima et al., 2014; Minatel et al., 2017), emphasizing the importance of these compounds in human nutrition.
Even though they have great antioxidant potential, food peels are usually discarded, either for domestic consumption or in industry. Peel separation may induce deterioration of plant tissues and browning induced by enzymes, such as polyphenoloxidase and peroxidase, which use phenolic compounds as substrate (Simões et al., 2020), damaging the quality of the final product.
A very interesting issue is the diversity of sweet potato tubers, with different colors of peel (white, cream, yellow, orange, pink and red) and flesh (white, cream, orange, yellow and purple). This coloration is generally influenced by the level of metabolites found in the tubers (Wang et al., 2018), e.g., orange sweet potatoes present β-carotene (carotenoid) as the majoritary compound (Van Jaarsveld et al., 2005); in contrast, purple sweet potatoes have in their chemical composition bioactive compounds such as phenolic acids, flavonoids and anthocyanins (De Albuquerque et al., 2019). The exploitation of this diversity, that is, the use of varieties which fleshes and peels with different contents of antioxidant compounds, might be explored by the food and pharmaceutical industries, adding value to the product and, concomitantly, increasing the income of the rural producer.
In recent surveys conducted by our group on the use of peels in tuberous foods (potatoes, sweet potatoes, beets, carrots, cassava, among others), 75.7% responded that they discarded this material, 11.6% that they consumed and 12.7% who reuse it for other purposes, such as animal feed and composting (unpublished data), demonstrating the need for studies aimed at the purpose of using this biomass. Most sweet potato consumption is domestic and, although it is a tuber with industrial potential, there is little visibility in the use of its by-products by the industry. The peel is an important by-product resulting from the processing of food and may be a potential source of bioactive compounds.
Thus, our objective was to determine the contents of total phenolic compounds, total carotenoids and in vitro antioxidant activity in raw and thermally processed samples of flesh, peel and flesh + peel of different colored sweet potatoes.
MATERIAL AND METHODS
Purple-fleshed sweet potato genotypes 'JN RX1', 'JN RX2' and 'JN RX7' were purchased from a producer in the region of Presidente Prudente, São Paulo (latitude 22° 7′ 39″S, longitude 51° 23′ 8″W and altitude 457 m). The orange-fleshed sweet potato genotypes 'BRS Amélia', 'CPNH 1365' and 'CPNH 1358' were grown in Botucatu , São Paulo (22° 52' 47”S latitude, 48° 25' 12”W longitude and 810 m altitude), at the Center of Tropical Roots and Starches (CERAT).
After sanitizing the raw tubers, the peels were removed using a manual peeler. The extremities of the tubers were discarded and the flesh, peel and flesh + peel (whole tuber) were powdered in liquid nitrogen and stored at -20° C. In all genotypes, total phenolic compounds, total flavonoids and antioxidant capacity (DPPH and MDA) were analyzed. In the tubers 'BRS Amélia', 'CPNH 1365' and 'CPNH 1358' (orange flesh), the total carotenoids were also analyzed and in 'JN RX1', 'JN RX2' and 'JN RX7' (purple flesh), total anthocyanins monomeric were evaluated.
The total phenols content was analyzed using the reagent Folin Ciocalteu (Singleton & Rossi, 1965). Fresh samples were homogenized in MeOH: acetic acid: water (80:1:19 v/v/v) and submitted to an ultrasonic bath for 30 min and centrifugation at 6000 rpm, for 15 min (5° C). The supernatant was removed and placed in an amber glass container. The process was repeated, totaling two extractions and the supernatants were mixed for the analysis. The results were calculated from a standard curve and expressed in mg of gallic acid equivalent/100g.
The total flavonoids content was analyzed according to the method of Zhishen et al. (1999) adapted by Pekal and Pyrzynska (2014). Fresh samples were homogenized in MeOH: water (70:30, v/v), vortexed for 3 min, sonicated (20 min) and centrifuged (6000 rpm, 15 min, 5 °C). After the addition of sodium nitrite (5%) and homogenization, and after 30 min, aluminum chloride (2%) was added and the tubes were homogenized again. After 6 min, 0.5 ml of sodium hydroxide (1M) was added. The samples were read at 510 nm after 10 min. The results were expressed in mg rutin equivalent/100g.
The ability of the samples to reduce the DPPH radical (2,2-diphenyl-1-picrylhydrazyl) was assessed following the methodology of Brand-Williams et al. (1995). The samples were extracted in MeOH: acetic acid: water (80: 1: 19, v/v/v) and, for the reaction, 1 ml of sample was added to 3 ml ethyl alcohol and 0.3 ml DPPH reagent. After 30 minutes, the spectrophotometer (517 nm) was read and the absorbance of the samples correlated with the absorbance of the control (white) resulted in the percentage of free radical scavenging, which was then converted to mg of Trolox /100g.
Free malondialdehyde (MDA) was determined using the methodology described by Heath and Packer (1968). The samples were homogenized in 0.25% thiobarbituric acid, 10% trichloroacetic acid and 89.75% water. The material remained in a water bath (90° C) for 60 min and was cooled in an ice bath. After centrifugation (6000 rpm, 5° C, 10 min), the samples were read in a spectrophotometer at wavelengths 560 nm and 600 nm. The results were expressed in nmol TBARS/g.
Total monomeric anthocyanins were determined using the differential pH method (Giusti and Wrolstad, 2001). Flesh, peel and flesh + peel samples were homogenized in MeOH: acetic acid: water (80:1:19 v/v/v), sonicated (20 min) and centrifuged (6000 rpm, 5° C, 15 min). The absorbance was measured at 510 and 700 nm, and the result was expressed in mg cyanidin-3-o-glycoside/100g.
The total carotenoids were analyzed according to Lichtenthaler (1987). Fresh samples were homogenized in acetone by sonication for 30 min. The extracts were centrifuged at 6000 rpm (15 min) and the absorbance was measured in a spectrophotometer at wavelengths of 661, 641 and 450 nm. The results were calculated and expressed in mg /100g of fresh weight.
The physical-chemical characteristics were carried out according to the methodology proposed by the Adolf Lutz Institute (2008) in the fleshes + peels of raw and cooked sweet potatoes. Orange-flesh and purple-flesh sweet potatoes were washed in running water, followed by sanitization in sodium hypochlorite (75%) for 10 minutes. The extremities of the tubers were discarded. Fleshes with peel was cut into slices (2 x 1 cm) and cooked in 500 ml of boiling water (100 ° C) for 12 minutes. The water was drained and after cooling, the samples were immediately evaluated. Total titratable acidity (TA) was determined in aqueous extract, using 1 g of the material homogenized in 10 ml of distilled water and titrated in 0.1 N NaOH. The results were expressed in percentage of citric acid/100g of fresh weight. The hydrogen potential (pH) was measured using a potentiometer (model Q Quimis - 400ª). The content of soluble solids (SS) in the samples was analyzed using a digital refractometer (Atago model, PAL-1), with results expressed in °Brix.
The analyzes were performed in triplicate and the data were submitted to analysis of variance (ANOVA), followed by Scott-Knott test (comparison of means) (p <0.05), with the aid of the SISVAR program. Principal component analysis (PCA), cluster analysis (HCA) and correlation test were performed using the XLSTAT software (version 2017; Addinsoft, France).
RESULTS AND DISCUSSION
In orange flesh potatoes ‘BRS Amélia’, ‘CPNH 1365’ and ‘CPNH 1358’ there was a significant variation in the levels of bioactive compounds, depending on the genotype and part analyzed (flesh, peel or flesh + peel). Sweet potato 'CPNH 1358' (orange-fleshed, Figure 1) presented the lowest levels of total phenolic compounds and total flavonoids (Table 1), either in the peel (163.39 and 76.63 mg/100g, respectively), in the flesh (185.37 and 86.25 mg/100g, respectively) or when using the whole tuber (flesh + peel) (178.65 and 81.76 mg/100g, respectively). In contrast, ‘CNPH 1365’ contains the highest levels of phenolic compounds and total flavonoids in the flesh and in the peel (286.44 and 141.77 mg/100g, respectively) (Table 1).
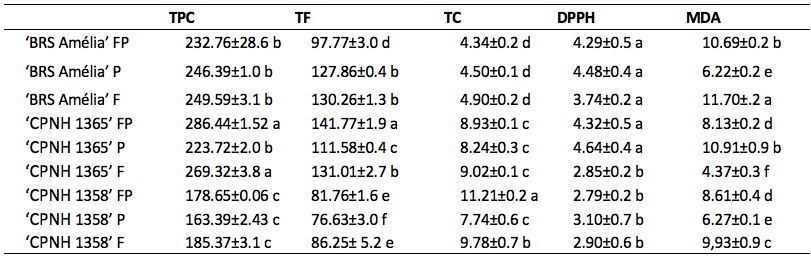
* Means followed by the same letter, in the column, do not differ statistically among themselves by Sckott-Knott test (p≤ 0.05).** FP= flesh + peel; P = peel and F = flesh.
In orange-fleshed sweet potato (cv. Paraná), Simões et al. (2020) described higher amounts of flavonoids (approximately 30% more), when compared to the cream-fleshed sweet potatoes (cv. ESAM 1). In the present study, ‘CPNH 1358’ contains higher total carotenoids in the flesh + peel (11.21 mg/100g) and in the flesh (9.78 mg/100g), while the other genotypes do not show differences between the analyzed parts. The total carotenoids content did not show great variations when the three genotypes were analyzed, regardless of analyzing only the flesh (4.90 to 9.78 mg/100g), only the skin (4.50 to 8.24 mg/100g) or the whole tuber (flesh + peel) (4.34 to 11.21 mg/100g) (Table 1). Although the genotype ‘CPNH 1358’ presents low levels of total phenolics, it shows high levels of total carotenoids when the whole tuber is analyzed (flesh + peel), compared to the other genotypes. It is worth emphasizing that ‘CPNH 1358’ present the most accentuated orange color (Figure 1).

FIGURE 1
Colored fleshed sweet potatoes genotypes: A) ‘BRS Amélia’; B) ‘CPNH 1358’; C) ‘CPNH 1365’; D) ‘‘JN RX1’’; E)’JN RX2’ and F)’JN RX7’
Orange-fleshed sweet potatoes contain carotenoids, mainly b-carotene in higher levels compared to α-carotene, both precursors of retinol (vitamin A) in the human body (Maoka, 2020; Simões et al., 2020). The intense orange color in fruits (Borges et al., 2019), vegetables (Diamante et al., 2020) and tubers (Simões et al., 2020) is a phenotypic characteristic that is correlated with the presence of provitamin A, corroborating with the results found in the present work. Vitamin A deficiency mainly affects children and pregnant women, which can cause developmental delays, blindness, inefficiency in the immune system and, depending on the severity, death (Alam et al., 2020). Thus, the consumption of ‘CNPH1358’, as well as ‘CNPH1365’, may be a good option for reducing hypovitaminosis A, considered a worldwide public health problem (Alam et al., 2020). It is worth mentioning that, in addition to provitamins, these pigments have antioxidant activity, reducing the effects promoted by free radicals and preventing a series of cardiovascular diseases (Kulczyński et al., 2017).
Aiming a grouping model of the different bioactive compounds analyzed in orange-fleshed sweet potato cultivars, principal component analysis (PCA) was applied (Figure 2). The PCA explained 80.27% of the data variance resulting from the analysis of the content of total phenols, total flavonoids and total carotenoids in orange-fleshed sweet potatoes (Figure 2). ‘CNPH 1365’ and ‘BRS Amélia’ contain the highest levels of (poly)phenols and show the highest antioxidant activities measured via DPPH, grouping into PC1 +, which explains 52.95% of the data variance. On the other hand, the ‘CNPH 1358’, which has the highest levels of carotenoids, showed less activity measured via DPPH and greater antioxidant capacity when analyzed by MDA (PC1-) method. Our results demonstrate the high antioxidant value of the peel, due to the presence of phenolic and carotenoid compounds, regardless of the method used. In the food industry, bio-waste sources of bioactive compounds are important in the incorporation of products such as natural dyes, improving the nutritional and functional quality of foods, demonstrating the potential for using this biomass (Martins and Ferreira, 2017). In the hierarchical cluster analysis (HCA) (Figure 3), 'CNPH 1358' presented specific grouping in relation to the analyzed parts of the tuber, differing from 'BRS Amélia' and from ‘CNPH1365’. In addition, it is possible to highlight that both in 'CNPH 1358' and in 'CNPH 1365', the levels of the compounds were predominant in the fleshes, influencing the similarity.
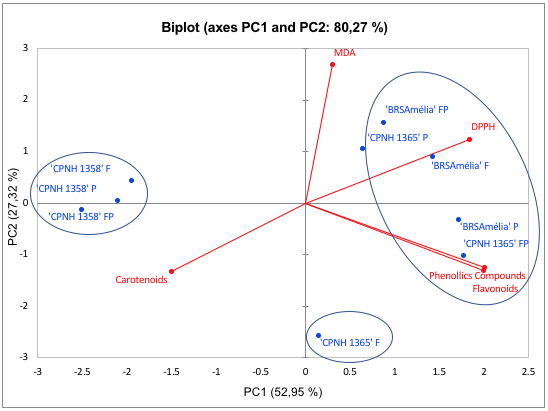
FIGURE 2
Two-dimensional projection and scores of the biochemical characteristics of total phenolic compounds, total flavonoids, total carotenoids, antioxidant activity DPPH and MDA in orange-fleshed sweet potatoes. The treatments are represented by the points, where F = flesh; P = peel and FP = flesh + peel
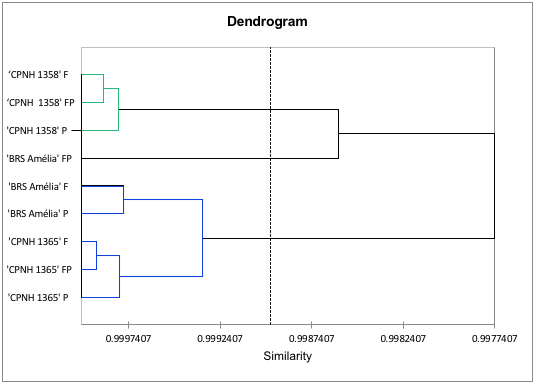
FIGURE 3
Hierarchical cluster analysis (HCA) of the biochemical characteristics of total phenolic compounds, total flavonoids, total carotenoids, antioxidant activity by DPPH and lipid peroxidation by MDA in orange-fleshed sweet potatoes. The treatments are represented by the points, where F = flesh; P = peel and FP = flesh + peel
In the three genotypes of purple sweet potatoes, the presence of high levels of total phenolic compounds and total flavonoids in the peels is clear (Figure 1 and Table 2), with emphasis on the phenols content in 'JN RX1' (324.78 mg/100g) and flavonoids in 'JN RX2' (160.17 mg/100g). However, these genotypes showed the lowest levels of phenolic compounds (162.50 to 177.88 mg/100g). In this study, it is worth to point out that purple sweet potatoes contain higher contents of phenolic compounds, including flavonoids, compared to the orange-fleshed ones. Other studies have also demonstrated higher levels of phenolic compounds in purple-fleshed sweet potatoes (0.949 mg/g), compared to white-fleshed (0.003 mg/g) (Teow et al., 2007).
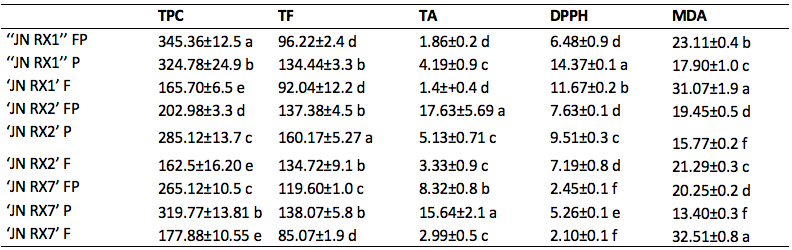
* Means followed by the same letter, in the column, do not differ statistically among themselves by the Sckott-Knott test (p≤ 0.05).** FP= flesh + peel; P = peel and F = flesh.
Purple sweet-potatoes containing abundant anthocyanins and several studies demonstrate their diverse biological activities, including scavenging reactive species (Cartier et al., 2017). 'JN RX1' (Table 2), which exhibits an intense purple color in the flesh, contains higher levels of anthocyanins in the peel (4.19 mg/100g), when compared to the other analyzed parts (Figure 1). However, when the comparison occurs between the genotypes, both 'JN RX2' and 'JN RX7' have higher levels of anthocyanins compared to 'JN RX1'. Anthocyanins are considered the most important phenolic group for the coloring of purple sweet potatoes (Salawu et al., 2015). Certainly, this flavonoid influenced the antioxidant activity, both by DPPH or MDA in ‘JN RX1’ and ‘JN RX7’, since phenolic compounds contribute to the ability to eliminate free radicals (Cartier et al., 2017). These results allow us to affirm that sweet potato peels, usually discarded by the consumer, might be used as a source of important bioactive agents - increasing the functional value, and may be an option for the food industry in the elaboration of by-products.
The principal component analysis was also applied to the purple sweet potato data. The two main components explain 83.63% of the data variance (Figure 4) and the results showed that there was no grouping between the genotypes in relation to the analyzed antioxidant compounds. In contrast, the fleshes were grouped in PC2- and this grouping was influenced by antioxidant activities, measured via MDA and DPPH. The hierarchical cluster analysis (HCA) also demonstrates the grouping of 'JN RX2' and 'JN RX7' fleshes (Figure 5) and it is worth noting that both have the lowest levels of total phenolic compounds, reflecting the low antioxidant activity measured by DPPH and high by MDA. The peels of all purple flesh genotypes showed the highest values of phenols, flavonoids and anthocyanins and were grouped in PC1 +, with emphasis on 'JN RX 1' and 'JN RX 2', which show high similarity.
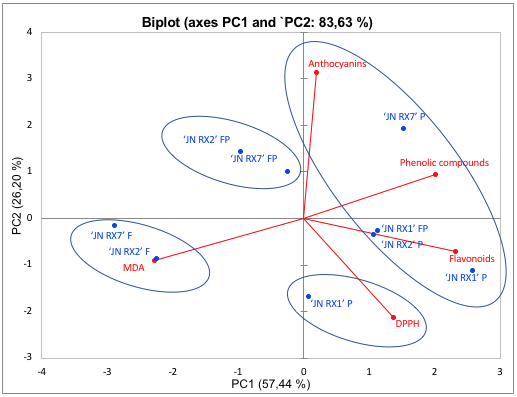
FIGURE 4
Two-dimensional projection and scores of biochemical characteristics, total phenolic compounds, total flavonoids, total anthocyanins, antioxidant activity by DPPH and lipid peroxidation by MDA in purple-fleshed sweet potatoes. The treatments are represented by the points, where F = flesh; P = peel and FP = flesh + peel
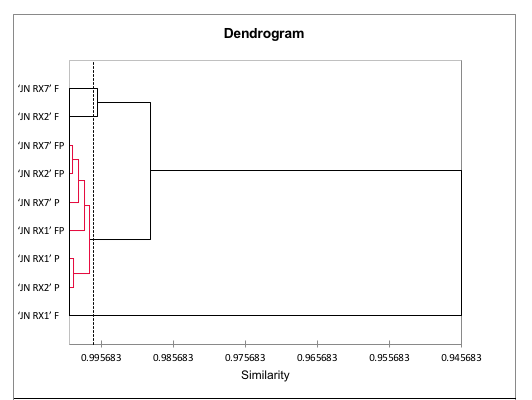
FIGURE 5
Dendrogram of biochemical characteristics, total phenolic compounds, total flavonoids, total anthocyanins, antioxidant activity by DPPH and lipid peroxidation by MDA in orange-fleshed sweet potatoes. The treatments are represented by the points, where F = flesh; P = peel and FP = flesh + peel
Aiming at a physicochemical characterization of the orange and purple sweet potatoes, the tubers were cooked, which is the preferred form of consumption, and the physicochemical attributes were characterized (Table 3). There was an increase in pH in 'BRS Amélia' after cooking in water. In purple-fleshed sweet potatoes, there was a decrease in pH in ‘JN RX1’ and ‘JN RX2’ after thermal processing. The pH values found in the orange and purple genotypes cooked in water were higher than 6.0, however a lower pH (5.5) was detected in raw 'BRS Amélia'. pH values below 5.5 may indicate low quality, due to oxidation processes (De Oliveira et al., 2019). All genotypes (orange flesh and purple flesh) showed decreased titratable acidity in response to cooking, except for the purple-fleshed sweet potato 'JN RX2', which may be attributed to losses of organic acids to the cooking water (Ogliari et al., 2020), improving the taste and consequently increasing consumer acceptance. The titratable acidity contents detected in our study were lower than those described for other sweet potato genotypes (De Oliveira et al., 2019) and can be attributed to cultivation factors, including abiotic factors, such as climate and fertilization.
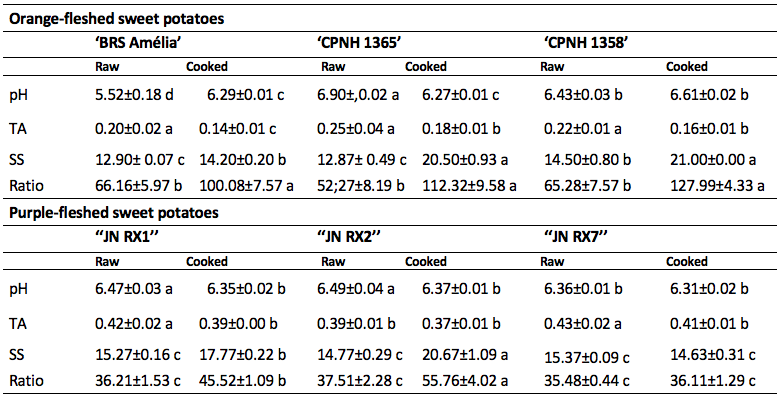
* Means followed by the same letter, in the line, do not differ statistically among themselves by the Sckott-Knott test (p≤ 0.05).
Cooking induced an increase in the content of soluble solids in all genotypes, except for the purple-fleshed sweet potato 'JN RX7' (Table 3). This increase may be due to the ease of extraction due to the rupture of the cell wall by the action of temperature (Borges et al., 2019; Lima et al., 2017). The increase in SS is important for consumer acceptance, due to the more sweet taste. Comparing the ratio between SS and TA (ratio), the orange genotypes show higher values, compared to the purple sweet potatoes, regardless of thermal processing. This result is very interesting, since the presence of sugars and acids influences the taste and the acceptance of the product (Jayasena and Cameron, 2008). It is worth mentioning that cooking in water promoted an increase in the ratio in all genotypes, except 'JN RX7', which was a result influenced by the absence of significant variations in the content of soluble solids after the action of high temperature.
Principal component analysis was performed to verify the physicochemical characteristics after thermal processes (boiling) in the genotypes studied (Figures 6 e 7). In relation to orange-fleshed sweet potatoes (Figure 6), PC1 represents 61.28% of the total data variance, grouping the genotypes according to the thermal processing. PC1+ grouped the cooked genotypes ‘CPNH 1365’ e ‘CPNH 1358’, which showed the higher SS and ratio (Table 3) than raw orange-fleshed sweet potatoes (PC1+ and PC2+). These raw genotypes also presented the highest levels of carotenoids (Table 1), an potent free radical scavenger, which are also related with inhibit the development of cardiovascular diseases and mitigate the risk of metabolic diseases, such as diabetes (Kulczyński et al., 2017). In this way, ‘CPNH 1365’ show good characteristics physicochemical and antioxidants, which make them new genotypes interesting for consumption.
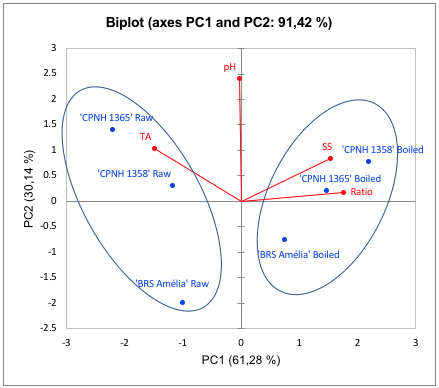
FIGURE 6
Two-dimensional projection and scores of physicochemical biochemical characteristics pH, soluble solids (°Brix), titratable acidity (% citric acid) and ratio (SS / TA), in raw and cooked orange-fleshed sweet potatoes
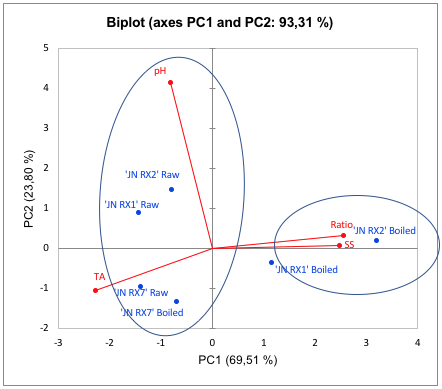
FIGURE 7
Two-dimensional projection and scores of physicochemical biochemical characteristics pH, soluble solids (°Brix), titratable acidity (% citric acid) and ratio (SS / TA), in raw and cooked purple-fleshed sweet potatoes
The principal component analysis applied to the physicochemical analyses of purple-fleshed sweet potatoes explained 93.31% of analyzed data (Figure 7). PC1 explained 69.51% of the evaluated parameters. PC1+ grouped both boiled genotypes ‘JN RX1’ and ‘JN RX2’ with the highest content of SS and ratio, which have also higher pH (PC1-) compared with ‘JN RX7’. Although raw ‘JN RX1’ show the highest antioxidant activity measured by DPPH radical, it present lower levels of anthocyanin (Table 2) than ‘JN RX7’. It is worth mentioning the levels of SS, ratio and antioxidants in ‘JN RX2’, which may be an indication of the quality of this genotype for consumption.
CONCLUSION
The bioactive compounds found in sweet potatoes are genotype-dependent and the color of the tubers is an interesting phenotypic characteristic for indicating the class and content of these phytochemicals in the different genotypes. In general, genotypes with orange coloration 'BRS Amélia', 'CPNH 1365' and 'CPNH 1358' are potential sources of carotenoids and the more intense the color of the tuber (i.e., 'CPNH 1358'), the higher the levels of these provitamins A. Purple-fleshed sweet potatoes' JN RX1 ',' JN RX2 'and JN RX7', however, have high levels of phenolic compounds such as flavonoids (i.e., anthocyanins) and greater antioxidant capacity, mainly the peels, where the highest levels of these bioactives compunds are found. The orange sweet potato 'CPNH 1365' and the purple sweet potato 'JN RX2' have a higher content of bioactive compounds and high SS levels, emphasizing the potential of these genotypes for selection and their use as high quality food and source of compounds that promote human health. It should be noted that colored flesh sweet potatoes are potential sources of bioactive compounds, demonstrating the importance of expanding the use and enhancement of vegetables for use in the food and pharmaceutical industries, especially the peels, where the greatest content of these compounds are verified.
Acknowledgments
The authors gratefully acknowledge the financial support of Sao Paulo Research Foundation (FAPESP) for financial support of this work (grant number 2016/22665-2 and 2017/08032-0) and for scholarship provided by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) to Letícia Silva Pereira Basílio (grant number 88882.433049/2019-01) and by Sao Paulo Research Foundation (FAPESP) to M.A.F. Belin (grant number 2019/26996-1) and National Council for Scientific and Technological Development (CNPq, Brazil) (grant number 307571/2019-0).
Literature
Alam, M.K., Sams, S., Rana, Z.H., Akhtaruzzaman, M., Islam, S.N. 2020. Minerals, vitamin C, and effect of thermal processing on carotenoids composition in nine varieties orange-fleshed sweet potato (Ipomoea batatas L.). J. Food Compos. Anal. 92, 103582. https://doi.org/10.1016/j.jfca.2020.103582
Borges, C.V., Minatel, I.O., Gomez-Gomez, H.A., Lima, G.P.P. 2017. Medicinal plants: Influence of environmental factors on the content of secondary metabolites. Med. Plants Environ. Challenges 259–277. https://doi.org/10.1007/978-3-319-68717-9_15
Borges, C. V., Minatel, I.O., Amorim, E.P., Belin, M.A.F., Gomez-Gomez, H.A., Correa, C.R., Lima, G.P.P. 2019. Ripening and cooking processes influence the carotenoid content in bananas and plantains (Musa spp.). Food Res. Int. 124, 129–136. https://doi.org/10.1016/j.foodres.2018.08.022
Brand-Williams, W., Cuvelier, M.E., Berset, C. 1995. Use of a free radical method to evaluate antioxidant activity. LWT - Food Sci. Technol. 28, 25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Cartier, A., Woods, J., Sismour, E., Allen, J., Ford, E., Githinji, L., Xu, Y. 2017. Physiochemical, nutritional and antioxidant properties of fourteen Virginia-grown sweet potato varieties. J. Food Meas. Charact. 11, 1333–1341. https://doi.org/10.1007/s11694-017-9511-8
De Albuquerque, T.M.R., Sampaio, K.B., de Souza, E.L. 2019. Sweet potato roots: Unrevealing an old food as a source of health promoting bioactive compounds – A review. Trends Food Sci. Technol. 85, 277–286. https://doi.org/10.1016/j.tifs.2018.11.006
De Oliveira, A.F., Soares, J.M., Da Silva, É.C., Filho, P.S.L., Candido, C.J., Do Amaral, L.A., Dos Santos, E.F., De Resende, J.T.V., Schwarz, K., Novello, D. 2019. Evaluation of the chemical, physical and nutritional composition and sensory acceptability of different sweet potato cultivars. Semin. Agrar. 40, 1127–1137. https://doi.org/10.5433/1679-0359.2019v40n3p1127
Diamante, M.S., Vanz Borges, C., Minatel, I.O., Jacomino, A.P., Basílio, L.S.P., Monteiro, G.C., Corrêa, C.R., de Oliveira, R.A., Pace Pereira Lima, G. 2020. Domestic cooking practices influence the carotenoid and tocopherol content in colored cauliflower. Food Chem. 340, 127901. https://doi.org/10.1016/j.foodchem.2020.127901
Giusti, M.M., Wrolstad, R.E. 2001. Characterization and Measurement of Anthocyanins by UV ‐ Visible Spectroscopy. Curr. Protoc. Food Anal. Chem. 0–13. https://doi.org/10.1002/0471142913.faf0102s00
Heath, R.L., Packer, L. 1968. Photoperoxidation in isolated chloroplasts. Arch. Biochem. Biophys. 125, 189–198. https://doi.org/10.1016/0003-9861(68)90654-1
IAL. 2008. Óleos E Gorduras, in: Odair Zenebon, N.S.P. e P.T. (Ed.), Métodos Físicos-Quimicos para análise de alimentos. São Paulo, pp. 589–625. https://doi.org/10.1017/CBO9781107415324.004
Islam, S.N., Nusrat, T., Begum, P., Ahsan, M. 2016. Carotenoids and β-carotene in orange fleshed sweet potato: A possible solution to Vitamin A deficiency. Food Chem. 199, 628–631. https://doi.org/10.1016/j.foodchem.2015.12.057
Jayasena, V., Cameron, I. 2008. °brix/Acid ratio as a predictor of consumer acceptability of crimson seedless table grapes. J. Food Qual. 31, 736–750. https://doi.org/10.1111/j.1745-4557.2008.00231.x
Jung, J.K., Lee, S.U., Kozukue, N., Levin, C.E., Friedman, M. 2011. Distribution of phenolic compounds and antioxidative activities in parts of sweet potato (Ipomoea batata L.) plants and in home processed roots. J. Food Compos. Anal. 24, 29–37. https://doi.org/10.1016/j.jfca.2010.03.025
Kulczyński, B., Gramza-Michałowska, A., Kobus-Cisowska, J., Kmiecik, D. 2017. The role of carotenoids in the prevention and treatment of cardiovascular disease – Current state of knowledge. J. Funct. Foods 38, 45–65. https://doi.org/10.1016/j.jff.2017.09.001
Lichtenthaler, H.K. 1987. [34] Chlorophylls and carotenoids: pigments of photosynthetic biomembranes, in: Methods in Enzymology. Elsevier, pp. 350–382.
Lima, G.P.P., Costa, S.M., Monaco, K. de A., Uliana, M.R., Fernandez, R.M., Correa, C.R., Vianello, F., Cisneros-Zevallos, L., Minatel, I.O. 2017. Cooking processes increase bioactive compounds in organic and conventional green beans. Int. J. Food Sci. Nutr. 68, 919–930. https://doi.org/10.1080/09637486.2017.1324563
Lima, G.P.P., Vianello, F., Corrêa, C.R., Campos, R.A. da S., Borguini, M.G. 2014. Polyphenols in Fruits and Vegetables and Its Effect on Human Health. Food Nutr. Sci. 05, 1065–1082. https://doi.org/10.4236/fns.2014.511117
Maoka, T. 2020. Carotenoids as natural functional pigments. J. Nat. Med. 74. https://doi.org/10.1007/s11418-019-01364-x
Martins, N., Ferreira, I.C.F.R. 2017. Wastes and by-products: Upcoming sources of carotenoids for biotechnological purposes and health-related applications. Trends Food Sci. Technol. 62, 33–48. https://doi.org/10.1016/j.tifs.2017.01.014
Minatel, I.O., Borges, C.V., Alonzo, H., Hector, G., Gomez, Chen, C.O., Pace, G., Lima, P. 2017. Phenolic Compounds: Functional Properties , Impact of Processing and Bioavailability, in: InTech (Ed.), Phenolic Compounds - Biological Activity. London, United Kingdom, pp. 1–24. https://doi.org/10.5772/66368
Ogliari, R., Soares, J.M., Teixeira, F., Aparecida, K., Schiessel, D.L., Novello, D. 2020. Chemical, nutritional and sensory characterization of sweet potato submitted to different cooking methods. Int. J. Res. 8, 147–156. https://doi.org/https://doi.org/10.29121/granthaalayah.v8.i10.2020.1881
Pekal, A., Pyrzynska, K. 2014. Evaluation of Aluminium Complexation Reaction for Flavonoid Content Assay. Food Anal. Methods 7, 1776–1782. https://doi.org/10.1007/s12161-014-9814-x
Sagar, N.A., Pareek, S., Sharma, S., Yahia, E.M., Lobo, M.G. 2018. Fruit and Vegetable Waste: Bioactive Compounds, Their Extraction, and Possible Utilization. Compr. Rev. Food Sci. Food Saf. 17, 512–531. https://doi.org/10.1111/1541-4337.12330
Salawu, S.O., Udi, E., Akindahunsi, A.A., Boligon, A.A., Athayde, M.L. 2015. Antioxidant potential, phenolic profile and nutrient composition of flesh and peels from Nigerian white and purple skinned sweet potato (Ipomea batatas L.). Pelagia Res. Libr. Asian J. Plant Sci. Res. 5, 14–23.
Simões, A. do N., de Almeida, S.L., Borges, C.V., Fonseca, K.S., Barros Júnior, A.P., de Albuquerque, J.R.T., Corrêa, C.R., Minatel, I.O., Morais, M.A. dos S., Diamante, M.S., Lima, G.P.P. 2020. Delaying the harvest induces bioactive compounds and maintains the quality of sweet potatoes. J. Food Biochem. 44, 1–13. https://doi.org/10.1111/jfbc.13322
Singleton, V., Rossi, J.R. 1965. Determination of tannins in wines. J. Enol. Vitic. 6, 114.
Teow, C.C., Truong, V. Den, McFeeters, R.F., Thompson, R.L., Pecota, K. V., Yencho, G.C. 2007. Antioxidant activities, phenolic and β-carotene contents of sweet potato genotypes with varying flesh colours. Food Chem. 103, 829–838. https://doi.org/10.1016/j.foodchem.2006.09.033
Van Jaarsveld, P.J., Faber, M., Tanumihardjo, S.A., Nestel, P., Lombard, C.J., Benadé, A.J.S. 2005. β-carotene-rich orange-fleshed sweet potato improves the vitamin A status of primary school children assessed with the modified-relative-dose- response test. Am. J. Clin. Nutr. 81, 1080–1087. https://doi.org/10.1093/ajcn/81.5.1080
Wang, A., Li, R., Ren, L., Gao, X., Zhang, Y., Ma, Z., Ma, D., Luo, Y. 2018. A comparative metabolomics study of flavonoids in sweet potato with different flesh colors (Ipomoea batatas (L.) Lam). Food Chem. 260, 124–134. https://doi.org/10.1016/j.foodchem.2018.03.125
Zhishen, Jia, Mengcheng, Tang, Jianming, W. 1999. The determiantion of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 64, 555–559.
Author notes