Reportes Verduras
Cultivation seasons change the antioxidant capacity and physicochemical quality of ‘Vanda’ lettuce
Las fechas de cultivo cambian la capacidad antioxidante y la calidad fisicoquímica de la lechuga 'Vanda'
Cultivation seasons change the antioxidant capacity and physicochemical quality of ‘Vanda’ lettuce
Revista Iberoamericana de Tecnología Postcosecha, vol. 22, núm. 1, 2021
Asociación Iberoamericana de Tecnología Postcosecha, S.C.
Recepción: 15 Abril 2021
Aprobación: 05 Junio 2021
Publicación: 30 Junio 2021
Resumen: La lechuga se consume ampliamente y tiene una cantidad aceptable de compuestos con actividad antioxidante, que puede variar debido a factores bióticos y abióticos. El objetivo de este estudio fue investigar si la temporada de cultivo es responsable de los cambios en la actividad antioxidante y la calidad fisicoquímica y fisiológica de la lechuga 'Vanda' almacenada en frío. Las cabezas de lechuga se cosecharon en verano e invierno y se mantuvieron almacenadas a 0 °C y 95-97% de humedad relativa durante 28 días, con evaluaciones semanales. El diseño experimental fue completamente al azar en un diseño factorial 2 x 5 (temporadas de cultivo x días de análisis). Los datos se presentaron como Análisis de Varianza Multivariante (MANOVA), Análisis de Correlación (CA), Análisis de Componentes Principales (PCA) (gráfico biplot) y Análisis de Conglomerados Jerárquicos (HCA). La lechuga cultivada durante la temporada de verano presentó mayor actividad antioxidante, mayor contenido de compuestos fenólicos y color más claro. Todos los atributos cualitativos se perdieron durante el almacenamiento, pero la lechuga cultivada en verano tuvo una mayor actividad antioxidante y mejor calidad fisicoquímica durante 21 días, mientras que la lechuga cultivada en invierno tuvo una pérdida importante desde el séptimo día de almacenamiento. Por lo tanto, los factores ambientales influyen en los atributos cualitativos y definen la mejor temporada de crecimiento, mientras que el almacenamiento en frío solo mantiene estos atributos y proporciona una larga vida útil. Se necesitan más estudios para verificar si este hallazgo también ocurre con otras variedades de lechuga, al igual que en otras partes del mundo.
Palabras clave: Lactuca sativa L., análisis multivariado, fisiología vegetal, verano, invierno.
Abstract: Lettuce is widely consumed and has an acceptable amount of compounds with antioxidant activity, which may vary due to biotic and abiotic factors. The aim of this study was to investigate whether the cultivation season is responsible for changes in the antioxidant activity and the physicochemical and physiological quality of cold stored 'Vanda' lettuce. Heads of lettuce were harvested in summer and winter and kept stored at 0°C and 95-97% RH for 28 days, with weekly evaluations. The experimental design was completely randomized in a factorial 2 x 5 design (growing seasons x days of analysis). Data were submitted Multivariate Analysis of Variance (MANOVA), Correlation Analysis (CA), Principal Component Analysis (PCA) (biplot graph) and Hierarchical Cluster Analysis (HCA). The lettuce cultivated during the summer season presented higher antioxidant activity, higher content of phenolic compounds and lighter color. All the qualitative attributes were lost during storage, but the summer-cultivated lettuce had a higher antioxidant activity and better physicochemical quality for 21 days, whereas winter-cultivated lettuce had major loss since the seventh day of storage. Therefore, environmental factors has an affect on the qualitative attributes and define the best growing season, while cold storage only maintains these attributes and provides long shelf-life. More studies are needed to verify if this found also occurs to other varieties of lettuce, just as in other parts of the world.
Keywords: Lactuca sativa L., multivariate analyse, plant physiology, summer, winter.
INTRODUCTION
Lettuce (Lactuca sativa L.) is the most widespread leafy vegetable in Brazil and in the world, being also the most consumed in natura (Henz and Suinaga, 2009). The high consumption is related to low acquisition cost, mild flavor, nutritional composition and the elevated offer, which is due to the ease of cultivation during the whole year, with varieties suited to the climate of each region (Costa and Sala, 2005).
Given the high consumption of lettuce in natura, its nutritional quality is of particular interest for human diet. Its ingestion provides vitamins A, C and E, minerals, such as calcium and iron and, especially, antioxidant compounds. These compounds are related to the prevention of carcinogenic and cardiovascular diseases, inhibiting oxidative stress caused by free radicals. The main compounds with antioxidant activity found in green lettuce are chlorophylls, carotenoids, folic acid, caffeic acid and phenolic compounds (Caldwell, 2003; Ferreres et al., 1997; Hooper and Cassidy, 2006; Perucka and Olszowka, 2011; Serafini et al., 2002).
The compounds with antioxidant activity are secondary metabolites synthesized during the plant’s growth and development, which biosynthesis, as well as the quality of lettuce, may be influenced by abiotic and biotic factors, such as solar radiation, temperature, moisture, injury and attacks by pathogens (Dixon and Paiva, 1995; Ferreres et al., 1997; Llorach et al., 2008; Romani et al., 2000; Zhao et al., 2007; Zhao, Iwamoto and Carey, 2007). The influence of the environment of cultivation in the quality of lettuce, comparing the vegetable produced in the field with the one produced in the greenhouse, has been previously studied (Krizek, Britz and Mirecki, 1998; Romani et al., 2002; Wells, 1996). However, studies on the environmental influence in terms of season of cultivation of this vegetable are lacking.
The technologies used in the postharvest also interfere in the antioxidant activity of lettuce. This vegetable has a short shelf-life and the cold storage is the main technique used for the maintenance of its physicochemical quality and the increase of its offer. Physiological studies, especially those related to the respiratory activity and ethylene production, are important because they become the main processes to be controlled by cooling.
In Brazil, the main types of lettuce grown in order of economic importance are Grand Rapids types, crisphead, butterhead and romaine. Since the last decade, the Grand Rapids type lettuce dominates more than 50% of the domestic market, unique event in the world market of lettuce. Among the green cultivars of Grand Rapids type, the ‘Vanda’ is the most commercialized and also the most preferred by growers, given the rapid cycle, mildew and mosaic virus resistance, great size, clear color and good consumer acceptance (Sala and Costa, 2012).
In this sense, the goal of the present study was to investigate if the cultivation season is responsible for changes in the antioxidant activity and in the physicochemical and physiological quality of the lettuce 'Vanda' stored under refrigeration.
MATERIAL AND METHODS
Acquisition of the raw material
Heads of lettuce 'Vanda' were purchased from a local grower. The lettuce were grown in open and conventional system in the city of Piracicaba, SP, Brazil (22° 43' 30" S, 47° 38' 56" W). The choice of this variety also was due to its hardiness of cultivation, which allowed their planting in all the seasons. For this study, two growing seasons were evaluated: winter and summer. The summer cultivation was planted in November 2009 and harvested in January 2010 (50 days of cultivation cycle), and the winter planting was held in May with the harvest in July the same year, 2010 (70 days of cultivation cycle).
During the summer cultivation, the average minimum temperature was 20.0 °C and the maximum was 30.5 °C, with mean relative humidity and solar radiation of 79% and 459.30 Ly day-1, respectively. In the winter cultivation, the average minimum temperature was 11.9°C, with the maximum of 26.1°C, mean relative humidity was 72% and mean solar radiation was 342.6 Ly day-1.
The growing technologies (plant in tray, transplanted, fertilization and weed and pest control) used were the same for both seasons. Lettuce cultivation was irrigated and the harvest carried out manually at the time the leaves got tender and without evidence of tasseling. After harvesting, the heads of lettuce were transported in plastic boxes, without cooling, to the laboratory of the University of São Paulo, also in Piracicaba, within an hour.
Experiment
As soon as the heads of lettuce arrived at the laboratory, in both periods of harvesting, a selection was made regarding size, color, absence of damage, insects and other potential contaminants, to achieve as much homogeneity of the lot as possible. All heads were sanitized through immersion in solutions of 100 mg L-1 of active chlorine for 10 min. For drying, the heads were placed on benches lined with absorbent paper, right after being stored in the chamber at 0 °C and 95-97% RH, for 28 days.
Analyses were carried out immediately after receiving the lettuce (day 0) and at days 7, 14, 21 and 28. The experimental design was completely randomized, in a factorial 2 x 5 design (seasons of cultivation x days of analysis). For the variables related to ethylene production and respiratory activity, four repetition of one head each were used, while for the other analyses, four repetitions composed of three heads each were employed. Biochemical analyses were carried out in triplicate.
Ethylene production and respiratory activity
The heads of lettuce were placed in 5.0 L glass bottles, which remained hermetically sealed for 2 hours, in the same storage conditions. Samples of 0.5 mL of the internal atmosphere of the flasks were collected through a silicone septum present on the caps of the flasks and measured by a gas chromatograph (Thermo Electron Corporation, MA, USA, model Trace GC Ultra), equipped with two flame ionization detectors (FID), two injectors, two nitrogen columns and a methanator at 350 °C. Results were determined considering the chromatographic values, the mass of the lettuce heads, the volume of the flasks and the time they remained closed. Ethylene production was expressed in µL C2H4kg-1.-1 and respiratory activity in mg C02kg-1h-1.
Physicochemical and biochemical analyses
The color of the leaves was determined by the parameters L, a* and b*, using a colorimeter (Konica Minolta Inc., Japan, model CR-400), evaluating 4 leaves of each head of the replicate, for 4 times. Total chlorophyll (Chl) was determined according to Lichtenthaler (1987), with the addition of 5 mL of 80% acetone at 4°C to 0.5 g of mashed samples and centrifugation for 15 min at 12298 RCF and 4°C (Clinical Centrifuge, IEC). The absorbance was measured at 645, 663 and 470 nm in a spectrophotometer (Biochrom Libra S22) and the results were expressed in mg g-1. Polyphenol oxidase activity (PPO) was determined according to Matsuno and Uritani (1972), in which 5 mL of potassium phosphate buffer 0.05 M (pH 6.0) were added to 0.5 g of mashed samples and the homogenate was centrifuged for 10 min at 12298 RCF and 4°C. PPO was determined by the addition of 0.85 mL of 0.1 M catechol and incubation in a water bath (Tecnal, TE-0541) for 30 min at 30°C, following addition of 0.7 mL of 5% sulfuric acid. The absorbance was measured at 395 nm and the activity was expressed in AU min-1g-1. The determination of phenolic compounds (PhC) was performed according to Singleton, Orthofer and Lamuela-Raventos (1999), using 80% ethanol as extractor, ultrasonic bath (Unique, USC-1400A), centrifugation for 15 min at 5000 RCF, addition of 0.5 ml of 10% Folin-Ciocalteu and 2.0 mL of 4% sodium carbonate, absorbance read at 740 nm. Gallic acid was used as standard, and the results were expressed in mg GAE g-1. Free radical scavenging activity of 2,2-diphenyl-1-picrylhydrazyl (DPPH) was performed according to Mensor et al. (2001) and had the same extraction of PhC, with the addition of 3 mL of ethanol and 0.03 mL of 0.5 mM DPPH to 0.5 mL of the extract . The absorbance was measured at 575 nm and the results, expressed as µmol TEAC g-1. The autoxidation of β-carotene system/linoleic acid (Beta) was assessed using BHT and α-tocopherol as standards (Emmons, Peterson and Paul, 1999), with the addition of 40 mg of linoleic acid and 400 mg of Tween® 40 to 3 mL of a β-carotene/chloroform solution (10 mg of β-carotene in 100 mL of chloroform) in order to obtain the emulsion. 3 mL of the emulsion were added to 0.05 mL of the extract (same as the PhC) and the absorbance was measured at 470 nm at time (0) and after 2 hours of incubation, with results expressed as % of relative inhibition to the control.
Statistical analyses
Multivariate analyses were conducted on the data through Multivariate Analysis of Variance (MANOVA), Correlation Analysis (CA), Principal Component Analysis (PCA) (biplot graph) and Hierarchical Cluster Analysis (HCA). MANOVA was performed to compare the vectors of average, defining the significance among the observations. For this purpose, normality and homogeneity of variance tests were performed for all the variables studied. CA was performed to evaluate the interdependence of the variables, calculating the correlation matrix. PCA was employed to characterize and establish the relationship and similarity between the two growing seasons and the five periods (days of analyses). For HCA, the grouping technique was given through the single linkage cluster with method Euclidean distance, cutting at 0.40. The statistical software used to perform all the tests was the SAS 9.3 (SAS Institute, Cary, NC, USA, 2010).
RESULTS AND DISCUSSION
Multivariate Analysis of Variance
The MANOVA test is a rigorous statistical analysis and was performed to test the null hypothesis, which indicates that there is no significant pairwise difference between any of the growing seasons and periods evaluated (Table 1). The null hypothesis was rejected for almost all of the pairwise comparisons, being valid only for VS21 x VS28, VW7 x VW14, VW14 x VW21, VW14 x VW28 and VW21 x VW28. This test showed that (1) the growing season of lettuce indeed influenced the postharvest attributes; and (2) these attributes were changed during the cold storage period. This means that ‘Vanda’ lettuces cultivated on summer showed changes on their qualitative atributes until the 21st day, while the attributes of winter lettuces didn't show any changes from the 7th day. The total group of data and the differences between the treatments and variables are presented in items 3.2 (CA) and 3.3 (PCA).
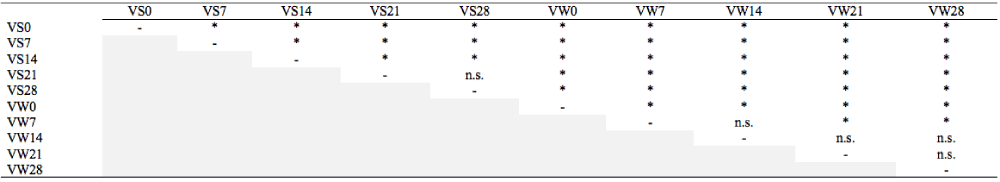
VS0 = Vanda harvested in summer, day 0; VS7 = Vanda harvested in summer, day 7; VS14 = Vanda harvested in summer, day 14; VS21 = Vanda harvested in summer, day 21; VS28 = Vanda harvested in summer, day 28; VW0 = Vanda harvested in winter, day 0; VW7 = Vanda harvested in winter, day 7; VW14 = Vanda harvested in winter, day 14; VW21 = Vanda harvested in winter, day 21; VW28 = Vanda harvested in winter, day 28. *= significantly different by Wilks's lambda test (P<0.05), n.s.= not significantly different by Wilks's lambda test (P<0.05).
Correlation Analisis
To evaluate the correlation between the physicochemical, physiological and biochemical variables, we calculated the correlation matrix and selected the coefficients equal to or above 0.70, which are considered significant according to Sounis (1985) (Table 2). This way, we observed that all the selected variable pairwise had high and positive coefficients, hence the variables are directly proportional. As an example, the variable PhC, which formed the greatest number of pairwise numbers, was correlated with DPPH, value a*, Chl, value L and PPO variables, indicating that when PhC has a high value, the other variables will also do and vice-versa. Due to the high correlation coefficients obtained, the application of PCA and HCA to better characterize the lettuce produced at different seasons was justified.

PhC: Total phenolic content; DPPH: Antioxidant capacity by the free radical scavenging activity DPPH; Chl: Quantity of chlorophyll; L*: Lightness; PPO: Polyphenol oxidase activity; C2H4: Ethylene production; CO2: Respiratory activity.
Principal Component and Hierarchical Cluster Analyses
The number of principal components (PC) used is the one that accumulates 70% or more of the total variance (Larrigaudiere et al., 2004). In this work, two PC were taken from the total group of data (Table 3), explaining 78.68% of the variance.
SD= Standard Deviation; VW= Vanda harvested in winter; VS= Vanda harvested in summer.
The PCA is represented by the projection of the variables (Figure 1A) and observations (Figure 1B). The first principal component (PC1) explained 56.32% of the statistical variance and was characterized by green color and antioxidant activity. PC1 is positively correlated with variables PhC, value a*, DPPH and Chl. The second principal component (PC2) explained 22.37% of the statistical variance and was characterized by the physiological quality. PC2 is negatively correlated with the variables ethylene production and respiratory rate.
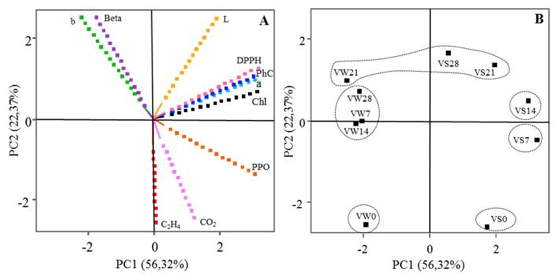
Figure 1
Principal Component Analysis, using the postharvest parameters of lettuce ‘Vanda’: projections of variables (A) and observations (B).
(A) PC= Principal Component. Variables: b = value b* of color, Beta = antioxidant activity using the β-carotene/linoleic acid system, L* = value of Lightness, DPPH = Antioxidant capacity by the free radical scavenging activity DPPH, PhC = Total phenolic Content, a = value a* of color, Chl = Quantity of chlorophyll, PPO = Polyphenol Oxidase Activity, CO2 = Respiratory activity, C2H4 = Ethylene production. (B) Observations: VS0 = Vanda harvested in summer, day 0; VS7 = Vanda harvested in summer, day 7; VS14 = Vanda harvested in summer, day 14; VS21 = Vanda harvested in summer, day 21; VS28 = Vanda harvested in summer, day 28; VW0 = Vanda harvested in winter, day 0; VW7 = Vanda harvested in winter, day 7; VW14 = Vanda harvested in winter, day 14; VW21 = Vanda harvested in winter, day 21; VW28 = Vanda harvested in winter, day 28.
In the HCA, the observations could be broken into 6 groups by a linkage distance around 0.40, viewed in the dotted line (Figure 2). The same break was used for the separation of groups (dotted circles) shown in the projection of the observations (Figure 1B).
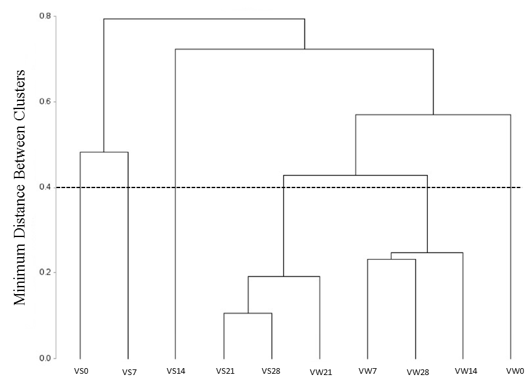
Figure 2
Dendrogram constructed based on the postharvest variables profiles of lettuce ‘Vanda’.
VS0 = Vanda harvested in summer, day 0; VS7 = Vanda harvested in summer, day 7; VS14 = Vanda harvested in summer, day 14; VS21 = Vanda harvested in summer, day 21; VS28 = Vanda harvested in summer, day 28; VW0 = Vanda harvested in winter, day 0; VW7 = Vanda harvested in winter, day 7; VW14 = Vanda harvested in winter, day 14; VW21 = Vanda harvested in winter, day 21; VW28 = Vanda harvested in winter, day 28.
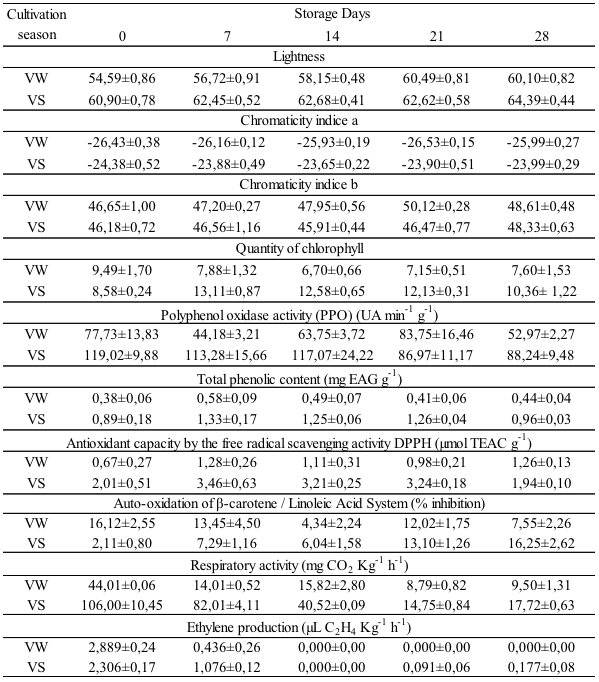
The observations regarding lettuce VS stored for 0, 7 and 14 days (VS0, VS7 and VS14) were individually separated in HCA because they had characteristics that differed among themselves and also between them and the others. These three groups showed medium to high antioxidant capacity and fresh green color, corresponding to fresh lettuces. However, VS0 had the highest physiological activity (106.0 mg CO2Kg-1h-1 and 2.306 µL C2H4Kg-1h-1) and VS14 the least (40.52 mg CO2Kg-1h-1 and 0.0 µL C2H4Kg-1h-1), while VS7 presented intermediate values of ethylene production and respiratory rate (82.01 mg CO2Kg-1h-1 and 1.076 µL C2H4Kg-1h-1), denoting a decrease in the metabolism during the cold storage period. Although the variables PPO, values L and b* and Beta did not enter in both PC, their projections indicated that VS0, VS7 and VS14 showed high PPO (119.02, 113.28 and 117.07 UA min-1g-1, respectively) and low value b* (46.18, 46.56 and 45.91, respectively) and Beta (2.11, 7.29 and 6.04 % inhibition).
An individual separation also occurred for the observation of 'Vanda' lettuce harvested in winter on day 0 (VW0). This was characterized by low antioxidant capacity, a darker green color than VS (low values a* and L) and high physiological activity, since these lettuce heads had not been refrigerated yet. Unlike VS, VW showed intermediate PPO (77.73 UA min-1g-1) in the harvest day (day 0).
In the same way as VW0, the group formed by VW7, VW14 and VW28 was characterized by low antioxidant capacity and dark green color. Moreover, this group presented medium to high values of b* color (47.20, 47.95 and 50.12, respectively), indicating yellowing leaves during storage, or in other words, the senescence of the lettuce. Concerning physiological activity, the group didn’t present the same behavior as VW0. The VW7 (14.01 mg CO2Kg-1h-1) and VW14 (15.82 mg CO2Kg-1h-1) had intermediate values, while VW28 (9.50 mg CO2Kg-1h-1) had low ones, which is due to the cold and senescence.
Even though the observation VW21 presents the same characteristics of the previous group in relation to antioxidant capacity, color, PPO and Beta, it formed a new group with VS21 and VS28. The three observations of the second group showed at least one important feature in common: low physiological activity, particularly the ethylene production, which was more relevant in PC2 (0.62) than the respiration rate (0.49).
In addition to the physiological activity, VS21 and VS28 are characterized by high antioxidant capacity, average values of PPO and high values L. VS21 presented a medium Beta and value b*, whereas in VS28 these values were high, indicating lettuce senescence. These results confirm the observation in MANOVA, wherein VS21 and VS28 had no difference between each other.
Thus, two fundamental results can be extracted from our data. Firstly, the cultivation season was responsible for changes in the antioxidant activity and physicochemical quality of the 'Vanda' lettuce. Secondly, lettuce heads produced in different seasons presented different behaviors during cold storage.
We found that the highest values of antioxidant activity, represented by PhC and DPPH, were found in lettuces cultivated in the summer. Several studies have shown that high temperature, humidity level and solar radiation increase the incidence of agricultural pests, diseases and physiological stress in lettuce, and these factors induce the phenylpropanoids metabolism and increase the biosynthesis of phenolic compounds (such as flavonoids) and chlorophyll (Dixon and Paiva, 1995; Dussi, Sugar, and Wrolstad, 1995; Howard et al., 2002; Romani et al., 2000; Taiz and Zeiger, 2017). Thus, we believe that the high maximum average temperature (30.5 °C) and solar radiation (459.30 Ly day-1) observed at the summer time of production could have provided a pathological stress, resulting in higher values of PhC and DPPH of these lettuce heads. This behavior has also been observed in spinach produced in different seasons (Howard et al., 2002).
The cold storage conditions of the lettuce heads harvested in different growing seasons were the same, but VS lettuce showed a higher antioxidant activity compared to VW lettuce during the 28 days of study. The highest PhC and DPPH values recorded at the harvest, which remained superior in spite of the losses, might be a reason for this observation.
The same logic regarding the difference between the cultivation seasons and the behavior during the days of study can be applied to Chl. The presence of a greater amount of this pigment in VS is possibly due to the fact that its metabolic energy was larger than in VW, considering that the chlorophyll is also influenced by the intensity of the radiation. Some plants add more chlorophyll into photosystem II in order to dissipate the excess of energy absorbed in the form of fluorescence (quenching) (Taiz and Zeiger, 2017). This might have happened in VS due to the high solar radiation observed during its cultivation. Quenching is one of the main processes that regulate the excitation energy distribution for the photosystem reaction centers, and may be regarded as a regulator of the excitation flow to the photosystem II reaction center at an adequate level, depending on the radiation intensity (Taiz and Zeiger, 2017). This dissipation does not allow the occurrence of chlorophyll’s photo-oxidation.
Once value a* discussed in this study is related to the green color, we believe that the high radiation detected during VS production culminated in bright green color on the day of harvest, and a lighter green than lettuce VW during storage. Color intensity is related to the different types of chlorophyll that compose leaf pigmentation, hence leaves built in higher light conditions increase the production of chlorophyll a, that is the predominant type of chlorophyll in photosystem II (Taiz and Zeiger, 2017).
In contrast to what we observed for the antioxidant activity and color, physiological activity was not influenced by the cultivation seasons. Lettuce cultivated in both seansons presented high physiological activity at day 0 due to harvesting stress and ‘field heat’. During storage, ethylene production and respiration rate decreased for both VS and VW, with no difference in this behavior. Refrigeration minimizes respiratory heat generation by the product to be stored, by decreasing the intensity of the respiratory rate, ethylene production and the action of degradative and oxidative enzymes. The decrease in physiological activity during refrigerated storage has also been observed in some cultivars of lettuce (Chandra et al., 2006; Chandra et al., 2008; Mattos et al., 2008; Suthumchai et al., 2006 and 2007).
CONCLUSION
The environmental factors affect the qualitative attributes of ‘Vanda’ lettuce and determine the best cultivation season, while refrigeration only maintains those attributes and provides long shelf-life (28 days). Summer cultivation provides a greater antioxidant activity and better physicochemical quality of the 'Vanda' lettuce at the moment of harvesting, and high values of these attributes can be maintained for a period of 14 days of cold storage. More studies are needed to verify if this found also occurs to other varieties of lettuce, just as in other parts of the world.
Acknowledgments
The authors would like to thank the ‘São Paulo Research Foundation (FAPESP)’ for granting the scholarships (grant nº 2009/10806-7).
REFERENCES
Caldwell, C. R. 2003. Alkylperoxyl radical scavenging activity of red leaf lettuce (Lactuca sativa L.) phenolics. Journal of Agricultural and Food Chemistry 51: 4589-4595.
Chandra, D. et al. 2006. Postharvest changes in some physiological traits and activities of ammonia-assimilating enzymes in lettuce during storage. Asian Journal of Plant Sciences 5. 378-384.
Chandra, D. et al. 2008. Physio-biochemical changes in lettuce stored at ambient Temperature with prior storage at low temperature. Europe-Asia Symposium on Quality Management in Postharvest Systems 804: 585-592.
Costa, C.P.; Sala, F.C. 2005. A evolução da alfacicultura brasileira. Horticultura Brasileira 23 (artigo de contracapa).
Dixon, R. A.; Paiva, N. L. 1995. Stress-induced phenylpropanoid metabolism. Plant Cell 7: 1085-1097.
Dussi, M. C.; Sugar, D.; Wrolstad, R. E. 1995 .Characterizing and quantifying anthocyanins in red pears and the effect of light quality on fruit color. Journal of the American Society for Horticultural Science 120: 785-789.
Emmons, C. L.; Peterson, D. M.; Paul, G. L. 1999. Antioxidant capacity of oat (Avena sativa L.) extracts. In vitro antioxidant activity and contents of phenolic and tocol antioxidants. Journal of Agricultural and Food Chemistry 47: 4894-4898.
Ferreres, F. et al. 1997. Phenolic metabolites in red pigmented lettuce (Lactuca sativa). Changes with minimal processing and cold storage. Journal of Agricultural and Food Chemistry 45: 4249-4254.
HENZ, G. P.; SUINAGA, F. A. 2009. Tipos de alface cultivados no Brasil. In: Embrapa Hortaliças, Comunicado Técnico 75.
Hooper, L.; Cassidy, A. 2006. A review of the health care potential of bioactive compounds. Journal of the Science of Food and Agriculture 86: 1805-1813.
Howard, L. R. et al. 2002. Antioxidant capacity and phenolic content of spinach as affected by genetics and growing season. Journal of Agricultural and Food Chemistry 50: 5891-5896.
Krizek, D. T.; Britz, S. J.; Mirecki, R. M. 1998. Inhibitory effects of ambient levels of solar UV-A and UV-B radiation on growth of cv. New Red Fire lettuce. Physiologiae Plantarum 103: 1-7.
Larrigaudiere, C. et al. 2004. Biochemical characterisation of core browning and brown heart disorders in pear by multivariate analysis. Postharvest Biology and Technology 31: 29-39.
Llorach, R. et al. 2008. Characterisation of polyphenols and antioxidant properties of five lettuce varieties and escarole. Food Chemistry 108: 1028-1038.
Matsuno, H.; Uritani, I. 1972. Physiological behavior of peroxidase isoenzymes in sweet potato root tissue injured by cutting or with black rot. Plant and Cell Physiology 13: 1091-1101.
Mattos, L. M. et al. 2008. Respiratory activity and ethylene evolution of fresh-cut crisphead lettuce stored under two temperatures. Ciencia e Agrotecnologia 32: 1985-1990.
Mensor, L. L. et al. 2001. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytotherapy Research 15: 127-130.
Perucka, I.; Olszowka, K. 2011. Accumulation of potassium, magnesium, calcium in fresh and cold stored leaves of lettuce (Lactuca sativa L.) After CaCl2 foliar treatment before harvest. Journal of Elementology 16: 445-454.
Romani, A. et al. 2002. Polyphenols in greenhouse and open-air-grown lettuce. Food Chemistry 79: 337-342.
Romani, A. et al. 2000. HPLC analysis of flavonoids and secoiridoids in leaves of Ligustrum vulgare L. (Oleaceae). Journal of Agricultural and Food Chemistry 48: 4091-4096.
Sala, F.C., Costa, C.P. 2012. Retrospective and trends of Brazilian lettuce crop. Horticultura Brasileira 30: 187-194.
Serafini, M. et al. 2002. Effect of acute ingestion of fresh and stored-lettuce (Lactuca sativa) on plasma total antioxidant capacity and antioxidant levels in human subjects. British Journal of Nutrition: 88: 615-623.
Singleton, V. L.; Orthofer, R.; Lamuela-Raventos, R. M. 1999. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Oxidants and Antioxidants 299: 152-178.
SOUNIS, E. 1985. Bioestatística: princípios fundamentais, metodologia estatística, aplicação às ciências biológicas (3 ed.). Rio de Janeiro: Atheneu, 317p.
Suthumchai, W. et al. 2006. Changes in acid invertase activity and sugar contents in lettuce during storage at ambient temperature. Asian Journal of Plant Sciences 5: 304-310.
Suthumchai, W. et al. 2007. Sugar metabolizing enzymes activities in lettuce head during low temperature storage. Asian Journal of Plant Sciences 6: 568-576.
Taiz, L.; Zeiger, E. 2017. Fisiologia e Desenvolvimento vegetal (6 ed.): Porto Alegre: Artmed, 858p.
Wells, O. S. 1996. Rowcover and high tunnel growing systems in the United States. Hort-Technology, 6: 172-176.
Zhao, X., et al. 2007. Influences of organic fertilization, high tunnel environment, and postharvest storage on phenolic compounds in lettuce. Hortscience 42: 71-76.
Zhao, X.; Iwamoto, T.; Carey, E.E. 2007. Antioxidant capacity of capacity of leafy vegetables as affected by high tunnel environment, fertilisation and growth stage. Journal of the Science of Food and Agriculture 87: 2692-2699.
Notas de autor