Artículos
Soybean pod and grain isoflavones accumulation during the filling period in field conditions
Soybean pod and grain isoflavones accumulation during the filling period in field conditions
RIA. Revista de Investigaciones Agropecuarias, vol. 47, núm. 3, pp. 338-343, 2021
Instituto Nacional de Tecnología Agropecuaria

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial 4.0 Internacional.
Recepción: 10 Noviembre 2020
Aprobación: 16 Diciembre 2020
Publicación: 01 Diciembre 2021
Abstract: The objective of this work was to determine the isoflavones accumulation in soybean pods and grains during the filling period in field conditions. Pods of the same phenological stage (marked in R5, i.e. beginning of grain filling period) were collected at R5, R6 (full grain), and R7 (beginning of physiological maturity) stages. At each analyzed stage, grains always exceed pods isoflavone concentrations. A decreasing tendency of isoflavone concentration was observed in pods towards the late grain filling. In this maternal tissue, no acetyl glucosides at any stage or aglycones at physiological maturity were detected. In grains, glucosides, malonyl glucosides, and total isoflavones increased and acetyl glucosides decreased from R5 to R7, whereas aglycones increased to R6, but then decreased. It is concluded that grains are the main accumulation tissue of these bioactive compounds, whereas pods seem to contribute to their accumulation in advanced stages of grain development.
Keywords: Glycine max, aglycones, glycosides, malonyl glycosides, acetyl glycosides, maternal tissue and seed isoflavones.
Resumen: El objetivo de este trabajo fue determinar la acumulación de isoflavonoides en vainas y granos de soja durante el período de llenado en condiciones de campo. Vainas de la misma fenología (marcadas en R5, es decir, inicio del llenado de granos) fueron colectadas en los estadíos R5, R6 (máximo tamaño de grano) y R7 (inicio de madurez fisiológica). En cada estadio analizado, los granos siempre superaron la concentración de isoflavonoides de las vainas. Se observó una tendencia decreciente de la concentración de isoflavonoides en las vainas hacia el llenado tardío de granos. En este tejido materno no se detectaron acetil glucósidos en ningún estadio ni agliconas a madurez fisiológica. En los granos los glucósidos, los malonil glucósidos y los isoflavonoides totales aumentaron y los acetil glucósidos disminuyeron de R5 a R7, mientras que las agliconas aumentaron a R6, pero luego disminuyeron. Se concluyó que los granos son el principal tejido de acumulación de estos compuestos bioactivos, mientras que las vainas parecen contribuir a su síntesis más tarde en el desarrollo de los granos.
Palabras clave: Glycine max, agliconas, glicósidos, malonil glicósidos, acetil glicósidos, isoflavonoides del tejido materno y del grano..
INTRODUCTION
Isoflavones, abundant in soybean meal, are physiologically active compounds with great nutraceutical value. The health benefits of isoflavones consumption are prevention of hormone-dependent cancers, cardiovascular diseases, osteoporosis, adverse menopausal manifestations and age-related cognitive decline (Pilsakova et al., 2010) and prostate cancer (Mahmoud et al., 2014). Although soybean grain isoflavone concentration is influenced by genotype, environment and their interaction (Carrera et al., 2014b), it is widely reported that, in the processed soybean meal, malonyl glucosides represent approximately 72% of the total isoflavones. Glucosides, acetyl glucosides and aglycones forms represent 20%, 6% and less than 4%, respectively (Wilson, 2004). Despite their low quantity, aglycones are proposed as responsible for the physiological activities attributed to the entire group of grain isoflavones (Izumi et al., 2000).
Isoflavones are ubiquitous natural organic compounds that display several physiological activities in soybean crops. For instance, they were shown to induce nodulation genes in Bradyrhizobium japonicum during the symbiosis process (Ferguson and Mathesius, 2003), and also are one of the main chemical defenses against insect attack (Zavala et al., 2015) or pathogen infection (Graham and Graham, 1996). Thus, large amounts of isoflavones were found in leaves, stems, and roots tissues, which would be channeled into sink tissues such as grains (Dhaubhadel, 2011). Indeed, an in vitro study revealed that although soybean embryos have the ability to synthesize isoflavones by themselves, the maternal tissues might contribute to the total grain isoflavones accumulation at maturity (Dhaubhadel et al., 2003). Therefore, information about the contribution of total isoflavones in grains derived from translocation of maternal tissues (pods) under field conditions still needs quantification (Carrera and Dardanelli, 2016).
The synthesis of isoflavones in soybean grains takes place 35–60 days after flowering, i.e. during the grain filling period (Kim and Chung, 2007; Medic et al., 2014). During this period many environmental factors (climatic variations, soil types) and the cropping management’s diversity (sowing date, plant density, genotypes, water regimes, among others) affect the final amount of these compounds in grains (Al-Tawaha and Seguin, 2006; Carrera and Dardanelli, 2016). Even when there are some studies addressing the changes of isoflavones from beginning of grain filling to physiological maturity, all of them are focused on grain and they present discrepancies regarding the patterns of accumulation followed by the different isoflavones conjugates in the grains (Carrão-Panizzi et al., 2019; Kim and Chung, 2007; Kim et al., 2006). Therefore, the objective of this study was to quantify isoflavones accumulation in soybean maternal tissue (pods) as well as on grains during the filling period in field condition. The information generated may be useful i) physiologically, to understand the deposition pattern of these health-promoting compounds in the fruit tissues during grain filling in field conditions, and ii) agronomically, to improve decisions related to harvest time in order to obtain specific raw material for the functional foods industry.
MATERIALS AND METHODS
An experiment was carried out at the field station of the National University of Lomas de Zamora (34º45ʹS, 58º29ʹW), under rainfed conditions. The soybean cultivar DM40R16 (Don Mario seeds) was sowed on the 29th of December 2017 on a typic Argiudoll soil (USDA Soil Taxonomy). This cultivar (of indeterminate growth habit and maturity group IV) is widely cropped in the Rolling Pampas. Inoculated seeds were hand-planted in three rows 0.52m apart and three meters long using a stand density of 35 plants/m2. The experimental field trial was laid out as a completely randomized design with three replications. At the beginning of grain filling (R5 phenological stage), 50 pods were hand-marked per plot (2-4 pods per plant) on the upper six nodes of the main stem. The selected pods contained grains of 3 mm length indicative of R5 stage (according to the scale of Fehr and Caviness 1977). Marked pods were collected at the stages R5, R6 (full grain) and R7 (beginning of physiological maturity), placed on ice in the field, and immediately after stored in the freezer (-20ºC). To ensure enough sample size, the marked pods were collected as follows: 25 pods per plot were harvested at R5 stage, 20 pods per plot were harvested at R6 stage, while 5 pods per plot were sampled at R7 stage. Meteorological data during the grain filling period, namely average daily maximum and minimum air temperature, as well as daily precipitation, were provided by the National University of Lomas de Zamora´s Meteorological Station. Mean air temperature was calculated as the average between maximum and minimum air temperature.
Quantitative analysis of isoflavones was carried out by ultra-high-performance liquid chromatography (UPLC, Waters, USA) equipped with a photodiode array detector according to Berhow (2002). The extraction was done separately in both tissues of the fruit, i.e. pod (without grains) and grain in triplicate according to Carrão-Panizzi et al. (2002). The isoflavones were eluted in a reverse phase column Acquity UPLC BEH C18 (Waters, Milford, MA, USA), the gradient system was compounded by methanol and water. Since the different isoflavones isomers vary in their molecular weights, isoflavones concentration were expressed as mg of isoflavones per 100 g of defatted samples on a dry weight basis. Total isoflavones were calculated as the sum of the 12 forms whose peaks were identified from 12 standards (Sigma Co and Fuji Co). The following variables were defined by adding the concentrations of their respective isomers, namely: aglycones = daidzein + genistein + glycitein; glycosides = daidzin + genistin + glycitein; malonyl glycosides = malonyl daidzin + malonyl genistin + malonyl glicitin, and acetyl glycosides = acetyl daidzin + acetyl genistin + acetyl glicitin. All determinations were made at the Physicochemical and Chromatographic Analysis Laboratory, located at EMBRAPA Soja, Londrina, Paraná, Brazil.
Chemical analyses were performed in the three replicates. The data obtained from the field trial was modeled according to a bifactorial experiment with tissues of the fruit and phenological stage as factors, using ANOVA and following the mentioned experimental design. The level of significance was 5%. The comparison of means was performed using Fisher’s least significant difference (LSD) test. From this analysis, the variance components were also calculated as a percentage (as part of total sums of squares) of the total variance for the four isoflavone conjugates (aglycones, glycosides, malonyl glycosides, acetyl glycosides) as well as the total isoflavones, accounted by the effects of the mentioned factors and their interaction. All calculations were done with the statistical software InfoStat (InfoStat, http://www.infostat.com.ar).
RESULTS
Average daily mean, maximum and minimum air temperature and daily precipitation during soybean grain filling period are shown in figure 1. The time elapsed between beginning grain filling (R5 stage) and physiological maturity (R7 stage) was 45 d, being within the mean duration for this region and soybean cultivar, during which the daily mean air temperature ranged was 15.1°C to 29.2°C, whereas the daily maximum air temperature ranged from 21.5°C to 36.3°C. Moreover, the proportion of the grain filling duration having a daily maximum temperature above 30°C was 49% of days and occurred mainly during the first half of this period. Precipitations during grain filling totalized 171mm, however, the first half of the period was fast dry with only one day of 23mm of rainfall, and with 60mm rain registered three days before physiological maturity (figure 1).
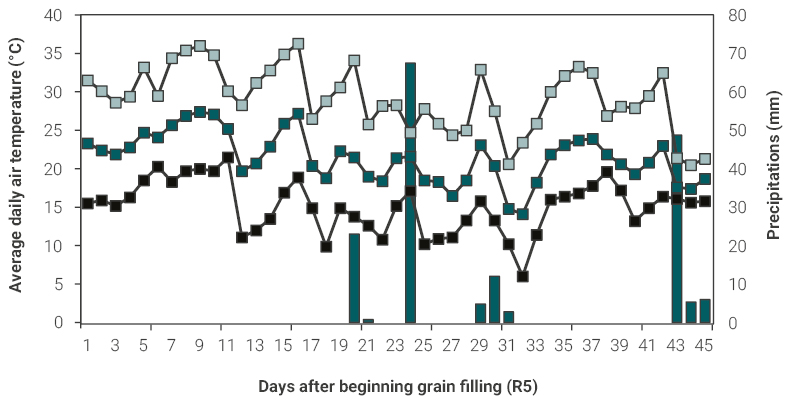
Figure 1
Average daily maximum (grey square), mean (green square), and minimum (black square) air temperature, and precipitations (grey bars) in field station of the National University of Lomas de Zamora (34º45 S, 58º29 W) during soybean grain filling period (R5-R7), growing season 2017-2018.
The analysis of variance (table 1) showed that tissues of the fruit, phenological stage, and tissues of the fruit × phenological stage interaction were significant (p < 0.0001) for all isoflavones, including the total concentration. Nevertheless, the tissues of the fruit were the most important source of variation for all of them, explaining up to 70% of the isoflavones total variability (table 1).
Interestingly, at the level of maternal tissue (pods), the major conjugated isoflavones, namely aglycones, glycosides, and malonyl glycosides decreased 100%, 73%, and 93% between R5 and R7 stages (figure 2A-C), resulting in a 91% decrease of total isoflavones concentration in this period (figure 2E). No presence of the acetyl glucosides conjugates was detected in any of the sampled stages (figure 2D) and of aglycones at physiological maturity (figure 2A).
In grains conjugates and the total isoflavones concentration varied significantly (P < 0.001, table 1) throughout the late reproductive development exhibiting different patterns of accumulation. For instance, glycosides and malonyl glycosides increased 192% and 270% between stages R5 and R7 (figure 2B-C) resulting in a 222% increase in total isoflavones, from 89.1 mg/100 g in R5 to 286.9 mg/100 g in R7 (figure 2E).
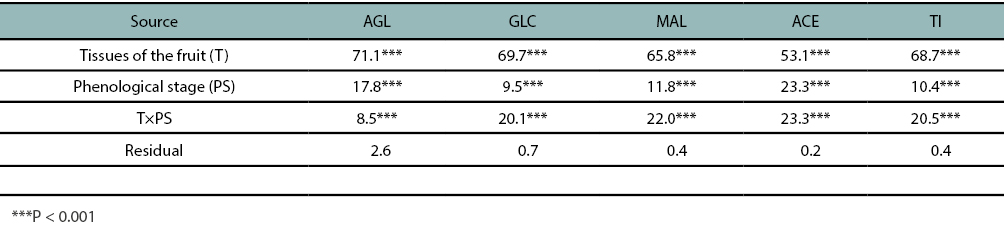
Table 1
Variance components as a percentage (as part of total sums of squares) of the total variance for aglycones (AGL), glucosides (GLC), malonyl glucosides (MAL), acetyl glucosides (ACE), total isoflavones (TI) (mg/100 g of defatted sample), accounted by the effects of the tissues of the fruit, phenological stage, and their interaction.
The conjugate forms aglycones represented less than 7% at R5 and R6, and 2% at R7 of grains total isoflavones, whereas acetyl glucosides accounted for less than 6% at R5 and less than ~ 0.5% of grains total isoflavones at R6 and R7 (figure 2A and 2D, respectively). Although representing a small proportion of the total isoflavones, we found that aglycones forms, responsible for the biological activity, increased 82% between R5 to R6 stages, then decreased steadily to R7 stage, reaching values similar to those at R5 (6.27 mg/100 g) (figure 2A). In contrast, acetyl glucosides showed an inverse trend to the rest of isoflavones, decreasing 74% from R5 to R6, and then remained unchanged until R7 (figure 2D).
DISCUSSION AND CONCLUSIONS
Results showed that grains always exceed pods isoflavone concentrations at each of the analyzed phenological stages (Figure 2A-E). Chennupati et al. (2012) observed that individual aglycones as well as their sum (considered by these authors the total isoflavones) were consistently greater in grains than in pods across the filling period. Nevertheless, it is important to note that we did not find studies addressing the accumulation dynamics extended to the other conjugated forms of isoflavones (glycosides, malonyl glycosides, and acetyl glucosides) in pods. The highest grain concentration compared to pods is expected, since developing grains become highly specialized storage tissues compared to maternal organs, which supply many products synthesized in different plant tissues to grains. In turn, this higher concentration of isoflavones in grains contributes to conferring this tissue, antioxidant properties and defense against stinkbug attacks for example.
Conjugates glycosides and MAL significantly increased throughout the whole filling period, resulting in a significant total isoflavones increases, which is expected since both conjugates accounted for 97.4% of total isoflavones at physiological maturity. Likewise, previous field studies by Kim and Chung (2007), Kim et al. (2006) and Carrão-Panizzi et al. (2019) reported malonyl glycosides and glycosides as the most abundant conjugates and also observed increases of both forms during grain development. Even though it has been found that malonyl glycosides are highly thermolabile compounds during the course of extraction (Kudou et al., 1991), the majority of soybean grain isoflavones accumulate in this form. This has been related to facilitation of glycoside isoflavones transport through the tonoplast into the cell vacuole where they are located, remaining unknown what stabilizes this form in the plant (Dhaubhadel, 2011).
Regarding the other two isoflavone conjugates, namely aglycones and acetyl glucosides, discrepancies emerge in the literature regarding their accumulation patterns in grains. Kim et al. (2006) found that these two conjugates increased during the filling period. Later studies also described this rising tendency during grain filling (Berger et al., 2008; Carrão-Panizzi et al., 2019; Chennupati et al., 2012; Kumar et al., 2009). The opposite trend was observed by Kim and Chung (2007), who found a decrease of aglycones and acetyl glucosides during grain development. Our results showed that the direction followed by both isoflavones conjugates varied across the filling period. While aglycones increased during the early grain filling (from R5 to R6 stages) and then decreased steadily to physiological maturity; acetyl glucosides showed an inverse trend to the rest of isoflavones, decreasing from R5 to R6, and then remaining unchanged until R7. According to the general theory, acetyl forms are degradative products generated from the malonate forms (heat unstable compounds), either in the grain or during the course of extraction and processing (Kim et al., 2006). Probably, the prevailing heat and drought conditions at the beginning of the filling (Figure 1) exposed immature grains to rising temperatures promoting degradation products of malonyl-glucose conjugates into acetyl glucosides explaining, at least in part, the higher acetyl glucosides levels at stage R5 compared to levels at stages R6 and R7 (figure 2D). In an advanced grain filling the increase of the other conjugates (malonyl glucosides, glucosides and aglycones) would have diluted acetyl glucosides concentrations to negligible amounts in the minimally processed grains. Regarding aglycones, this conjugate significantly increased between R5 and R6 (figure 2A), the most active period of isoflavones accumulation (Kumar et al., 2009), matching with the higher rate of glycosides and malonyl glycosides accumulation (figure 2B-C). Considering that aglycones synthesized via the phenylpropanoid pathway are then derivatized to the corresponding glucosides and malonyl glucosides (Kudou et al., 1991), then the higher the amount of aglycones, the greater the possibility of converting them into glycosides and malonyl glycosides and vice versa. The significant decrease of aglycones from R6 to R7 also matched with the decreasing increments of glycosides and malonyl glycosides conjugates in the mentioned stages. Besides, the decrease of aglycones during the late grain filling could be related to reductions in leaf primary metabolism towards the end of the cycle in a crop characterized by accelerated senescence processes and loss of leaves. This hypothesis is supported by the fact that isoflavones are secondary metabolites that need carbon skeletons synthesized by photosynthesis, given rise to carbon fluxes from the primary to the secondary metabolism (Caretto et al., 2015). Moreover, in a field study it was found that factors enhancing soybean photosynthetic rates such as cooler canopies (canopy temperature lower than air ones) with higher total leaf chlorophyll content were significantly related with higher grain levels of isoflavones (Carrera et al., 2015).
On the other hand, discrepancies regarding the patterns of accumulation followed by the different isoflavones conjugates in the grains among literature (Carrão-Panizzi et al., 2019; Kim and Chung, 2007; Kim et al., 2006) could be also related to the methodology followed when sampling. In previous studies at each phenological stage during the grain filling period, samples corresponded to pooled pods, whereas in our research we collected at each sample date marked pods of the same phenological age. This is an important difference since it is well documented that canopy position has a profound effect on soybean grain composition, due to the asynchrony between grains growth of different nodal positions in the plant (Huber et al., 2016; Bellaloui and Gillen, 2010).
Although the magnitude of the decrease in the proportions, between R5 and R7 stages, observed for most of isoflavone conjugates including the total at the level of maternal tissue (pods) was high (up to 100%), differences between both stages of the grain filling were not statistically significant. The lack of significance could be related to the great differences in all isoflavones concentration between pod and grain tissues, which were exacerbated at R7, indeed glycosides, malonyl glycosides and total isoflavones were 22-fold, 104-fold, 71-fold higher in grains respect to pods (figure 2). The decreased tendency of isoflavones in pods towards physiological maturity indicates translocation from the maternal tissues to the growing grains. Dhaubhadel et al. (2003) reported that the maternal tissue constitutes a substantial source of contribution to the total isoflavones concentration of grains in vitro. In control conditions data from Chennupati et al. (2012) limited to aglycones, also suggested that pods play a key role in the translocation of synthetized isoflavones and/or metabolic precursors into grains. It is particularly important that bioactive compounds such as the one analyzed in this study accumulate in grains, minimizing the rest that is wasted in the residual biomass (pods). On the other hand, the analysis of the isoflavone profile in both tissues showed that in grains as well as in pods the predominant forms were the malonyl glucosides conjugates followed by the glucosides conjugates. This characteristic was maintained through the different sampled phenological stages. Although from previous studies it is known that both conjugates are the predominant ones in soybean grains at maturity (Carrera et al., 2014a; Wang and Murphy, 1994; Wilson, 2004), these results would not be obvious in the case of pods isoflavone composition which as mentioned above, has been less explored. Indeed, to the best of our knowledge, this is the first report that described the dynamic of accumulation of all 12 isoflavones in soybean pods, and also the first that analyzed pods of the same phenological age marked at the beginning of the grain filling which represents an improvement in the methodology followed respect to previous research studies. Additional studies on the dynamics of accumulation expanding to other compounds should be carried out to provide a more comprehensive chemical composition characterization of fruit tissues during grain filling. This information will help growers to improve decisions related to harvest time in order to obtain a specific raw material for the functional foods industry, key for the majority of the population who uses this crop as a main nutrition source. Besides this study is also relevant for the edamame (immature soybean) production strategy, which is an imported vegetable in the world, especially in East Asia.
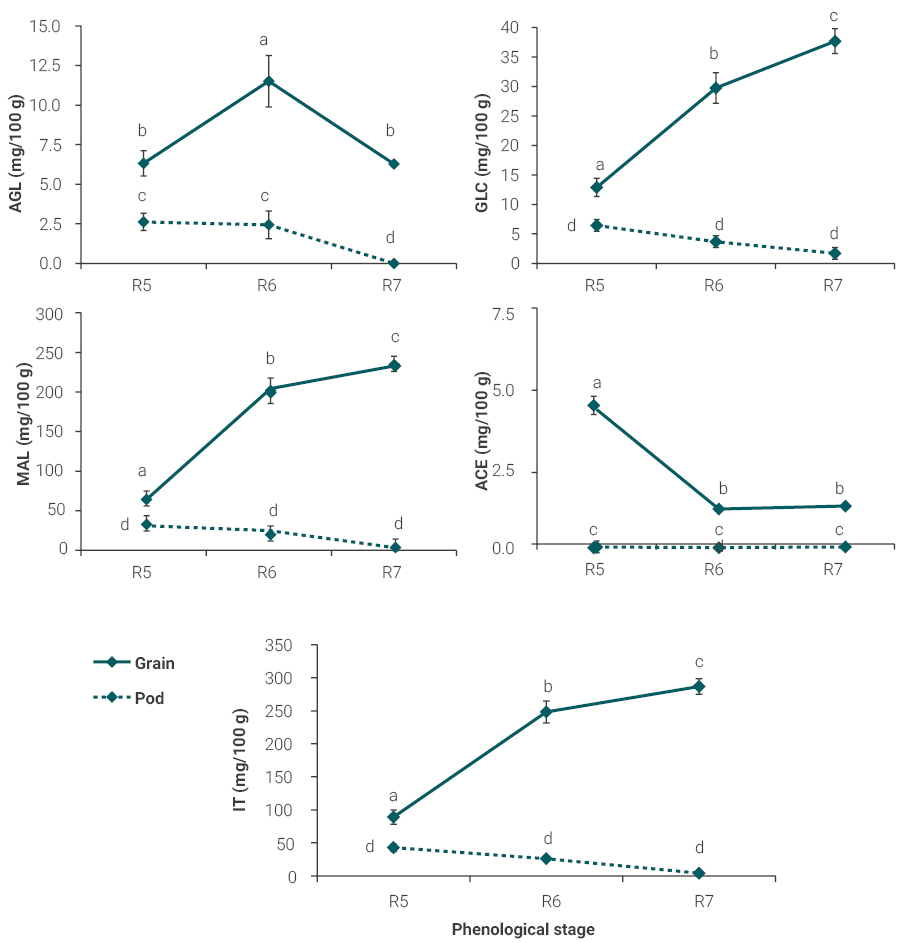
Figure 2
Concentration of the conjugated forms of isoflavones: A) aglycones (AGL), B) glycosides (GLC), C) malonyl glycosides (MAL), D) acetyl glycosides (ACE), and E) total isoflavones (TI) in pod (dashed line) and grain (continuous line) of the DM40R16 soybean cultivar. Determinations correspond to pods and grains harvested at the following phenological stages: R5 (beginning grain filling), R6 (full grain) and R7 (physiological maturity) according to Fehr and Caviness (1977). Values are expressed as mean ± SD (n = 3). Means with different letters were significantly different (P < 0.05).
Acknowledgments
To C. Fernández, M. Rosasco, F. García and Y. Ibañez (UNLZ) for technical assistance in field experiments. This study was supported by research grants from: CONICET, INTA, LOMASCYT and EMBRAPA.
C.S. Carrera and D.P. Rondanini are members of CONICET, the Scientific Research Council of Argentina.
REFERENCES
AL-TAWAHA, A.; SEGUIN, P. 2006. Seeding date, row spacing, and weed effects on soybean isoflavone concentrations and other seed characteristics. Canadian Journal of Plant Science 86, 1079-1087.
BELLALOUI, N.; GILLEN, A.M. 2010. Soybean seed protein, oil, fatty acids, N, and S partitioning as affected by node position and cultivar differences. Agricultural Sciences 1(03), 110.
BERGER, M.; RASOLOHERY, C.A.; CAZALIS, R.; DAYDÉ, J. 2008. Isoflavone accumulation kinetics in soybean seed cotyledons and hypocotyls: distinct pathways and genetic controls. Crop Science 48, 700-708.
BERHOW, M.A. 2002. Modern analytical techniques for flavonoid determination, Flavonoids in cell function. Springer, 61-76 pp.
CARETTO, S.; LINSALATA, V.; COLELLA, G.; MITA, G.; LATTANZIO, V. 2015. Carbon fluxes between primary metabolism and phenolic pathway in plant tissues under stress. International Journal of Molecular Sciences 16, 26378-26394.
CARRÃO-PANIZZI, M.C.; FAVONI, S.P.D.G.; KIKUCHI, A. 2002. Extraction time for soybean isoflavone determination. Brazilian Archives of Biology and Technology 45, 515-518.
CARRÃO-PANIZZI, M.C.; SILVA, B.D.S.; LEITE, R.S.; GODOY, R.L.D.O.; SANTIAGO, M.C.P.D.A.; FELBERG, I.; OLIVEIRA, M.C.N.D. 2019. Isoflavone, anthocyanin, and fatty acid contents of vegetable-type soybean grains at different maturity stages. Pesquisa Agropecuária Brasileira 54.
CARRERA, C.; CARRÃO PANIZZI, M.; GONTIJO MANDARINO, J.; LEITE, R.; ERGO, V.; ANDRADE, F.; PAROLA, R.; LASCANO, R.; VEGA, C. 2015. Relationship between soybean industrial-nutritional quality and the assimilate source under heat and water stress during seed filling. In: Proceedings of VII Congreso Brasilero de Soja - Mercosoja 2015. Florianopolis, Brasil.
CARRERA, C.S.; DARDANELLI, J.L. 2016. Changes in the relationship between temperature during the seed‐filling period and soya bean seed isoflavones under water‐deficit conditions. Journal of Agronomy and Crop Science 202, 421-432.
CARRERA, C.S.; DARDANELLI, J.L.; SOLDINI, D.O. 2014a. Chemical compounds related to nutraceutical and industrial qualities of non-transgenic soybean genotypes. Journal of the Science of Food and Agriculture 94, 1463-1469.
CARRERA, C.S.; DARDANELLI, J.L.; SOLDINI, D.O. 2014b. Genotypic and environmental variation in seed nutraceutical and industrial composition of non-transgenic soybean (Glycine max) genotypes. Crop and Pasture Science 65, 1311-1322.
CHENNUPATI, P.; SEGUIN, P.; CHAMOUN, R.; JABAJI, S. 2012. Effects of high-temperature stress on soybean isoflavone concentration and expression of key genes involved in isoflavone synthesis. Journal of Agricultural and Food Chemistry 60, 12421-12427.
DHAUBHADEL, S. 2011. Regulation of isoflavonoid biosynthesis in soybean seeds, Soybean-Biochemistry, Chemistry and Physiology. IntechOpen.
DHAUBHADEL, S.; MCGARVEY, B.D.; WILLIAMS, R.; GIJZEN, M. 2003. Isoflavonoid biosynthesis and accumulation in developing soybean seeds. Plant molecular biology 53, 733-743.
FEHR, W.R.; CAVINESS, C.E. 1977. Stages of soybean development. Iowa State University of Science and Technology Ames, Iowa.
FERGUSON, B.J.; MATHESIUS, U. 2003. Signaling interactions during nodule development. Journal of Plant Growth Regulation 22, 47-72.
GRAHAM, T.L.; GRAHAM, M.Y. 1996. Signaling in soybean phenylpropanoid responses (dissection of primary, secondary, and conditioning effects of light, wounding, and elicitor treatments). Plant Physiology 110, 1123-1133.
HUBER, S.C.; LI, K.; NELSON, R.; ULANOV, A.; DEMURO, C.M.; BAXTER, I. 2016. Canopy position has a profound effect on soybean seed composition. PeerJ 4, e2452.
IZUMI, T.; PISKULA, M.K.; OSAWA, S.; OBATA, A.; TOBE, K.; SAITO, M.; KATAOKA, S.; KUBOTA, Y.; KIKUCHI, M. 2000. Soy isoflavone aglycones are absorbed faster and in higher amounts than their glucosides in humans. The Journal of nutrition 130, 1695-1699.
KIM, J.A.; CHUNG, I.M. 2007. Change in isoflavone concentration of soybean (Glycine max L.) seeds at different growth stages. Journal of the Science of Food and Agriculture 87, 496-503.
KIM, S.-L.; BERHOW, M.A.; KIM, J.-T.; CHI, H.-Y.; LEE, S.-J.; CHUNG, I.-M. 2006. Evaluation of soyasaponin, isoflavone, protein, lipid, and free sugar accumulation in developing soybean seeds. Journal of Agricultural and Food Chemistry 54, 10003-10010.
KUDOU, S.; FLEURY, Y.; WELTI, D.; MAGNOLATO, D.; UCHIDA, T.; KITAMURA, K.; OKUBO, K. 1991. Malonyl isoflavone glycosides in soybean seeds (Glycine max Merrill). Agricultural and Biological Chemistry 55, 2227-2233.
KUMAR, V.; RANI, A.; DIXIT, A.K.; BHATNAGAR, D.; CHAUHAN, G. 2009. Relative changes in tocopherols, isoflavones, total phenolic content, and antioxidative activity in soybean seeds at different reproductive stages. Journal of Agricultural and Food Chemistry 57, 2705-2710.
MAHMOUD, A.M.; WANG, W.; BOSLAND, M.C. 2014. Soy isoflavones and prostate cancer: A review of molecular mechanisms. Journal of Steroid Biochemistry and Molecular Biology 140, 116-132.
MEDIC, J.; ATKINSON, C.; HURBURGH JR, C.R. 2014. Current knowledge in soybean composition. Journal of the American Oil Chemists’ Society 91, 363-384.
PILSAKOVA, L.; RIECANSKÝ, I.; JAGLA, F. 2010. The physiological actions of isoflavone phytoestrogens. Physiological Research 59, 651.
USDA. 2019. United States Department of Agriculture. World Agricultural Supply and Demand Estimation. WASDE-566. 40p. (Available at: http://www.usda.gov/oce/commodity/wasde/latest.pdf verified: October 25th 2019).
WANG, H.J.; MURPHY, P.A. 1994. Isoflavone composition of American and Japanese soybeans in Iowa: effects of variety, crop year, and location. Journal of Agricultural and Food Chemistry 42, 1674-1677.
WILSON, R.F. 2004. Seed composition. In: STEWART, B.A.; NIELSEN, D.R. (Eds.). Soybeans: improvement, production, and uses, 3rd edn. Agronomy Monograph 16. ASSA, CSSA, and SSSA, Madison. 621-677 pp.
ZAVALA, J.A.; MAZZA, C.A.; DILLON, F.M.; CHLUDIL, H.D.; BALLARE, C.L. 2015. Soybean resistance to stink bugs (Nezara viridula and Piezodorus guildinii) increases with exposure to solar UV‐B radiation and correlates with isoflavonoid content in pods under field conditions. Plant, Cell & Environment 38, 920-928.