Artículo científico

This work is licensed under Creative Commons Attribution-NonCommercial 4.0 International.
Abstract: To know the reproductive cycle of Scomberomorus sierra, from January 2003 to March 2004, individuals were monthly obtained in the coast of Colima, Mexico. Individuals were obtained with hand lines, troll and gillnet, which helped obtain different lengths and age groups. Sex ratio was 1:1.03 female: males. Average length of sexual maturity L50 = 54.3 cm of total length (TL, cm) in both males and females with 3.79 years of age, length of first maturity (L25) for both was 47.3 cm corresponding to 3.08 years of age. The gonadosomatic index was at its maximum values in March and August. The allometric relationship between the liver weight and the fish weight is isometric (b = 1.004). Monthly values of the hepatosomatic index are higher in March, May, and November - December. The condition factors show two main seasons of wellness. Total fecundity was 416,483 to 4’354,860 oocytes for females of 4 to 15 years old. Relative fecundity was average: 703.7 oocytes·g-1 (312.6 - 1,438.4 oocytes·g-1 in females from 4 to 15 years old). Average oocytes diameter was 0.44 mm (0.34 - 0.59 mm). The species should be protected from fishing from February to May and August to October, and under 50 cm TL.
Keywords: Reproduction length (L50), first maturity length (L25), hepatosomatic index, gonadosomatic index, condition factor.
Resumen: Para conocer el ciclo reproductivo de Scomberomorus sierra, se colectaron mensualmente individuos en las costas de Colima (México), entre enero de 2003 y marzo de 2004. Los individuos se obtuvieron con líneas de mano, curricán, caña y redes agalleras para obtener individuos de diferentes longitudes y grupos de edad. La proporción de sexos fue de 1: 1.03 hembras: machos. La longitud promedio de la madurez sexual L50 = 54.3 cm de longitud total (TL, cm) tanto para machos como hembras correspondientes a 3.79 años de edad; la longitud promedio de primera madurez L25 = 47.3 cm TL para ambos sexos, 3.08 años de edad. El índice gonadosomático alcanza sus máximos valores en marzo y agosto. La relación alométrica entre el peso del hígado y el peso del pez es isométrica (b = 1.004). Los valores mensuales del índice hepatosomático son mayores en marzo, mayo, noviembre y diciembre. El índice de repleción gástrica muestra sus valores más altos en enero, marzo, abril, julio-agosto, noviembrediciembre. Los factores de condición muestran dos épocas principales de mayor gordura. La fecundidad total fue 416,483 a 4’354,860 ovocitos en hembras de 4 a 15 años de edad. La fecundidad relativa promedio fue 703.7 ovocitos·g-1 (312.6 - 1,438.4 ovocitos·g-1 en hembras de 4 a 15 años de edad). El diámetro promedio del ovocito fue 0.44 mm (0.34 - 0.59 mm). La especie debe protegerse de la pesca de febrero a mayo y de agosto a octubre, y organismos de menos de 50 cm TL.
Palabras clave: Longitud de reproducción (L50), longitud de primera madurez (L25), índice hepatosomático, índice gonadosomático, factor de condición.
Introduction
The Pacific sierra Scomberomorus sierra Jordan & Starks, 1895, is an epipelagic neritic species which forms schools and spawns close to the coast on the continental shelf. Its entire distribution area goes from South California to Peru, including the Galapagos Islands. It feeds mainly on anchovies and sardines (Collette and Nauen, 1983). Its major captures occur in Mexico, where it is captured by sport fishers and abundant enough to sustain a commercial fishery (Fischer et al., 1995).
Captures of the Pacific sierra in Colima, Mexico, show two marked seasonal periods due to migration: in summer they swim north and in winter they migrate south (MárquezMillán, 1972). S. sierra is more abundant in the coasts of Manzanillo during February and March and from September to November (Chávez-Ruiz, 1986). S. sierra is commercially considered a second class species with a price to fishermen from $3.00 to $4.00 USD per kilogram.
Studies on its taxonomic description have been done by Castro-Aguirre (1978),Chirichigno et al. (1982) and Collette and Nauen (1983). General features of its biology and fisheries are known by the studies of Márquez-Millán (1972),Chávez-Ruiz (1986), Espino-Barr et al. (1990) and Aguirre-Villaseñor et al. (2006), and an analysis of otolith’s shape by Ramírez-Pérez et al. (2010).
Although the Pacific sierra is a commercially important species, studies on its reproduction are limited and have been done by Collette and Nauen (1983), Chávez-Ruiz (1986), Aguirre-Villaseñor et al. (2006), Lucano-Ramírez et al. (2011) and Vega et al. (2013). This is the first study of S. sierra reporting data on fecundity of the species.
Studies on reproduction of species are important because length of first maturity and reproduction are calculated and evaluate if the species is captured as an adult or before it has spawned at least once, assuring the recruitment of individuals to the adult population and avoid overexploitation. Likewise, periods of massive spawning are obtained, to formulate fishing closure of banned tables and understand the reproduction phenomena associated to the storing of reserve substances, feeding indexes and fecundity of the species, which also explain their evolutionary success.
According to this, the objective of this paper is to analyze the reproductive cycle of Scomberomorus sierra determining: Sex ratio, average length of sexual maturity (L50) and length of first maturity (L25), gonadosomatic index, the relationship between the liver weight and the fish length and weight, hepatosomatic index, gastric repletion index, condition factor of Fulton, Clark and Safran, total and relative fecundity, oocytes diameter and comparison of the results obtained in the present study with those obtained by other authors.
Material and methods
From January 2003 to March 2004, individuals of S. sierra were obtained monthly from the commercial captures of the coastal fishery in Manzanillo, Colima, Mexico (18°40’ to 19°10’N and 103°45’ to 104°35’W) (figure 1). Total length (TL) and total weight (TW) from 972 organisms were obtained directly in situ throughout the study period.
From these, 464 were transported to the laboratory, where total length (TL), total (TW) and eviscerated weight (EW) and sex were registered for each organism. Individuals were captured with hand lines and hooks No. 7-9, troll and gillnet (3.5-4.5 inch mesh size), which helped obtain different lengths and age groups.
Sex and gonad maturation were determined in visu, on fresh organisms the same day they were caught. The stages of sexual maturity were determined using the key described in Espino-Barr et al. (2008), where:
Phase I: undefined, sexual glands are a fine filament, and females and males cannot be differentiated.
Phase II: Immature, the gonads start developing, ovaries are rose translucent and testes resemble a whitish lace.
Phase III: Maturing, sexual glands are well differentiated. Ovaries look granular, pink-yellowish color, oocytes small and opaque; testes are ivory white.
Phase IV: Mature, sexual glands are well developed, ovaries are rose-orange color, oocytes are big and transparent, and testes are whitish.
Phase V: Spawning, ovaries are brilliant orange color; sexual products are ready to be expelled and are pushed out at the slightest pressure, veins are well developed irrigating the entire gonad; testes are white pearly white, sperm emerges at a light pressure.
Phase VI: Post-spawn, product has been expelled; sexual glands are flaccid, swelled and brownish-grey. Residual oocytes are reabsorbed.
The first spawning TL for males and females was determined by 50% of the accumulative frequency (L50) of stages IV and V of sexual maturation (Sparre and Venema, 1995), and the minimum TL of first spawning (L25) was also recorded to compare with other authors findings (Rodríguez-Gutiérrez, 1992). The logistic function was described by the equation (Gaertner and Laloe, 1986; Sparre and Venema, 1995):
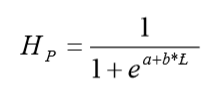
Where: HP = percentage of females or males sexually mature, a and b are constants. Its logarithmic transformation is: ln(1(1/Hp-1)) = a-b ∙ TL, and the length at which 50% of the population is sexually mature (L50) corresponds to: L50 = a/b. The original equation is modified to include L50: Y = 1/[1+a(1-TL/L50)].
To calculate the length of recruitment (Lr) and first capture (Lc), data of all individuals were used, separated by sex. The same analysis of the logistic function was used (Gaertner and Laloe, 1986; Sparre and Venema, 1995).
The gonadosomatic index (GSI) for females and males was calculated according to Rodríguez-Gutiérrez (1992), where gonad weight (GW) is expressed as a function of body weight: GSI = 100∙GW/TW (TW = total weight). As a measure of physical fitness of the fishes, we obtained the condition factor K = (EW∙TL-3) ∙ 100 (Clark, 1928), K = (TW∙TL-3) ∙100 (Fulton, 1902) and a = TW∙TL-b and a = EW∙TL-b (Safran, 1992).
The hepatosomatic index (HSI), expressed as the percentage of liver weight (LW) with respect to the total weight HSI = 100∙LW/TW (Rodríguez-Gutiérrez, 1992). Gastric repletion index (GRI) was calculated as GRI = 100∙SW/TW, as a measure of the more intense feeding seasons, where SW = stomach weight.
Fecundity (F) and relative fecundity were obtained by the gravimetric method using the wet weight of 42 phase V female gonads of S. sierra. To estimate total fecundity, two subsamples of 0.1 g were obtained of each individual and put in a modified Gilson fluid (Simpson, 1951) to preserve. All oocytes were counted with the help of a stereoscopic microscope and measured with a micrometric ocular.
The following expression was used in the calculation: F = n · GW/gi, where F = fecundity of a sample; n = number of oocytes in the subsample; GW = weight of the gonad (grams) and gi = weight of the subsample (grams) (Holden and Raitt, 1975). The relationship between fecundity and total length and weight was calculated with the formula F = a∙xb where F= fecundity, x = individual weight or length, a = origin ordinate or initial number of oocytes, b = trend or oocyte number changing rate.
The relationships among TL, TW, LW, testis weight (TeW), ovary weight (GW), and fecundity were defined for different ages. Age groups were obtained by sagittal otolith analysis; five age groups of S. sierra were established by Gallardo-Cabello et al. (2011) and Nava-Ortega et al. (2012).
Results
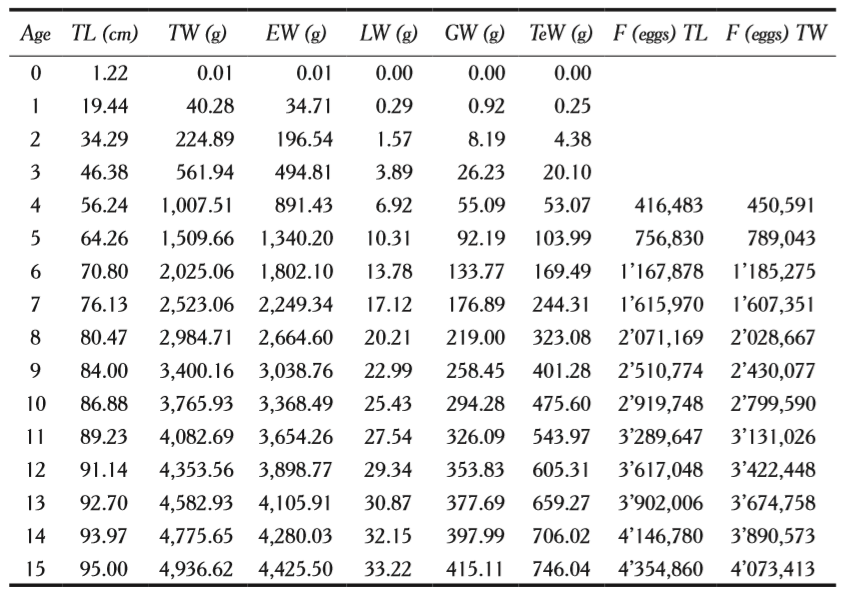
Scomberomorus sierra does not show sexual dimorphism, therefore sex can only be determined by dissecting and observing the gonads. The virgin individuals cannot be determined by simple sight. Ovaries, as in many fish species are cylindrical when females have matured and oocytes can be observed in different tones from yellow to orange. Testes are long and whitish and pearly tones. Table 1 shows average values of the gonad weight (GW, g) for each age group, as well as the length (TL, cm), total weight (TW, g) and eviscerated weight (EW, g), and liver weight (LW, g). Testes are heavier than ovaries from age 5 on; for example, for an individual of 5 years old, the weight of the ovaries is 92.19 g and that of the testis of 103.99 g (table 1).
Oocyte diameter was 0.44 mm (±0.07 mm standard deviation, SD, n = 90), minimum 0.34 mm and maximum 0.59 mm. Fecundity values ranged from 416,483 to 4’354,860 oocytes (± 1’158, 956 SD, n = 42) in females of four to fifteen years of age and lengths of 56.24 cm to 95.00 cm, and 1,007.51 g to 4,936.62 g of weight (table 1). Average value of the relative fecundity was of 703.7 oocytes·g-1 (ranging from 312.6 to 1,438.4 oocytes·g-1, 320.7 SD, n = 42).
Length (TL, cm), total weight (TW, g), eviscerated weight (EW, g), liver (LW, g), ovary weight (GW, g) and testis weight (TeW, g) for each age group (years), of Scomberomorus sierra.
Sample size was of 476 organisms of S. sierra of which 233 (48.95%) were females, and 242 (50.84%) males, and 1 (0.20%) undetermined. The proportion of female: male was 1: 1.03 (X2 = 0.827, p > 0.10). Lengths went from 30 to 97 cm (average 49.6 cm, ±11.3 SD), but females were bigger (30 to 97 cm, average 52.2 cm, ±12.7 SD), than males (30 to 83 cm, average 47.2 cm ±9.0 SD) (figure 1).
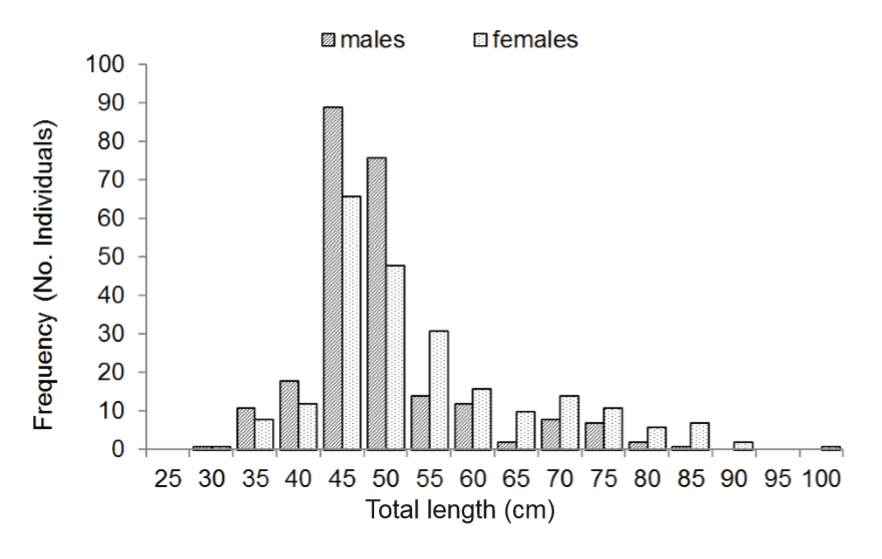
Figure 1
Length distribution of Scomberomorus sierra.
Monthly variations of the relative frequency of gonad maturity phases (figure 2) show that phase II or immature reaches its higher values from May to July, and January to March. Phase III was more abundant during August 2003 (46.34 %), showing values between 10% and 16% in the months of February to April, September to November. Phase IV, mature, the higher values are observed from February to and April. Phase V or spawning stage: spawning occurs in the months of March to May, and August to November. High values were also observed in February. Phase VI, post-spawning was observed in its highest values in January to April and a second period in September to December, but most months showed post-spawned organisms. These data assume two spawning seasons throughout the year.
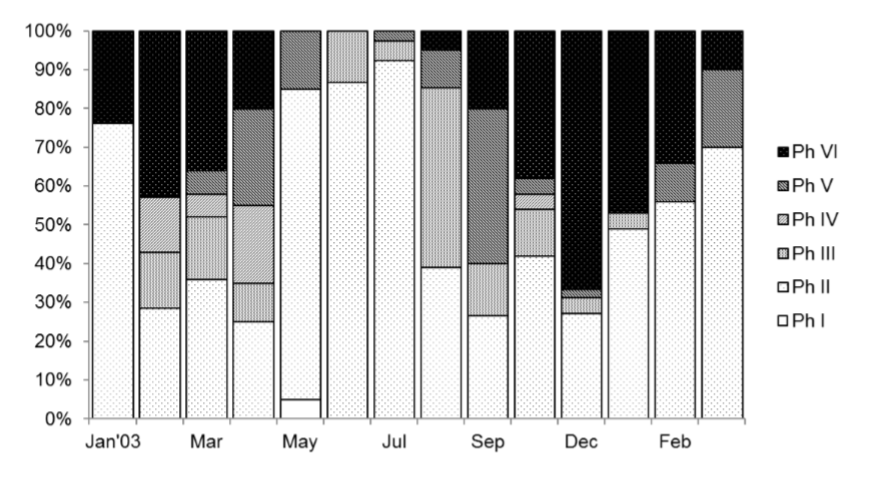
Figure 2
Monthly relation of gonad maturity in Scomberomorus sierra.
Length of first maturity was L25 = 47.30 cm and first reproduction length was
L50 = 54.30 cm for the specie (table 2, figure 3 a) corresponding to three years of age. For females, the values were: L25 = 47.0 cm and L50 = 53.5 (figure 3b), and in males L25 = 52.80 cm and L50 = 60.7 cm in males (figure 3c).
Length (TL), weight (TW) and age of Scomberomorus sierra at recruitment (Lr), first capture (Lc), first maturity (L25) and reproduction (L50).
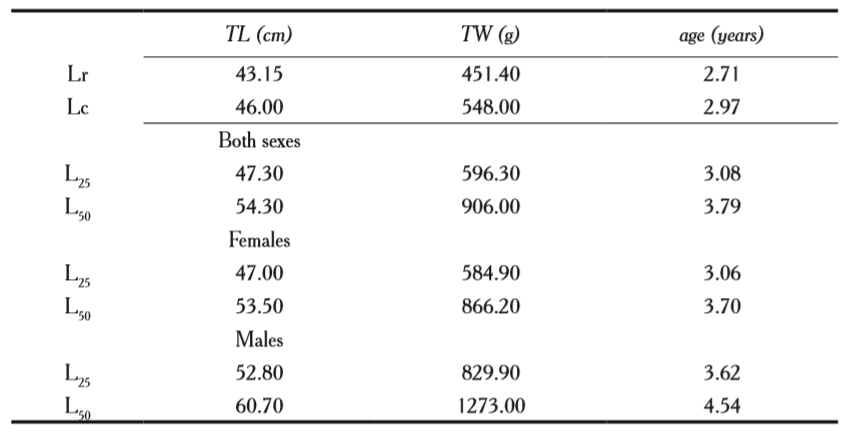
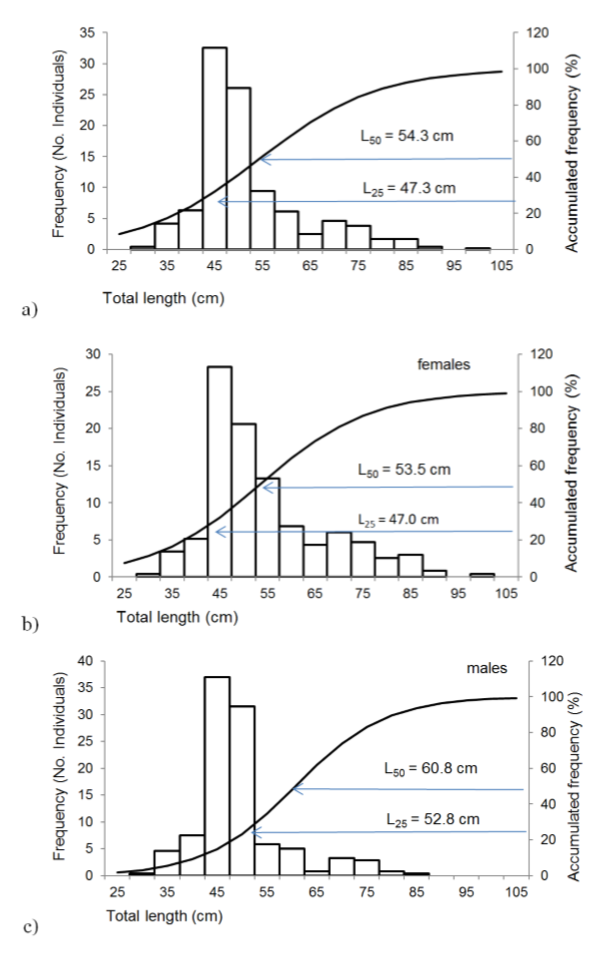
Figure 3
First maturity length (L25) and first reproduction length (L50) of a) all individuals of the species, b) females and c) males of Scomberomorus sierra.
The gonadosomatic index (GSI) reaches its highest values during March, May, July, August and September, and decreases in November (figure 4a).
The allometric relationship between the liver weight and total weight of the organism obtained in the present study was (LW = 0.008 TW1.004, r2 = 0.847). The allometric index b indicates that the liver grows in the same proportion (b = 1.004, t = 0.241, p > 0.05) in terms of the organism weight, which results in an isometric growth of the fish, incrementing its fatty reserves as it ages in the same proportion. But there are changes throughout the year: HSI variations show higher values during March, May, November, December 2003 and February 2004. Lower values are observed during February, April, September 2003 and March 2004 (figure 4b).
Variations in the gastric repletion index (figure 4c), showed higher values during January, March, April, July, August, November and December 2003 and March 2004; lower values are observed in February, April, September 2003 and March 2004, meaning there are changes in the food availability, therefore changes in the body indexes.
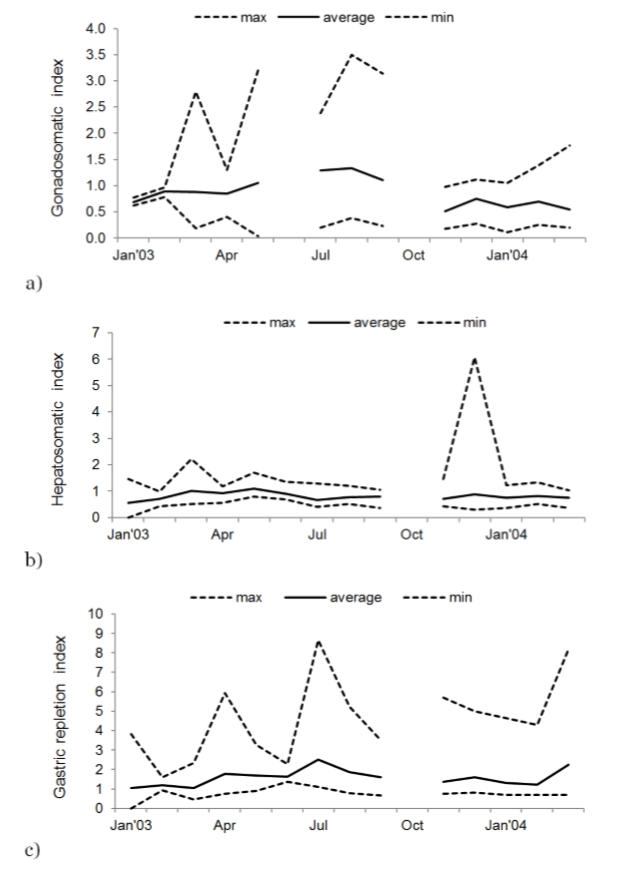
Figure 4
Monthly variation of a) the gonadosomatic index, b) hepatosomatic index, and c) gastric repletion index all individuals of Scomberomorus sierra.
The condition factor values calculated with Fulton’s equation are higher during the months of March, May, June, November and December 2003 and January, February and March 2004 (figure 5a). The condition factor values obtained with Clark’s equation are higher during the months of May, November, December of 2003 and February and March 2004 (figure 5b).
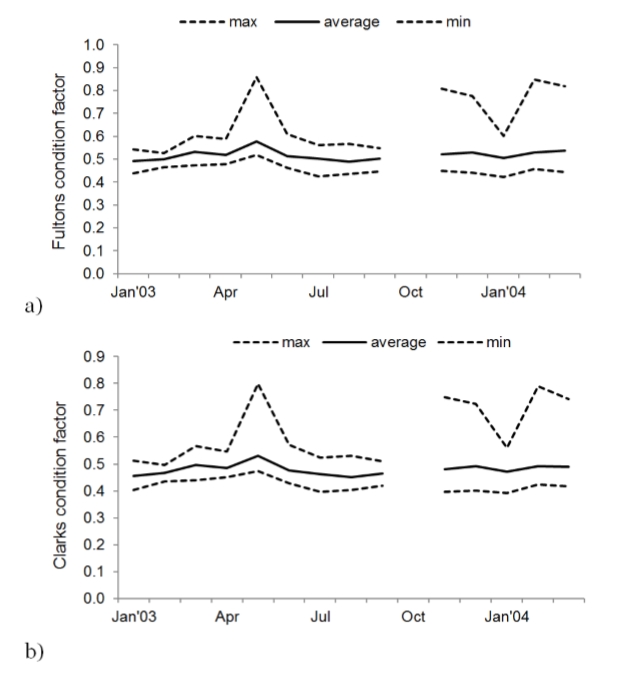
Figure 5
Monthly values of the relative condition factor: a) by the method of Fulton (1902), b) by the method of Clark (1928), of all individuals of Scomberomorus sierra.
The condition factor for total weight and eviscerated weight obtained by Safran’s method reached its higher values during May, November and December 2003 and February and March 2004 (figure 6).
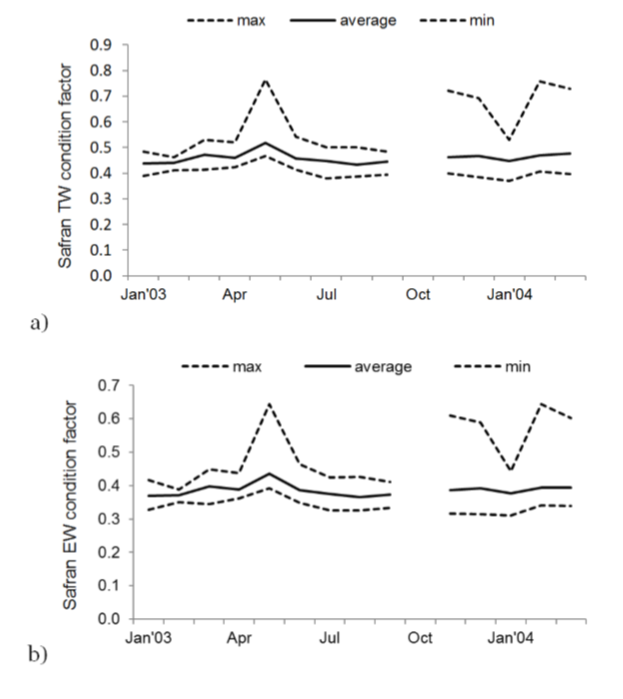
Figure 6
Monthly values of the relative condition factor of all individuals of Scomberomorus sierra: a) Safran’s method with total weight, b) Safran’s method with eviscerated weight.
Discussion
The highest values of the length growth rate of Scomberomorus sierra are in groups one to three years of age, where the lengths are from 18.22 in group 0 to 1, 14.85 cm from group of age 1 to 2, and 12.09 cm in group of age 2 to 3, obtained by difference of the length, observed in table 1. This means that from age 3 on the gonadal maturity begins and the growth rate values in length diminish; the growth rate in total weight, eviscerated weight, ovary weight, testis weight and the fatty reserve index start rising up.
Therefore, the highest increments in length of S. sierra occurs before the sexual maturity, period in which most part of the energy goes to the growth in length, to lessen the predators attacks and intraspecific competence (Nikolsky, 1963; Gallardo-Cabello et al., 2015;Espino-Barr et al., 2016).
Later, once the sexual maturity has begun, the energy is focused in the production of the sexual products, in the reproduction, to assure recruitment of new individuals to the area (Gallardo-Cabello et al., 2015,2016;Espino-Barr et al., 2008, 2015). In our case, analyzed data come from the fishery, which means that the sample was taken during the migration in front of the coast of Jalisco and Colima.
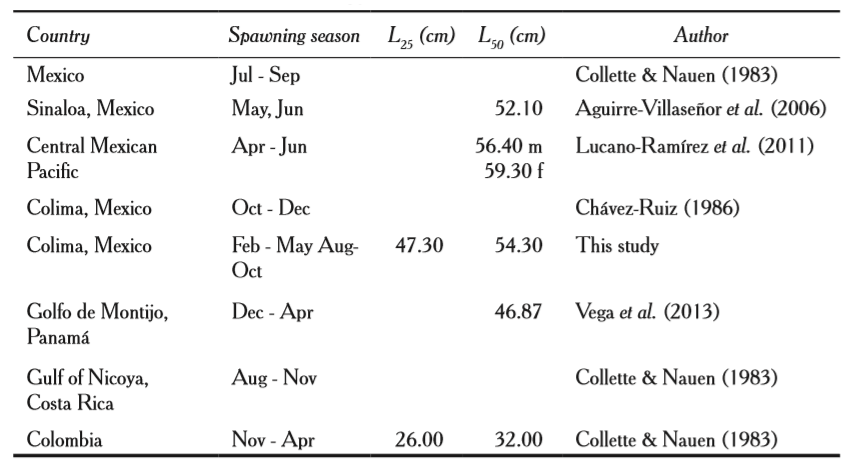
In the coasts of Jalisco and Colima, S. sierra presents two periods of massive reproduction: from February to May and from August to October (table 3). The second period is similar to that reported in the Gulf of Nicoya by Collette and Nauen (1983). The first period described in our study coincides partially in August and September by that reported for Colombia (Collette and Nauen, 1983).
Similarly, data reported for Mexico by these authors are comparable with those found in this analysis for the months of August and September (Collette and Nauen, 1983). Likewise, in the Central Mexican Pacific, our results are similar to those reported by Lucano-Ramírez et al. (2011) in the months of April and May. In the State of Sinaloa this coincides with the month of May (Aguirre-Villaseñor et al., 2006).Vega et al. (2013) reported in the Gulf of Montijo, Panama, similar data that coincides with the reports for February to April. Differences can probably be associated with environmental conditions.
Spawning seasons and first maturity (L25) and reproduction (L50) lengths of Scomberomorus sierra.
Note: m = males, f = females.
Length of first maturity in Colombia was of 26.00 cm and the first reproduction of 32.00 cm reported by Collette and Nauen (1983), which are inferior to those found in this study (table 3). Similar values as our findings were published by Lucano-Ramírez et al. (2011), with a length of first reproduction L50 = 56.40 cm for males and L50 =59.30 cm for females (in our analysis males were L50 = 60.70 cm, and females L50 = 53.50 cm).
Aguirre-Villaseñor et al. (2006) reported a value of L50 = 52.10 cm, close to ours. Vega et al. (2013) obtained a value of L50 = 46.87 cm, similar to that obtained in this study. Differences in the results are related to the sample; in some places fishing has a higher pressure than in other, so the older individuals are not present any more.
The gastric repletion index indicates that there are months in which feeding is more active, due to the availability of food, of which the fish take advantage, preparing for the gonadic maturation. The use of several condition factor indexes allows comparing results between total weights and eviscerated weight of fish, also between the cubic parameter and the one obtained in the weight-length relationship with data of this study. In this analysis these indexes have a similar trend, which reinforces the results that there are two seasons in which the body is heavier compared to the body size.
Regarding other species of the Scomberomorus genus, it was observed that in S. concolor there are two massive spawning periods: a more intense in May and another less intense in September (Montemayor-López and Cisneros-Mata, 2000). Periods which are similar to those reported in this paper.
Table 4 shows fecundity values of other species of the genus Scomberomorus present in the Atlantic Ocean. S. regalis and S. maculatus show values of fecundity lower to those found in our study. S. regalis showed values of 160,000 to 2’230,000 oocytes in the Oriental Atlantic, according to Collette and Nauen (1983), and 161,000 to 2’234,000 oocytes South of Florida according to Finucane and Collins (1984).
In the case of S. maculatus values of minimum 28,300 were observed in the Mississippi Delta, US (Finucane et al., 1986), and a maximum of 2’113,000 oocytes in the Northeast of Florida, US (Finucane et al., 1986).
Fecundity equations, number of oocytes (Minimum and maximum) of species of the Scomberomorus genus
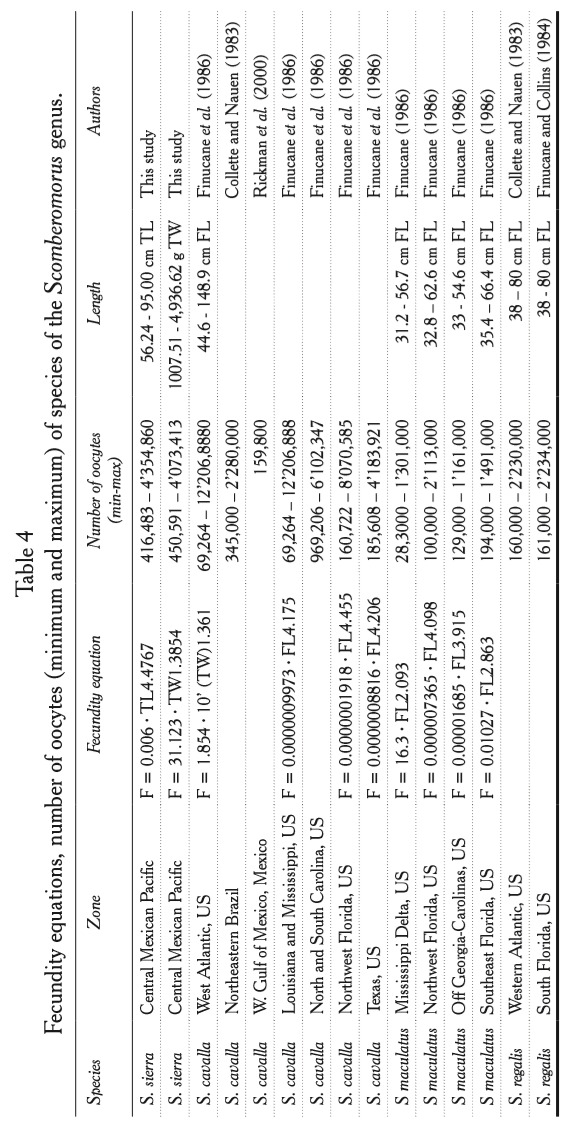
As for S. cavalla of the Northeast of Brazil, the observed values of fecundity were lower than those of S. sierra, 345,000 to 2’280,000 oocytes (Collette and Nauen, 1983), the same occurred in Texas, US, where S. cavalla showed values of 185,608 to 4’183,921 oocytes (Finucane et al., 1986).
On the other hand, Southeast of US, North and South Carolina, S. cavalla reaches maximum values higher than S. sierra: 6’102,347 oocytes, 8’070,585 oocytes Northwest of Florida US, 12’206,888 oocytes in Louisiana and Mississippi US, and 12’207,000 oocytes Southeast of US (Finucane et al., 1986).
Conclusions
According to these analyses, we conclude that two ban seasons should be implemented: May and September, so mature organisms can complete their reproduction process and the newborn organisms during these periods can recruit to the growth area, assuring an increment of the population biomass for the next fishing seasons.
Acknowledgement
We want to express our gratefulness to fishermen who kindly give us always their wisdom and help collect the samples.
References
Aguirre-Villaseñor, H.; Morales-Bojórquez, E.; Morán-Angulo, R. E.; Madrid-Vera, J. and Valdez-Pineda, M. C. (2006). Biological indicators for the Pacific sierra (Scomberomorus sierra) fishery in the southern Gulf of California, Mexico. Ciencias Marinas 32(3): 471-484.
Castro-Aguirre, J. L. (1978). Catálogo sistemático de los peces marinos que penetran a las aguas continentales de México, con aspectos zoogeográficos y ecológicos. Depto. Pesca. INP, México. Serie Científica 19. 298 pp.
Chávez-Ruiz, E. J. (1986). Análisis preliminar biológico-pesquero de la sierra, Scomberomorus sierra, Jordan y Starks 1895, en la Bahía de Manzanillo, Colima. Tesis de licenciatura, ENEP Iztacala, Universidad Nacional Autónoma de México. México, 52 pp.
Chirichigno, N.; Fischer, W. and Nauen, C. E. (1982). INFOPESCA. Catálogo de especies marinas de interés económico actual o potencial para América Latina. Parte 2. Pacífico Centro y Suroriental. Roma FAO/PNUD, SIC/82/2: 588 pp.
Clark, F. (1928). The weigth-length relationship of the Californian sardine (Sardina coerulea) at San Pedro. Fish. Bull. U.S. 12: 22-44.
Collette, B. B. and Nauen, C. E. (1983). Scombrids of the world. An annotated and illustrated catalogue of tunas, mackerels, bonitos and related species known to date. FAO Species Catalogue. Rome 125(2): 137 pp.
Espino-Barr, E.; Cruz-Romero, M. y García-Boa, A. (1990). Biología pesquera de tres especies de la familia Scombridae en el litoral de Colima, México. In: Dailey, M. and Bertsch, H. (Ed.). Memorias del VIII Simposium de Biología Marina. Ensenada, México. pp. 65-74.
Espino-Barr, E.; González-Vega, A.; Santana-Hernández, H. y González-Vega, H. (2008). Manual de biología pesquera. Universidad Autónoma de Nayarit. ISBN 968-833-076-0, 168 pp.
Espino-Barr, E.; Gallardo-Cabello, M.; Cabral-Solís, E. G.; Puente-Gómez, M. and García-Boa, A. (2015). Reproduction of Gerres cinereus (Percoidei: Gerreidae) off the Mexican Pacific coast. Revista Ciencias Marinas y Costeras 7:83-98.
Espino-Barr, E., Gallardo-Cabello, M.; Puente-Gómez, M. and García-Boa, A. (2016). Reproduction of Mugil cephalus (Percoidei: Mugilidae) off the Central Mexican Pacific Coast. Fisheries and Aquaculture Journal 7(4): 1-9.
Finucane, J. H. (1986). Reproduction of Spanish Mackerel, Scomberomorus maculatus, from the southeastern United States. Northeast Gulf Sci. 8(2): 97-106.
Finucane, J. H. and Collins, L. A. (1984). Reproductive biology of Cero, Scomberomorus regalis, from the coastal waters of South Florida. NE Gulf Science 7 (1): 101-107.
Finucane, J. H.; Collins, L. A.; Brusher, H. A. and Saloman, C. H. (1986). Reproductive biology of King Mackerel, Scomberomorus cavalla, from the Southeastern United States. Fishery Bulletin 84(4): 841-850.
Fischer, W.; Krupp, F.; Schneides, W.; Sommer, C.; Carpenter, K. E. y Niem, U. H. (1995). Guía FAO para la identificación de especies para los fines de la pesca. Pacífico Centro Oriental. Vertebrados Vols. II y III, Roma, FAO, pp. 644-1813.
Fulton, T. W. (1902). The rate of growth of fishes. 20th Annual Report of the Fishery Board of Scotland 3: 326-446.
Gaertner, D. and Laloe, F. (1986). Etude biometrique de la talle a’premier maturité sexualle de Geryon maritae, Maning et Holthuis, 1981 de Senegal. Oceanologica Acta 9(4): 479-487.
Gallardo-Cabello, M.; Espino-Barr, E.; Nava-Ortega, R. A.; García-Boa, A.; Cabral-Solís, E. G. and Puente-Gómez, M. (2011). Analysis of the otoliths of Sagitta, Asteriscus and Lapillus of Pacific Sierra Scomberomorus sierra (Pisces: Scombridae) in the coast of Colima México. Journal of Fisheries and Aquatic Science 6(4): 390-403.
Gallardo-Cabello, M.; Espino-Barr, E.; García-Boa, A.; Puente-Gómez, M. and Cabral-Solís, E. G. (2015). Fishery analysis of Diapterus brevirostris in the central Mexican Pacific. Avances en Investigación Agropecuaria 19(1): 77-93.
Gallardo-Cabello, M.; Espino-Barr, E.; García-Boa, A.; Puente-Gómez, M. and Cabral-Solís, E. G. (2015). Reproduction of Diapterus brevirostris (Percoidei: Gerreidae) in the Mexican Pacific coast. Global Journal of Fisheries and Aquaculture. Science Research Journals 3(5): 221-229.
Gallardo-Cabello, M.; Espino-Barr, E.; García-Boa, A. and Puente-Gómez, M. (2016). Fishery analysis of Mugil cephalus in Central Mexican Pacific coast. Journal of Marine Biology & Oceanography 5:1. doi:10.4172/2324-8661.1000151.
Holden, M. J. and Raitt, D. F. S. (1975). Manual de Ciencia Pesquera. Parte 2.- Métodos para investigar los recursos y su aplicación. ONU/FAO. Doc. Tec. sobre pesca. No. 115. Rev. 1. 207 pp.
Lucano-Ramírez, G.; Ruíz-Ramírez, S.; Palomera-Sánchez, F. I. y González-Sansón, G. (2011). Biología reproductiva de la sierra Scomberomorus sierra (Pisces, Scombridae) en el Pacífico Central de México. Ciencias Marinas 37(3): 249-260.
Márquez-Millán, R. (1972). Sierra del Pacífico. Unidades de producción. 171-182. Diagnóstico de la situación actual de las pesquerías de México. Programa de Investigaciones y Fomento Pesquero. México/ PNUD/FAO. SC/Depto. Pesca, INP, 685 pp.
Montemayor-López, G. y Cisneros-Mata, M. A. (2000). Sierra del Golfo de California. In: Sustentabilidad y pesca responsable en México: evaluación y manejo 1999-2000. INAPESCA-SEMARNAP. p. 250-274.
Nava-Ortega, R. A.; Espino-Barr, E.; Gallardo-Cabello, M.; García-Boa, A.; Puente-Gómez, M. and Cabral-Solís, E. G. (2012). Growth analysis of the Pacific Sierra Scomberomorus sierra in Colima, México. Revista de Biología Marina y Oceanografía 47(2): 273-281.
Nikolsky, G. V. (1963). The Ecology of Fishes. Academic Press, London, 351 pp.
Ramírez-Pérez, J. S.; Quiñónez-Velázquez, C.; García-Rodríguez, F. J.; Félix-Uranga, R. and MeloBarrera, F. N. (2010). Using the shape of sagittal otoliths in the discrimination of phenotypic stocks in Scomberomorus sierra (Jordan & Starks, 1895). Journal of Fisheries and Aquatic Science 5(2): 82-93.
Rickman, S. J.; Dulvy, N. K.; Jennings, S. and Reynolds, J. D. (2000). Recruitment variation related to fecundity in marine fishes. Can. J. Fish. Aquat. Sci. 57: 116-124.
Rodríguez-Gutiérrez, M. (1992). Técnicas de evaluación cuantitativa de la madurez gonádica en peces. AGT Ed. 79 pp.
Safran, P. (1992). Theoretical analysis of the weight-length relationship in fish juveniles. Mar. Biol. 112: 545-551.
Simpson, A.C. (1951). The fecundity of the Plaice. Fishery Investigations London, Serie 2, 18(5): 1-27.
Sparre, P. and Venema, S. C. (1995). Introducción a la evaluación de recursos pesqueros tropicales. Parte 1 - Manual. FAO Doc. Tec. de Pesca 306/1. Roma, 420 pp.
Vega, A. J.; Quezada, G. F. and Robles P. Y. A. (2013). Aspectos biológicos y pesqueros se Scomberomorus sierra (Perciformes: Scombridae) en el Golfo de Montijo, Pacífico de Panamá. Tecnociencia 15(2): 53-70.
Author notes
elespino@gmail.com
