Artículo científico
Arbuscular Mycorrhiza Interaction with Medicago sativa Plants: Study of Abiotic Stress Tolerance in Sustainable Agriculture
Micorrizas arbusculares en interacción con plantas de Medicago sativa: Estudio de la tolerancia al estrés abiótico en agricultura sustentable
Arbuscular Mycorrhiza Interaction with Medicago sativa Plants: Study of Abiotic Stress Tolerance in Sustainable Agriculture
Avances en Investigación Agropecuaria, vol. 25, núm. 3, pp. 26-40, 2021
Universidad de Colima
Recepción: 08 Febrero 2021
Aprobación: 26 Julio 2021
Resumen: Objetivo:Investigar los efectos de la inoculación con Rhizophagus intraradices sobre parámetros fisiológicos y defensa antioxidante en plantas de Medicago sativa bajo estrés salino, sequía y frio. Materiales y métodos: Semillas de M. sativa, var. Dekalb (DK166), fueron desinfectadas y sembradas en una mezcla de tierra y arena esterilizada; 10 g de R. intraradices fue adicionado a la mitad de las macetas y todas ellas recibieron5 ml de un cultivo de Shinorhizobium meliloti. Crecieron en un invernáculo a 25/20 °C (día/noche), con 16 h luz, 50-60% humedad relativa y riego normal durante 45 días. Después de ese tiempo se realizaron los tratamientos de control, sequía, salinidad y frio. La colonización de R. intraradices fue calculada en todos los tratamientos. La conductancia estomática fue medida usando un porómetro y la eficiencia del fotosistema II (PE) fue medida conFluor Pen FP100. El peso seco de tallos y raíces, el contenido de prolina, el daño oxidativo de lípidos y la actividad enzimática de SOD, CAT, APX fueron determinadas en todos los tratamientos después del estrés. Resultados:Todos los parámetros fisiológicos y enzimáticos medidos fueron significativamente (t-Student) mejores en plantas inoculadas, mostrando una interacción positiva entre micorrización-estándar fisiológico. La conductancia estomática, en plantas inoculadas, incrementó bajo sequía y salinidad, mostrando protección frente a estos estreses abióticos. La eficiencia fotosintética en plantas no micorrizadas disminuyó bajo todos los estreses, pero en plantas micorrizadas se mantuvo similar en todos los tratamientos de estrés, mostrando su mitigación. El peso seco aéreo y de raíces fue similar bajo diferentes estreses abióticos en plantas no micorrizadas, pero en plantas micorrizadas fueron mayores y mostraron diferencias entre ellos. La peroxidación lipídica en plantas no micorrizadas aumentó en condiciones de sequía y salinidad, mientras que en plantas inoculadas se mantuvo constante, mostrando mitigación. Las plantas inoculadas incrementaron las actividades de superóxido dismutasa (SOD) y ascorbato peroxidasa (APX) en tallos y raíces bajo todos los estreses, mostrando defensa antioxidante. Catalasa tuvo un comportamiento variable. Conclusión:La micorrización tuvo una interacción positiva con diferentes parámetros fisiológicos y defensa antioxidante en M. sativa,y promovió mitigación ante el estrés abiótico.
Palabras clave: Antioxidantes, fotosíntesis, frio, hongos, prolina, salinidad, sequía.
Abstract: Objetive: This study investigated the effects of inoculation with Rhizofagus intraradices on physiological parameters and antioxidant defense in Medicago sativa plants under salt, drought and cold stress. Material and methods: M. sativa seeds, var. Dekalb (DK166), were disinfected and seeded in sterilized mixture of soil/sand. 10 g of R. intraradices was added to half of the pots and all plants received 5 ml of a culture of Shinorhizobium meliloti. They were grown in a greenhouse at 25/20°C (day/night), with 16 light, 50-60% relative humidity and regular watered during 45 days. After this time, were place the treatment of control, drought, salinity and cold. R. intraradices colonization was calculated in all treatments. Stomatal conductance was measured using a porometer and the efficiency of photosystem II (PE) was measured with Fluor Pen FP100. The shoot and root dry weight (DW), proline content, the oxidative damage to lipids and the enzyme activities of SOD, CAT, APX, were determined in all treatments after stress. Results: All physiological and enzymatic measurements were significative (t-Student) better in inoculated plants, showing a positive interaction mycorrhization-physiological status. Stomatal conductance, in inoculated plants, increased under drought and salinity, showing protection to these abiotic stresses. Photosynthetic efficiency in nonmycorrhizal plants decreased under all stresses, but in mycorrhizal plants, it remained similar in all stress treatment, showing their mitigation. The aerial and roots dry weights were similar under different abiotic stress in non-mycorrhizal plants, but in mycorrhizal plants, were higher and showed different from each stress. Lipids peroxidation in non-inoculated plants increased under drought and salinity conditions, while in inoculated plants remained constant showing mitigation. Inoculated plants increased activities of superoxide dismutase and ascorbate peroxidase in shoots and roots under all stresses showing antioxidant defense. Catalase had a variable performance. Conclusion:The mycorrhization have a positive interaction with different physiological parameters and antioxidant defense in M. sativa and promoted mitigation under abiotic stresses.
Keywords: Antioxidants, cold, drought, fungi, photosynthesis, proline, salinity.
Introduction
Alfalfa (Medicago sativa L.) is a fundamental resource for agricultural production in temperate regions of the world, based on its high nutritional quality, forage production, growth habit, persistence, plasticity and capacity for symbiotic fixation of atmospheric nitrogen (Basigalup et al., 2007).
Arbuscular mycorrhizal fungi (AMF) represent the most widespread symbiosis with land plants. The associated fungi colonize the plant roots and reside in the internal tissues of their host plant. This mutualistic association not only plays a key role enhancing plant growth by facilitating the uptake of water and essential nutrients but also protects the plant from adverse soil conditions. The application of mycorrhizal fungi is a promising alternative strategy for sustainable crop production under normal as well as biotic and abiotic stress conditions. The mycorrhizal plants have an improved ability for nutrient uptake and have ability to tolerate stress environments (Nadeem et al., 2017)
The abiotic stresses such as drought, salinity, and extreme temperature regimes are linked by the fact that they all decrease the availability of water and nutrients to plant cells. The production of crops and their sustainability is mostly influenced by abiotic stress, such as drough (Barea et al., 2011), salinity (Al-Khaliel, 2010), extreme temperature (Canci and Toker, 2009).
Arbuscular mycorrhizal fungi (AMF) can promote plant growth increasing plants production under stress due to their benefits: establishments of extensive hyphal networks and secretion of biomolecules like glomalin, which ameliorates soil structure and enhances water and nutrient uptake (Pagano, 2011). One of the most used species to alleviate the biotic stress in plants and increase their yields is R. intraradices.
R. intraradices inoculation improved the shoot and root dry weights, total root length, root surface area, root volume, and the number of root forks under heavy metal stress, in Robinia pseudoacacia (Zhang et al., 2020) and increasing abiotic stress tolerance in Digitharia eriantha (Pedranzani et al., 2016) and increased the essential oil yield and dry biomass in aromatic plant (Colombo et al., 2013).
Under stress conditions, AMF can modify plant physiology in order to cope with those environmental factors (Miransari, 2010). Several reports have shown that mycorrhizal symbiosis improves plant health through increased protection against environmental stresses, either biotic (e.g., pathogen attack) or abiotic (e.g., drought, salinity, heavy metals, organic pollutants) (Azcón and Barea, 2010).
Plant stress responses are dynamic and involve complex cross-links among different regulatory levels, including adjustment of metabolism and gene expression for physiological and morphological acclimation (Krasensky and Jonak, 2012). During abiotic stress, different metabolic pathways are uncoupled and electrons are transferred to molecular oxygen to form reactive oxygen species (ROS), such as superoxide radicals (O2•−), hydrogen peroxide (H2O2), or hydroxyl radicals (OH•) (Noctor et al., 2014). The scavenging of ROS is achieved through the action of different enzymatic and non-enzymatic compounds, including superoxide dismutase (SOD), glutathione reductase (GR), catalase (CAT), ascorbate peroxidise (APX), and the enzymes of the ascorbate-glutathione pathway. Non-enzymatic mechanisms include compounds, such as ascorbic acid, glutathione, and α-tocopherol, capable of directly scavenging several ROS (Scheibe and Beck, 2011). Abiotic stress symptoms include plant wilting, reductions in the rate of net photosynthesis, reductions in stomatal conductance, and the gradual decrease in total chlorophyll content and biomass.
The aim of this study was to determine the effects of the symbiotic association of Medicago sativa and the mycorrhiza R. intraradices in relation to the physiological parameters and antioxidant response under different abiotic stresses.
Materials and methods
Treatments and experimental design
Eight treatments were compared under a randomized complete block design with five replications. Treatments came from the combination of two factors: (1) inoculated and non-inoculated (control) plants with Rhizophagus intraradices, (2) four growing conditions non-stressed (control), and any of three stresses drought, salt, or cold. The experimental unit was a pot as seen in figure 1.

Figure 1
Experimental units (pots) lay-out
Soil and biological material
A loamy soil was collected from the ground at the Zaidin Experimental Station (Granada, Spain). The soil had a pH of 8.1 (measured in water, 1:5 w/v); 1.5% organic matter; nutrient concentrations (g/kg): N, 1.9; P, 1 (NaHCO3-extractable P); K, 6.9. The soil was sieved (5 mm), diluted with quartz-sand (2 mm) (1:1, soil: sand, v/v), and sterilized by steaming (100 ºC for 1 h on 3 consecutive days).
M. sativa seeds, var. Dekalb (DK166., were disinfected with 70% ethanol for 3 min and rinsed three times with sterile water. One seed per pot was sown in 500 ml pots containing the sterilized mixture of soil/sand.
Mycorrhizal inoculum of R. intraradices (Schenck and Smith) strain EEZ 58 (Ri) was prepared as described by Porcel et al. (2006) and 10 g of the inoculum was added to half of the pots at the time at sowing. All plants received 5 ml of a culture of Shinorhizobium meliloti, 106 cell per ml.
Growing conditions
All plants were grown in a greenhouse at 25/20 °C (day/night), with 16 light, 50-60% relative humidity, an average photosynthetic photon flux density of 800 µmol m-2 s-1, (LI-COR, Lincoln, NE, USA, model LI-188B) and watered to field capacity for 45 days.
Stress conditions were applied after the 45-day mentioned above, protocol for each stress condition 1-25 °C and soil at field capacity; 2-25 ºC and 60% of field capacity for one week (drought); 3-25 ºC and soil irrigated with 200 mMNaCl for two weeks (salinity); 4-4 °C for 72 h (cold) continuously (Pedranzani et al., 2016). Before harvest, stomata conductance and photosynthetic efficiency were taken. At the end of each stress, the plants were harvested and the weights were taken and the samples were placed in liquid nitrogen for enzymatic measurements.
Mycorrhizal development
R. intraradices colonization was estimated by visual inspecting of fungal structures after treatments with different stresses, clearing of roots in 10% KOH and staining with 0.05% (w/v) trypan blue in lactic acid according to Philips and Hayman (1970). The percentage of R. intraradices colonization was calculated according to the gridline intersects method (Giovannetti and Mosse, 1980).
Stomatal conductance, photosynthetic efficciency and aerial and root dry weight
Stomatal conductance was measured using a porometer (Porometer AP4, Delta-T Devices Ltd., Cambridge, UK). The efficiency of photosystem II (PE) was measured with Fluor Pen FP100 (Photon Systems Instruments, Brno, Czech Republic). Fluor Pen quantifies the quantum yield of photosystem II as the ratio between the actual fluorescence yield in the light-adapted state (F′V) and the maximum fluorescence yield in the lightadapted state (FM), according to Oxborough and Baker (1997) both were measured in five different plants per treatment. After finishing the treatments, aliquots of aerial and root tissue were extracted and place in an oven at 70 C° for 48 h for shoot and root dry weight (DW) measured.
Proline content
Free proline content was quantified according to the method of Bates et al. (1973) and was measured in five different plants per treatment.
Oxidative damage to lipids
500 mg of leaves were ground with mortar on ice with 6 mL of 100 mM of potassium phosphate buffer (pH7) and the homogenate was filtered with Miracloth layer and centrifuged at 15 000 g for 20 min. The chromogen was formed by mixing 200 µL of supernatants with 1 mL of a reaction mixture containing 15% (w/v) trichloroacetic acid (TCA), 0.375% (w/v) 2-thiobarbituric acid (TBA), 0.1% (w/v) butylhydroxytoluene and 0.25 N HCl, incubating the mixture at 100 °C for 30 min (Minotti and Aust, 1987). After cooling at room temperature, tubes were centrifuged at 800 g for 5 min and the supernatant measurement at 532 nm in the spectrometer. Lipid peroxidation was estimated as the content of 2-thiobarbituric acid-reactive substances (TBARS) and expressed as equivalents of malondialdehyde (MDA) according to Halliwell and Gutteridge (1989).
Determination of enzyme activities
Among the enzymes known to be involved in ROS detoxification, SOD, CAT, APX were selected and their activities were determined in shoots and roots of all treatments.
Oneg of leaves or roots, with 50 mg polyvinylpolypyrrolidone (PVPP) and 10 ml of 50 mM K-phosphate buffer (pH 7.8) at 0-4 ºC were used for enzyme extraction, containing 0.1 ml EDTA for superoxide dismutase (SOD), catalase (CAT), and ascorbate peroxidase (APX). Extracts were filtered through four layers of nylon cloth and centrifuged at 20 000 gfor 20 min at 0-4 ºC. The supernatants were kept at -70 ºC for subsequent enzymatic assays. Total SOD activity (EC 1.15.1.1) was measured according to Beyer and Fridovich (1987), based on the ability of SOD to inhibit the reduction of nitrobluetetrafolium (NBT) by superoxide radicals generated photochemically. One unit of SOD was defined as the amount of enzyme required to inhibit the reduction rate of NBT by 50% at 25 ºC. CAT activity (EC 1.16.1.6) was measured by the disappearance of H2O2 (Aebi, 1984). The reaction mixture (3 ml) contained 10.6 mM H2O2. The reaction was initiated by adding 25 µl of the extract and monitoring the change in absorbance at 240 nm and 25 °C for 3 min. APX activity (EC 1.11.1.11) was measured in a 1 ml reaction volume containing 50 mM potassium phosphate buffer (pH 7.0), 0.1 mM H2O2 and 0.5 mM ascorbate. Adding the H2O2started the reaction and the decrease in absorbance at 290 nm was recorded for 1 min to determine the oxidation rate of ascorbate (Amako et al., 1994).
Statistical analysis
First, a t-test for independent samples (data were homoscedastic and normal) was performed to observe the correlation among measured variables and the two types of plants: inoculated and not inoculated. ANOVA's were carried out to identify the influence of the four growing conditions on measured variables, mean multiple comparisons analyses were done when a significant effect was found. Data manipulation and analysis were done by SPSS (Statistical Pack for Social Sciences) version 27.
Results
Mycorrhizal development
The application of different stresses did not affect root colonization. The mycorrhization percentage of R. intraradices to M. sativa plants was 60% in non-stress plants; 59.3% in drought stress plants; 66.6% in salinity and 63.8% in salinity and cold stress respectively.
Mycorrhizal and non-mycorrhizal plants interaction
A t-test for independent samples (data were homoscedastic and normal) was performed to observe the correlation between the study variables and the two groups: mycorrhizal (AMA) and non-mycorrhizal (NM). For all the parameters (variables) studied, the difference between AM and NM plants was significant, being a positive interaction.
Stomatal conductance, photosynthetic efficiency and aerial and root dry weight
Stomatal conductance increased considerably with AM colonization in all conditions in comparison with NM plants. In AM plants, drought and salinity stress increased stomata conductance and cold stress remained similar to non-stressed plants. In NM plants decreased by drought and cold stress (figure 2).
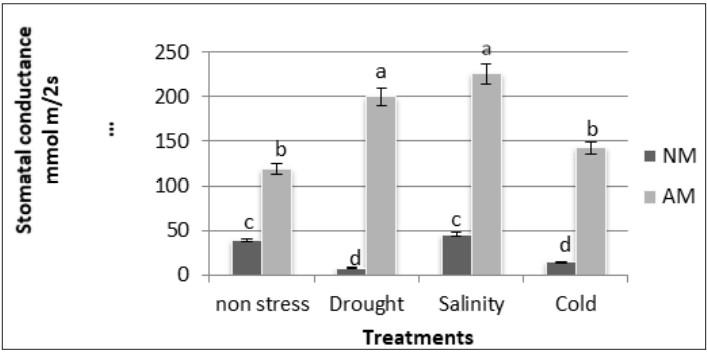
Figure 2
Stomatal conductance (mmol m/2s) in AM and NM plants of M. sativa under non-stress and drought, salinity and cold conditions
Different letters mean significant differences (P ≤ 0.05) as determined by the ANOVA test (n=5)
In NM plants, the photosynthetic efficiency decreased in salinity, drought and cold stress compared with non-stressed plants. The photosynthetic efficiency was not affected by abiotic stress in AM plants and it remained similar in all treatment respect the control (figure 3).
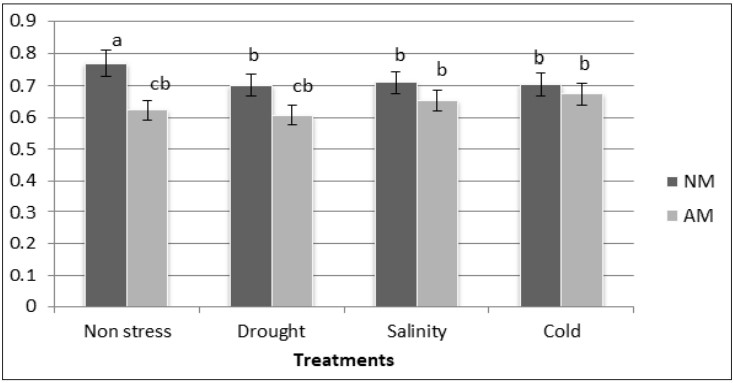
Figure 3
Photosynthetic efficiency in AM and NM plants of M. sativa under non-stress and drought, salinity and cold conditions
Different letters mean significant differences (P ≤ 0.05) as determined by ANOVA test (n=5)
The DW of shoots and roots of M. sativa plants varied in relation to colonization, thus AM plants increased DW, in relation to those that were NM (figure 4a and b). Aerial and roots DW increased in non-stressed, drought and salinity condition. Cold showed similar behavior in NM and AM plants, both aerial and roots DW (figure 4a and b).

Figure 4
Photosyntetic efficiency aerial and roots DW in AM and NM plants of M. sativa under non-stress and drought, salinity and cold.
Different letters significant mean differences (P ≤ 0.05) as determined by ANOVA test (n=5)
Proline
There is an interaction between mycorrhization and proline production under abiotic stress in M.sativa plants. Proline contents increased in NM and AM plants with drought and salinity stress in contrast with the control, and these changes were greater in AM plants compared with NM plants (figure 5a, b). Proline decreased under cold stress in NM and AM plants; in addition, there was mycorrhizal-proline interaction, in which it increased in AM plants concerning NM plants (figure 5c). This shows that proline was a protective osmolyte against drought and salinity being the protection greater, in AM plants.
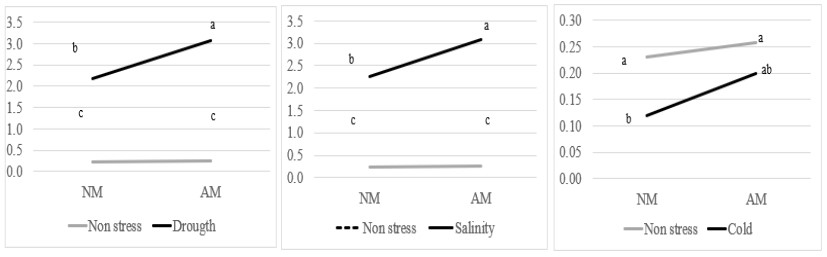
Figure 5
Determination of proline content (µmol/g FW) in M. sativa NM and AM plants under non-stress and drought (a), salinity (b) and cold (c) conditions
Oxidative damage to lipids (MDA)
Under non-stress conditions, Malondialdehyde (MDA) was significantly higher in AM plants than in NM plants. The MDA content increased significantly in shoots of NM plants when subjected to drought, salinity and, cold. In contrast, in shoots of AM plants, there were no significant differences between non-stressed plants and those subjected to abiotic stress (figure 6), showing that lipid peroxidation, as an alarm element against abiotic stress, is not necessary for the presence of symbiosis with mycorrhizae.
Antioxidant enzymes
In roots and shoots, SOD activity was significantly higher in AM plants with respect to NM plants under non-stress, drought, salinity and cold conditions (table 1). In roots and shoots from AM plants, CAT activity was elevated compared to NM plants in control and cold condition. In shoots, CAT activity decreased under drought and salinity stress in AM plants as compared with NM plants (table 1). APX activity was always significantly higher in AM plants than in NM plants in both roots and shoots tissues under the entire conditions tested (table 1).
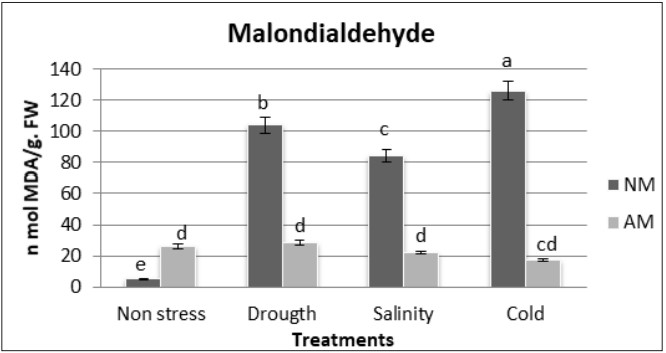
Figure 6
Oxidative damage to lipids (nmol MDA/g FW) in shoots (a) of nonmycorrhizal (NM) and mycorrhizal (AM) M. sativa plants under non-stress, drought, salt, or cold stress conditions
Different letters mean significant differences (P ≤ 0.05) as determined by the ANOVA test (n=5)
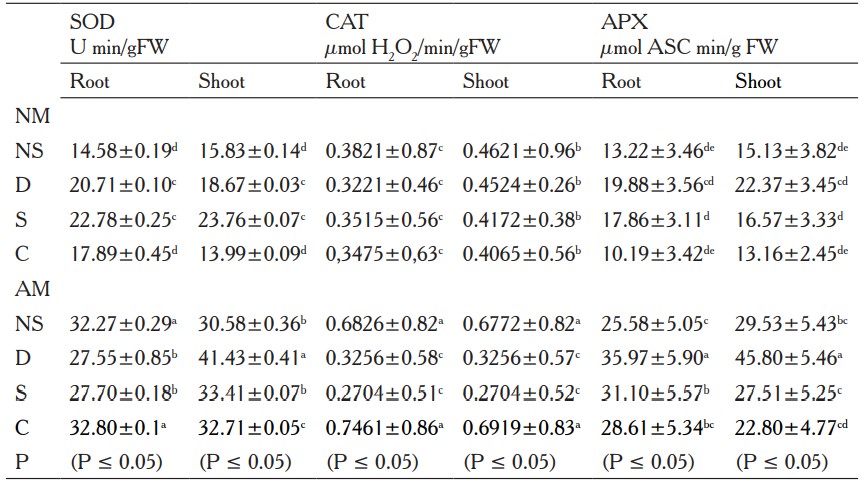
NS= Non stress, D= Drought, S= Salinity, C=Cold.
Columns represent means ±Standard error.
Different letters mean significant differences (P ≤ 0.05) undetermined by the analysis of variance (ANOVA) test (n=5).
Discussion
The values for colonization observed in M. sativa plants were similar than in other species (Abdel Latef and Chaoxing, 2011; Porcel and Ruiz-Lozano, 2004; Ruiz-Sánchez et al., 2010).
M. sativa plants inoculated with R. intraradices had higher DW than NM plants under control and all abiotic stress conditions and these results were consistent with those in maize (Feng et al., 2002), fenugreek (Evelin and Kapor, 2014), and Pigeon pea (Garg and Manchanda, 2009) under salt stress. Wu and Xia (2006) found that Citrus tangerine plants colonized by Glomus versiforme increased the DW compared with NM plants under similar drought stress conditions.
Stomatal conductance is regulated primarily by the aperture of the stomata pore and stomata density (Yan et al., 2012; Huang and Xu, 2015; Gamage et al., 2018). In this study stomatal conductance increased in AM plants under control, drought, salt and cold stresses compared with NM plants. AM symbiosis often alters the stomata behavior of the host (Augé et al., 2016; Chitarra et al., 2016). Smith and Read (2008) explained that the ability of AM plants to maintain stomata opening longer than NM plants as soils dry is because hyphae penetrate pores that are inaccessible to roots, and they spread beyond the root zone, effectively increasing the available volume of the soil solution. There is evidence that AM root systems can better exploit bound water in drying soils, in some cases providing access to soil water below the permanent wilting Ψ of NM plants (Augé et al., 2015). In NM plants, stomatal conductance decreased under drought and cold. Changes in stomatal conductance are always accompanied by changes in leaf water potential (Ψ) or osmotic adjustment. The influence of AM on stomatal conductance may also be associated with altered chemical signals and carbon dynamics of the plant leaves (Ruiz-Lozano and Aroca, 2010).
The photosynthetic efficiency remained unchanged in AM plants under different conditions, however decreased significantly with the drought, salt, and cold stress in NM plants, in correlation with the stomatal closure. A positive correlation between tolerance to abiotic stresses in AM plants and maintenance of photosystem II efficiency has been demonstrated, which, in turn, maintains (Porcel and Ruiz-Lozano, 2004) or even increases the productivity of the plant (Ruiz-Sánchez et al., 2010). In M. sativa AM plants, photosystem II efficiency under stress conditions remain similar to the unstressed control values because AM plants maintained higher water status and hence more opened stomata (Augé et al., 2015).
In order to tolerate abiotic stress, plants accumulate a high concentrations of low molecular mass organic solutes, such as soluble sugars, proline and other amino acids, to regulate the osmotic potential of cells aiming at improving absorption of water under drought stress (Zang et al., 2010). Our data indicate that the concentration of proline in leaves increased during drought and saline stress in both AM and NM-treated plants. The results are in agreement with previous reports of drought stress (Zang et al., 2010; Ruiz-Lozano and Aroca, 2010).
Abiotic stress is accompanied by the formation of ROS, such as superoxide radicals (O2-) and H2O2, that damage membranes and macromolecules and the oxidation of membrane lipids is an indication of uncontrolled free-radical production and oxidative stress (Noctor et al., 2014). Accordingly, the number of lipid peroxides was quantified in shoots of M. sativa, the lipid peroxidation in AM plants subjected to drought, salinity, and cold was 77, 81, and 85.6% lower than NM plants respectively, indicating a clear defense of the plant against stress by the AM symbiosis. These were observed in tomato plants subjected to salinity (Abdel Latef and Chaoxing, 2011) and in soybean and rice plants under drought stress conditions (Porcel and Ruiz-Lozano, 2004; Ruiz-Sánchez et al., 2010).
As a defense mechanism, the activity of antioxidant enzymes such as ascorbate peroxidase (APX), catalase (CAT), and superoxide dismutase (SOD) is enhanced under drought (Sankar et al., 2007; Manivannan et al., 2008), salinity (Jaleel et al., 2008), cold (Garbero et al., 2011) and oxidative stress (Pérez Chaca et al., 2014). In the present study, APX activities of M. sativa inoculated with R. intraradices increased significantly as compared to NM plants, whether host plants were under well-watered, and drought, salinity, or cold conditions. The behavior of SOD was identical to that of APX, increasing its activity in all treatments of mycorrhizal plants. The behavior of CAT was similar, but only for unstressed and cold treatment. Previous research has also reported increases in SOD, CAT, and APX activity in AM tomato plants subjected to saline stress (Abdel Latef and Chaoxing, 2011; Abbaspour et al., 2012) and in Digitaria eriantha, were CAT, APX and SOD showed significant increases in AM plants as compared to non-AM plants under salinity, drought and cold stress (Pedranzani et al., 2016). CAT and APX activities are both involved in the scavenging of hydrogen peroxide, although APX has a much higher affinity for H2O2 than CAT (Estrada et al., 2013). CAT activity was lower in the roots and shoots of AM plants subjected to drought and salinity than AM plants without stress, which may indicate that in M. sativa in these conditions, hydrogen peroxide could be preferentially scavenged by APX. Indeed, in these plants, APX activity was significantly higher than in control plants not subjected to stress.
Conclusion
Alfalfa var. Dekalb 166, which was considered moderately tolerant to drought and salinity stresses, associated with mycorrhizae, presents many antioxidant defense mechanisms that strongly prevent possible damage from drought and salinity, and cold improved performance and physiological parameters.
Acknowledgments
We thank Ministerio de Economía y Competitividad (Project AGL2014-53126-R), Junta de Andalucía, Spain (Project P11-CVI-7107) for supporting this research and Universidad Nacional de San Luis, Argentine (Research Project of Secretary of Science and Technology PROICO: N 02-3318 (Resol. CD126/18).
Cited literature
Abbaspour, H.; Saeidi-Sar, S.; Afshari, H. and Abdel-Wahhab, M. A. (2012) Tolerance of Mycorrhiza infected pistachio (Pistaciavera L.) seedling to drought stress under glasshouse conditions. J. Plant Physiol. 169: 704-709.
Abdel Latef, A. and Chaoxing, H. (2011) Effect of arbuscular mycorrhizal fungi on growth, mineral nutrition, antioxidant enzymes activity and fruit yield of tomato grown under salinity stress. Sci. Hortic. 127: 228-233.
Aebi, H. (1984). Catalase in vitro. Methods in Enzymology. 105: 121-126.
Al-Khaliel, A. S. (2010). Effect of salinity stress on mycorrhizal association and growth response of peanut infected by Glomus mosseae. Plant Soil Environ. 56: 318-324.
Amako, K.; Che, G. X. and Asada, K. (1994). Separate assays specific for ascorbate peroxidase and guaiacol peroxidase and for the chloroplastic and cytosolic isoenzymes of ascorbate peroxidase in plants. Plant Cell Physiol. 35: 497-504.
Augé, R. M.; Toler, H. D., and Saxton, A. M. (2015). Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: A meta-analysis. Mycorrhiza. 25: 13-24.
Augé, R. M.; Toler, H. D. and Saxton, A. M. (2016). Mycorrhizal stimulation of leaf gas exchange in relation to root colonization, shoot size, leaf phosphorus nitrogen: A quantitative analysis of the literature using meta-regression. Front Plant Sci. 7: 1-16.
Azcón, R. and Barea, J. M. (2010). Mycorrhizosphere interactions for legume improvement. In: Khan, M. S.; Zaidi, A. and Musarrat, J. (Eds.), Microbes for Legume Improvement. Springer, Vienna, Austria. Pp. 237-271.
Basigalup, D. H.; Rossanigo, R. and Ballario, M. V. (2007). Panorama actual de la alfalfa en Argentina. El cultivo de alfalfa en Argentina. Ediciones INTA. Buenos Aires, Argentina. 479 p.
Barea, J. M.; Palenzuela, J.; Cornejo, P.; Sánchez-Castro, I.; Navarro-Fernández, C.; Lopéz-García, A.; Estrada, B.; Azcón, R.; Ferrol, N. and Azcón-Aguilar, C. (2011). Ecological and functional roles of mycorrhizas in semi-arid ecosystems of Southeast. Spain Journal of Arid Environments.75: 1292-1301.
Bates, L. S.; Waldren, R. P. and Teare, I. D. (1973). Rapid determination of free proline for water-stress studies. Plant Soil. 39: 205-207.
Beyer, W. F. and Fridovich, I. (1987). Assaying for superoxide dismutase activity: Some large consequences of minor changes in conditions. Ann. Biochem. 161: 559-566.
Canci, H. and Toker, C. (2009). Evaluation of yield criteria for drought and heat resistance in chickpea (Cicer arietinum L.). J Agron. Crop. Sci. 195: 47-54.
Chitarra, W.; Pagliarani, C.; Maserti, B.; Lumini, E.; Siciliano, I.; Cascone, P.; Schubert, A.; Gambino, G.; Balestrini, R. and Guerrieri, E. (2016). Insights on the impact of arbuscular mycorrhizal symbiosis on tomato tolerance to water stress. Plant Physiol. 171: 1009-1023.
Colombo, R.; Fernández diPardo, A.; Fernández Bidondo, L.; van Baren, C.; and Godeas, A. (2013). Differential effects of two strains of Rhizophagus intraradiceson dry biomass and essential oil yield and composition inCalaminthanepeta. Revista Argentina de Microbiología. 45(2): 114-118.
Estrada, B.; Aroca, R.; Barea, J. M. and Ruíz-Lozano, J. M. (2013). Native arbuscular mycorrhizal fungi isolated from a saline habitat improved maize antioxidant systems and plant tolerance to salinity. Plant Sci. 201-202: 42-51.
Evelin, H. and Kapoor, R. (2014). Arbuscular mycorrhizal symbiosis modulates antioxidant response in salt stressed Trigonella foetum-gracum plants. Mycorrhiza. 24: 197-208.
Feng, G.; Zhang, F. S.; Li, X. L.; Tian, C. Y.; Tang, C. and Rengel, Z. (2002). Improved tolerance of maize plants to salt stress by arbuscular mycorrhiza is related to higher accumulation of soluble sugars in roots. Mycorrhiza. 12: 185-190.
Gamage, D.; Thompson, M.; Sutherland, M.; Hirotsu, N.; Makino, A. and Seneweera, S. (2018). New insights into the cellular mechanisms of plant growth at elevated atmospheric carbon dioxide concentrations. Plant Cell Environ. 41: 1233-1246.
Garbero, M.; Pedranzani, H. E.; Zirulnik, F.; Molina, A.; Pérez-Chaca, M. V.; Vigliocco, A. and Abdala, G. (2011). Short term cold stress in two cultivars of Digitaria eriantha: Effects on stress-related hormones and antioxidants defense system. Acta Phys Plant. 33: 497-507.
Garg, N. and Manchanda, G. (2009). Role of arbuscular mycorrhizae in the alleviation of ionic, osmotic and oxidative stresses induced by salinity in Cajanus cajan (L.) Mill sp. (Pigeonpea). J. Agron. Crop. Sci. 195: 110-123.
Giovannetti, M. and Mosse, B. (1980). An evaluation of techniques for measuring vesicular-arbuscular infection in roots. New Physiol. 84: 489-500.
Halliwell, B. and Gutteridge, J. M. C. (1989). Free radicals in biology and medicine. 2nd Ed. Claredon Press. Oxford, UK. 543 p.
Huang, B. and Xu, Y. (2015). Cellular and molecular mechanisms for elevated CO2 regulation of plant growth and stress adaptation. Crop Sci. 55: 1-20.
Jaleel, C. A.; Gopi, R.; Manivannan, P.; Gomathinayagam, M.; Murali, P. V. and Panneerselvam, R. (2008). Soil applied propiconazole alleviates the impact of salinity on Catharanthus roseus by improving antioxidant status. Pestic Biochem Physiol. 90: 135-139.
Krasensky, J. and Jonak, C. (2012). Drought, salt and temperature stress-induced metabolic rearrangements and regulatory networks. J. Exp Bot. 63: 1593-1608.
Manivannan, P.; Jaleel, C. A.; Somasundaram, R. and Panneerselvam, R. (2008). Osmoregulation and anti-oxidant metabolism in drought stressed Helianthus annuus under triadimef on drenching. C. R. Biol. 331: 418-425.
Minotti, G. and Aust, D. (1987). The requirement for iron (III) in the initiation of lipid peroxidation by iron (II) and hydrogen peroxide. J. Biol. Che. 262: 1098-1104.
Miransari, M. (2010). Contribution of arbuscular mycorrhizal symbiosis to plant growth under different types of soil stress. Plant Biology. 12: 563-569.
Nadeem, S.; Khan, M. Y.; Wagas, R.; Binyamin, R.; Akhtar, S. and Zahir, Z. A. (2017) Chapter: Arbuscular Mycorrhizas: An Overview. In: Wu, Qiang-Sheng (Eds.). Arbuscular Mycorrhizas and Stress Tolerance of Plants. 1er eds. Springer E-Book. Pp.1-24.
Noctor, G.; Mhamdi, A. and Foyer, C. H. (2014). The roles of reactive oxygen metabolism in drought: not so cut and dried. Plant Physiol. 164: 1636-1648.
Oxborough, K. and Baker, N. R. (1997). An instrument capable of imaging chlorophyll a fluorescence from intact leaves at very low irradiance and at cellular and sub cellular levels of organization. Plant Cell Environ. 20: 1473-1483.
Pagano, M. C. (2011). Soil tillage in agroforestry and agroecosystems: Mycorrhizal benefits. In: Miransari M. (Ed.). Soil Tillage and Microbial Activities. Research Signpost Publications, India. Pp. 65-84.
Pedranzani, H. E.; Rodríguez-Rivera, M.; Gutiérrez, M.; Porcel, R.; Hause, B. and Ruiz-Lozano, J. M. (2016). Arbuscular mycorrhizal symbiosis regulates physiology and performance of Digitaria eriantha plants subjected to abiotic stresses by modulating antioxidant and jasmonate levels. Mycorrhiza. 26: 141-152.
Phillips, J. M. and Hayman, D. S. (1970). Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 55: 158-161.
Pérez Chaca, M. V.; Rodríguez Serrano, M.; Molina, A.; Pedranzani, H.; Zirulnik, F.; Sandalio, L. and Romero Puertas, M. (2014). Cadmium induces two waves of reactive oxygen species in Glicine max L. roots. Plant Cell Environ. 37: 1683-1687.
Porcel, R.; Aroca, R.; Azcón, R. and Ruiz-Lozano, J. M. (2006). PIP aquaporin gene expression in Arbuscular mycorrhizal Glycine max and Lactuca sativa plants in relation to drought stress tolerance. Plant Mol Biol. 60: 389-404.
Porcel, R. and Ruiz-Lozano, J. M. (2004). Arbuscular mycorrhizal influence on leaf water potential, solute accumulation, and oxidative stress in soybean plants subject to drought stress. J. Exp. Bot. 55: 1743-1750.
Ruiz-Lozano, J. M. and Aroca, R. (2010). Host response to osmotic stresses: stomatal behavior and water use efficiency of arbuscular mycorrhizal plants. In: Koltai, Hinanit, Kapulnik, Yoram (Eds.). Arbuscular mycorrhizas: Physiology and Function. 2nd eds. Springer. E-book. Pp. 239-256.
Ruiz-Sánchez, M.; Aroca, R.; Muñoz, Y.; Polón, R. and Ruiz-Lozano, J. M. (2010). The arbuscular mycorrhizal symbiosis enhances the photosynthetic efficiency and the antioxidative response of rice plants subjected to drought stress. J. Plant Physiol. 167: 862-869.
Sankar, B.; Jaleel, C. A.; Manivannan, P.; Kishorekumar, A.; Somasundaram,R. and Panneerselvam, R. (2007). Effect of paclobutrazol on water stress amelioration through antioxidants and free radical scavenging enzymes. In Arachishypogaea L. Colloids Surf Biointerfaces. 60: 229-235.
Scheibe, R. and Beck, E. (2011). Drought, desiccation, and oxidative stress. In: Lüttge, U. et al. (Eds.). Plant desiccation tolerance, ecological studies. Springer Berlin, Germany. 209-231.
Smith, S. E. and Read, D. J. (2008). Micorrhizal Symbiosis. Academic Press 605. San Diego CA, USA. 800 p.
Wu, Q. S. and Xia, R. X. (2006). Arbuscular mycorrhizal fungi influence growth, osmotic adjustment and photosynthesis of citrus under well-watered and water stress conditions. J. Plant Physiol. 163: 417-425.
Yan, F.; Sun, Y.; Song, F. B. and Liu, F. L. (2012). Differential responses of stomatal morphology to partial root-zone drying and deficit irrigation in potato leaves under varied nitrogen rates. Sci Hortic. 145: 76-83.
Zang, Y.; Zong, C. L.; Chen, Y.; Jiang, Q. B.; Wu, C. and Pinyopusarerk, K. (2010). Improving drought tolerance of Casuarina equisetifolia seedlings by arbuscular mycorrizal under glass house conditions. New Forests. 40: 261-271.
Zhang, Q.; Gong, M.; Liu, K.; Chen, Y.; Yuan, J. and Chang, Q. (2020).Rhizoglomus intraradicesimproves plant growth, root morphology and phytohormone balance of Robinia pseudoacaciain Arsenic-Contaminated Soils.Front. Microbiol.11: 1428. doi: 10.3389/fmicb.2020.01428
Notas de autor