Artículos
Nematode populations affect grapevine seedlings: understanding the host-parasite interaction is critical for preventing negative effects
Nematode populations affect grapevine seedlings: understanding the host-parasite interaction is critical for preventing negative effects
RIA. Revista de Investigaciones Agropecuarias, vol. 50, núm. 3, pp. 140-147, 2024
Instituto Nacional de Tecnología Agropecuaria

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial 4.0 Internacional.
Abstract: The initial population density of nematodes is a primary factor that determines the damage caused to a crop. However, this factor has not been investigated adequately on grapevine. An investigation of the effects of initial population densities of Meloidogyne incognita on grapevine (cuttings) was carried out using a completely randomized design. The parameters recorded included shoot height, number of leaves, number of tendrils, and nematode population density. Low initial population densities of M. incognita had a stimulatory effect on grapevine growth. However, above 520 nematode eggs per pot, the performance of the crop (i.e. plant height, number of leaves, number of tendrils) started to decline gradually, culminating in the lowest performance at 4160 nematode eggs/pot. The mean nematode population density at the termination of the experiment revealed that the initial population density of 520 nematode eggs/pot resulted in more population build-up (4.73 and 9.55 times more nematodes than in the initial populations in Experiments 1 and 2, respectively) than that of 4160 nematode eggs/pot (with 1.46 and 1.68 times more nematodes than in the initial populations). The tolerance limit of grapevine to M. incognita (circa 520 nematode eggs) infection was determined to be the point where the magnitude of the plant character(s) started to decrease. It was concluded that this nematode is capable of severely disrupting the grapevine sector in tropical regions.
Keywords: initial population, Meloidogyne species, Vitisvinifera.
Resumen: El cultivo de la vid está empezando a ganar terreno en el trópico gracias al desarrollo de variedades locales. Sin embargo, la viticultura en esta región se enfrenta a varias enfermedades, incluidos los nematodos, que se encuentran entre los agentes patógenos duraderos y difíciles de eliminar en la viticultura. Los problemas graves de nematodos pueden comenzar en el vivero, lo que podría garantizar una infección generalizada y duradera y un alto costo de manejo. La densidad de población inicial de nematodos es un factor primario que determina el daño causado a un cultivo. Sin embargo, este factor no se ha investigado apropiadamente en la vid. No hay información adecuada sobre el efecto de las densidades de población iniciales de Meloidogyne incognita (una especie de nematodo principalmente tropical) en la vid en la vid que se cultiva en zonas tropicales. Por lo tanto, este estudio se concibió para llenar este vacío en nuestro conocimiento. Se llevó a cabo una investigación de los efectos de las densidades poblacionales iniciales de M. incognita en vid (esquejes) utilizando un diseño completamente al azar. Los parámetros de las plántulas registrados incluyeron la altura de los brotes, el número de hojas, el número de zarcillos y la densidad de población de nematodos. Las bajas densidades poblacionales iniciales de M. incognita tuvieron un efecto estimulante sobre el rendimiento de la vid. Sin embargo, por encima de 520 huevos por maceta, el rendimiento del cultivo (es decir, altura de la planta, número de hojas, número de zarcillos) comenzó a disminuir gradualmente hasta culminar en el rendimiento más bajo con 4,160 huevos/maceta. Se determinó que el límite de tolerancia de la vid a la infección por M. incognita (alrededor de 520 huevos de nematodos) era el punto en el que la magnitud de las características de la planta comenzaba a disminuir. Por tanto, este nematodo es capaz de perturbar gravemente el sector vitícola.
Palabras clave: población inicial, especies de Meloidogyne, Vitis vinifera..
INTRODUCTION
Gutierrez-Gutierrez et al. (2011) reported that grapevine (Vitis vinifera L. in the plant family Vitaceae) is cultivated for winemaking, as table grapes, and raisins. Grapevine is either processed into wine (71% of global grapevine is used for winemaking), consumed fresh (27%), or as raisins (2%). Grapevine is one of the most extensively cultivated fruit crops. Askary et al. (2018) pointed out that the leading producers of grapevine (2005-2006) included Italy (12.6%), the USA (10.5%), France (10.0%), and China (9.7%).
However, grapevine production is challenged by many pests and diseases, including plant-parasitic nematodes which cause significant yield losses (Deshar, 2019; Wallis, 2022; Iliya et al., 2023). The most serious direct damage is caused by Meloidogyne and Xiphinema species (Deshar, 2019). In northern Nigeria, many nematodes were found in grapevines, including M. incognita (Khan and Chindo, 1988; Chindo, 1991). Iliya et al. (2023) reported a total of ten plant-parasitic nematode species (Meloidogyne, Paratylenchus, Xiphinema, Scutellenema, Longidorus, Heterodera, Aphelenchoides, Trichodorus, Hoplolaimus, and Rotylenchus spp.) in northern Nigeria. M. incognita had the highest population density, the highest frequency of occurrence (100%), compared to the other varieties studied.
These nematodes can have a negative impact on the planting of new vineyards and reduce the vigour, yield, and productive lifespan of established vineyards. Moreover, nematodes may predispose grapevines to infection and damage by other pathogens (Hussey and Janssen, 2002; Ndifon, 2023). Askary et al. (2018) and Nadeem et al. (2022) asserted that plant parasitic nematodes (which can cause up to 12.5% yield loss) are amongst the major biotic constraints to grapevine production. Root-knot nematodes are one of the key parasites of grapevines in the tropics, including West Africa (Nigeria per se).
Hussey and Janssen (2002) emphasized that there are intraspecific variations in the virulence of Meloidogyne species which, when coupled with variations in the resistance of crop cultivars to nematodes, have a significant effect on the extent of loss that a nematode may induce. Moosavi (2014) stated that the yield of crops can be reduced due to high initial nematode population density at planting time, susceptibility or tolerance of the host plant, and environmental factors. Root-knot nematodes (Meloidogyne spp.) are the most destructive nematodes and account for about 5% of the total global plant yield loss annually.
Rahman et al. (2012) revealed that grapevine yields reduced significantly due to an increase in the initial nematode population densities per kg of soil. These researchers lamented that all the initial population densities assessed significantly reduced the number of bunches and yield of grapevine variety Semillon, but only the highest density (800 second stage juveniles (J2) kg-1 of dry soil) affected the bunches and yield of grapevine variety Chardonnay. They agreed that the initial population density of Meloidogyne species has a significant influence on grapevine yield loss.
Likewise, Jamali et al. (2012) confirmed that rice yield loss is influenced by the pathogenicity, initial population density, host sensitivity, host tolerance to rice white-tip nematode (Aphelenchoides besseyi), and environmental factors. Nadeem et al. (2022) propounded that M. javanica is particularly important since it spreads rapidly and has a wide host range. They revealed that a positive correlation was observed between J2 juveniles and the percentage reduction in the growth of grapevines.
Gutierrez-Gutierrez et al. (2011) in Spain reported that root-knot nematodes (especially Meloidogyne arenaria, M. incognita, and M. javanica) and Xiphinema index had a higher reproductive factor in all the varieties assessed, indicating that grapevines are suitable hosts for these nematodes.
Gharabadiyan et al. (2013) pointed out that root-knot nematode (M. javanica) management should be partly based on the knowledge of the economic threshold level. They indicated that the correlation coefficients among galling index, egg mass, and reproductive factors were positive. Thus, nematode population densities are mainly influenced by host growth, the reproductive potential of the species, and various environmental factors.
Furthermore, Claerbout et al. (2019) expounded that integrated management strategies require more knowledge about population densities and the damage threshold of pathogens and pests. Plant infection by Meloidogyne species is favored by the preservation of more inoculum in anti-dehydration egg masses (leading to a higher initial inoculum potential at the inception of infection), the polyphagous nature of the nematode (i.e. more alternative hosts all year round), the short life cycle of the nematode, and the polycyclic nature of the infection (thus enabling fast inoculum buildup and maintaining of the population), and the cosmopolitan nature of these plant parasitic nematodes. In vineyards where the plants are perennials, infections could result in higher population buildup every year which will never allow the plants to recover. In the tropics, plants tend to grow year-round so long as water is available. Yet there are very few reports on the effect of initial population densities of M. incognita on grapevine in most of the tropics. Consequently, this study was conceived to fill this gap in our knowledge.
MATERIALS AND METHODS
Preparation of potting soil
Two experiments were carried out at the Nematology Laboratory in the Department of Crop Protection, Ahmadu Bello University, Zaria (11.11 N by 7.38 E) in Nigeria. The soil utilized was dug from the campus, and its particles were pulverized by breaking the crumbs into smaller particles and sieving the soil using a metal sieve (1 cm mesh). The sieved soil (3:1 sieved soil to sand) was heat-sterilized using a cut metal drum and firewood. The soil was then allowed to cool down and any generated toxic gases were permitted to escape for four weeks.
Preparation of the grapevine cuttings
The grapevine canes were obtained from the Anab-i-sahib grapevine variety at the time of pruning. The grapevine cuttings were raised from hardwood canes in polythene planting bags for three weeks after sprouting before being used to set up the experiment. The polythene bags contained the sterilized soil prepared above.
Identification of the nematode species
The inoculum of M. incognita was obtained initially from tomato roots in the environs of the university. At the nematology laboratory, each egg mass was teased off using an inoculating needle (this egg mass was kept separate when the identification was ongoing). After the female was removed from the root, her vulvar cone was cut and she was identified by her perennial pattern using the microscope (Ndifon, 2023).
Thereafter, the egg masses from the identified nematodes were used to inoculate the sterilized soil on which three-week-old potted Roma VF tomato seedlings were transplanted (Sasanelli et al., 2006). The nematode culture was maintained by transplanting new tomato seedlings into the pots containing the nematodes. The nematodes were identified using manuals and microscopy (Mai and Lyon, 1975; Mekete et al., 2012; Ndifon, 2023).
Preparation of nematode inoculants
The nematodes were re-identified after raising the inoculum in the Nematology Laboratory by cutting the perennial patterns of the females. The identification of these nematodes was based on the pattern naturally found on female root-knot nematodes (Mai and Lyon, 1975; Mekete et al., 2012).
The egg masses were counted as they were being picked. The number of eggs per egg mass was determined for five egg masses that were selected at random and the average number of nematode eggs was obtained. The egg masses were left to hatch in counting dishes and the average hatching percentage was estimated (Deshar, 2019, Gómez-González et al., 2021, Iliya et al., 2023, Ndifon, 2023). No stimulant was needed to induce hatching during the six days.
Extracting nematode eggs from egg masses using sodium hypochlorite is considered by some as standard but the health of the nematode eggs is affected by the bleach. This may reduce the hatching of the nematode eggs and the survival of the larvae. Gómez-González et al. (2021) emphasized that the effect of the sodium hypochlorite on the viability of Meloidogyne enterolobii eggs is detrimental since it resulted in non-infective larvae, delay of hatching, and lower infectivity despite the improved hatching percent of the eggs. The hatched larvae were collected daily and counted using the counting dish and a microscope.
The middle way was taken by using egg masses to inoculate the plants. This inoculum was considered to be close to natural inoculum. The average count of nematode eggs was 130 nematode eggs per egg mass. The average hatchability of the nematode eggs was estimated to range from 92-96% (of course in a controlled environment).
Four egg masses were estimated to contain 520 nematode eggs. The number of nematode eggs was used to determine how many nematodes could be obtained bearing the hatching percent in mind and the number of nematode eggs per egg mass. The egg masses in water vials were poured into shallow holes at the bases of the plants (Di Vito et al., 2004). The holes were then filled with the soil.
Concerning the levels of nematodes utilized, Moosavi (2014) inoculated 3 kg of soil in pots with different densities of nematode eggs and juveniles (i.e. 0-512 eggs and juveniles g-1 of soil). Thus, this could have amounted to 512000 eggs and juveniles per 1000 g of soil, which is very high for a tomato plant. Carla et al. (2012) cultivated tomatoes for sixty days after they were inoculated with 2000, 4000, and 6000 M. incognita and observed decreased plant growth (shoot and root lengths, and fresh and dry root and shoot weights) with increasing nematode inoculum level.
In a different study, El-Sherif et al. (2007) applied levels of M. incognita eggs (0-1000 eggs) to each pot with tomato seedlings. This shows a wide variation in the use of inoculation levels. The M. incognita eggs El-Sherif et al. (2007) used as nematode inoculum were prepared using macerated Coleus plants with many egg masses that were not isolated.
The application of several rates or levels of nematodes per pot often seems to be exaggerated and one wonders why we do not get equivalent root galls on the root system. The nematodes compete with other microorganisms and other nematodes that feed on plants. Therefore, we used a moderate set of treatments that ranged from 0 to 4160 eggs per pot. The argument about the exact number of nematodes that hatched per treatment is valid. Nonetheless, it should be kept in mind that all the treatments received the same handling to provide equal conditions; thus, the number of nematode eggs as our reference point was used throughout the study.
Determination of the final population density of the nematodes
When extracting nematodes from the soil at the termination of the experiment, Cobb’s Sieving and decanting method was used (Van Bezooijen, 2006). The extracted nematodes were collected in measuring cylinders and used to determine how many nematodes were in the soil per replication. Counting the nematodes was done by drawing 2 ml of nematode suspension with a pipette and placing it into a counting dish.
The counting was done under a binocular compound microscope at 40x magnification (Deshar, 2019; Iliya et al., 2023; Ndifon, 2023). Three counts were taken and averaged per replication. The final number of nematodes per treatment was the average of these counts from the replications.
The experimental layout and design
The two experiments were performed using a completely randomized design (each trial had six treatments with four repetitions). The treatments included a control, 520, 1040, 2180, 3120, and 4160 nematode eggs per pot. The transplanting of the grapevine had to be done without disturbing the soil in the polythene bag. The bag was cut open with a razor and a grapevine cutting was placed inside a plastic pot (6 kg soil) and sterilized soil was then poured around the roots.
The egg masses were collected in vials containing 80 ml of distilled water to avoid dehydration while working in the screen-house. The control received placebo irrigation with 80 ml of distilled water. The inoculum was poured into four holes (each 5 cm deep) around the base of each plant. Immediately after applying the nematode egg mass suspension to the soil, the holes were carefully closed and the plants were irrigated with clean tap water (Claerbout et al., 2019).
The experiment was terminated 14 weeks later. The second experiment was set up using the same materials and methods as those used in this first experiment.
Data collection and analysis
The shoot height was measured with a tape while the number of leaves and tendrils were being counted. Data on the fresh weight of the severed shoots, fresh weight of the roots, weight of the fresh leaves, nematodes in g of soil, and nematodes (in g of roots) were collected for the thesis from which this article is drawn. The final nematode population density was obtained by using the Cobb Sieving and Decanting Method and a dissecting microscope.
The final population densities and reproduction factors were key to the study as shown herein. Al-Hazmi et al. (2017) utilized three important criteria (galling index, egg mass index, and nematode reproduction factor (Rf)) to group their assayed plant materials. The most common and accepted criterion is the reproduction factor which we adopted. We also utilized the galling index herein. López-Gómez and Verdejo-Lucas (2017) and Ndifon (2023) reported that the reproduction factor (Rf) is used as an indicator of the suitability of a host plant for a nematode.
The Rf expression (1) used is as follows:
Rf = Pf/Pi (1)
Where Pf = final population and Pi = initial population.
Thus, susceptible plants show an Rf >1, whereas resistant or non-host plants have an Rf<1.
The data were subjected to an analysis of variance procedure and the means were separated using Student Newmann-Keul’s test (p≤0.05), which was carried out using the 2004 version of System Analytical Software (SAS).
RESULTS
To determine the tolerance limit of grapevine to M. incognita, the number of leaves was plotted on a line graph (figure 1). It was observed that the number of leaves produced increased at low population density (until 520 nematode eggs per pot), but thereafter the number of leaves declined gradually. This point (520 nematode eggs per pot), at which the number of leaves began to decline, was termed the tolerance limit.
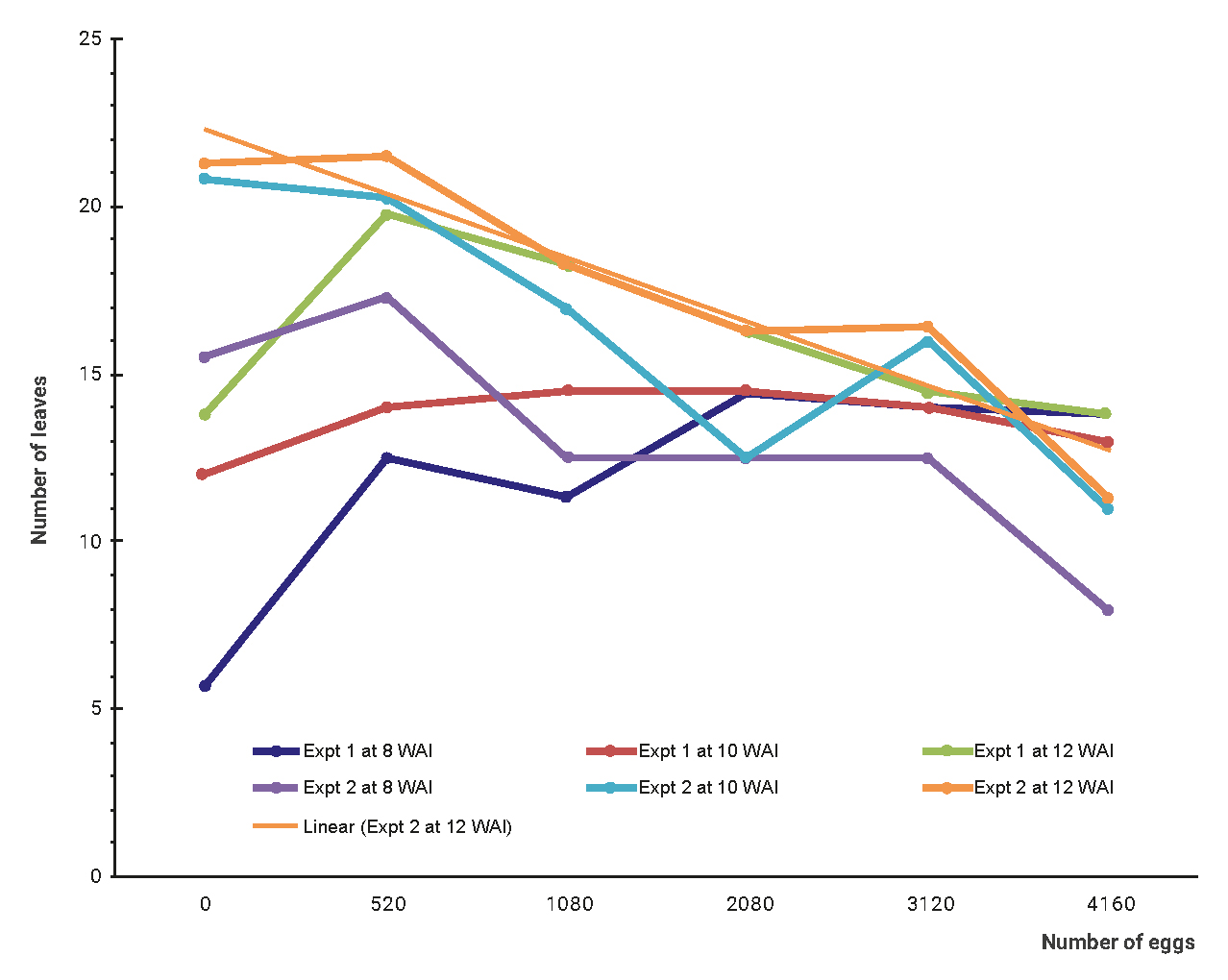
Figure 1
The effect of initial population of M. incognita eggs on the number of leaves of grapevine for Experiments 1 and 2 at 8, 10 and 12 weeks after inoculation.
The mean population density at the termination of the experiment indicated that the low initial population (520 nematode eggs per pot) resulted in more population build-up (4.73 times larger than the initial population in Experiment 1 and 9.55 times more than the initial population density in experiment 2) compared to the highest initial population of 4160 nematode eggs per pot in both experiments (table 1).
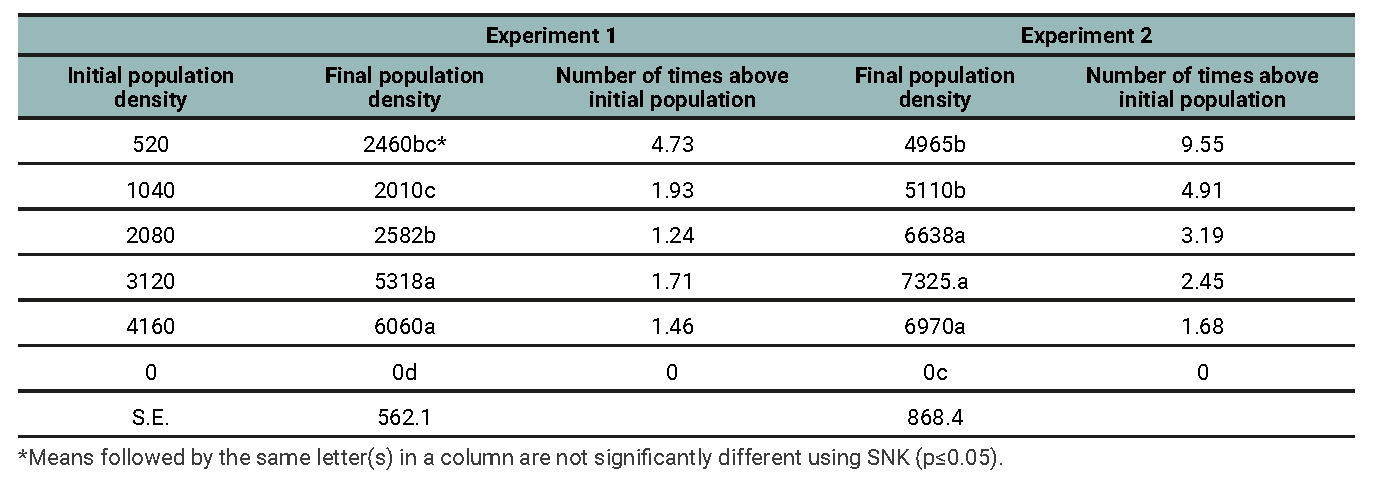
Table 1
Population densities of M. incognita on grapevine 12 weeks after inoculation for Experiments 1 and 2.
The percentage increase at 4160 nematode eggs per pot was only 146% and 168% (higher/larger increase in the population density) over the initial population in experiments 1 and 2, respectively. The treatments in between these two treatments produced population increases that were inversely proportional to their initial population densities.
It was observed that, before reaching 520 nematode eggs per pot, an increase in the number of tendrils occurred (table 2). The lowest number of tendrils was obtained at 4160 nematode eggs per pot, while the highest number of tendrils was obtained from the control plants. It was observed that, at the beginning of experiment 2 (up to 4 weeks after inoculation (WAI)), there were no tendrils available. The reason for this observation was not known. The tallest plants were obtained from the treatment that received 520 nematode eggs per pot, while the shortest plants were obtained from the treatments that received 4160 nematode eggs per pot. As the population densities of the nematodes were increased, so did the damage (table 3).2
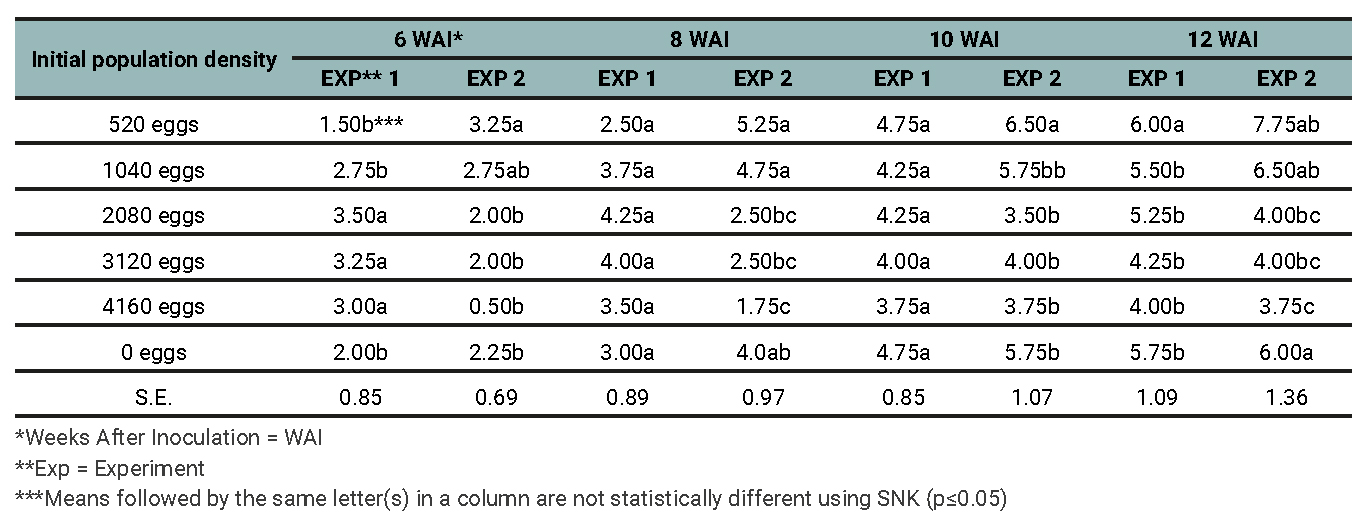
Table 2
Effects of initial population densities of M. incognita on the number of tendrils of grapevine in screen-house 6, 8, 10, and 12 weeks after inoculation for Experiments 1 and 2.
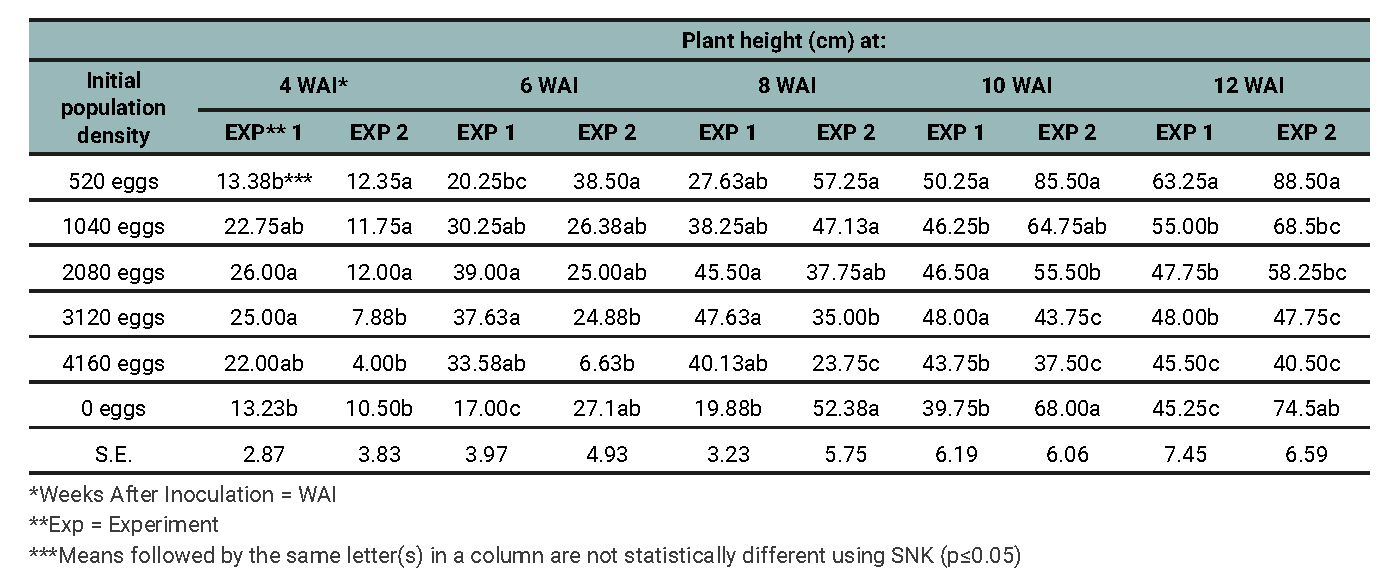
Table 3
Effects of initial population densities of M. incognita on plant height of grapevine 6, 8, 10, and 12 weeks after inoculation for Experiments 1 and 2.
DISCUSSION
The effect of population densities of nematodes on the growth and yield parameters of grapevine has remained blurred; consequently, farmers may not take action against nematode infections, which could be disastrous. As observed above, M. incognita sig nificantly reduced the growth of grapevine (that is plant height, number of leaves, and number of tendrils) as the densities of nematodes increased and as time passed. The reduction of plant height was more evident 10 weeks after inoculation in both experiments. These findings on plant height partially concurred with those of Carla et al. (2012), who reported that, after having cultivated the tomato sixty days after inoculation, the oldest tomato seedlings (5 weeks old) showed the least effects of root-knot nematode infection compared to younger seedlings (3 and 4 weeks old).
It is probable that, at low population density, the nematodes had enough feeding sites, so the damage was reduced or the plant may have compensated for the damage at low nematode populations. It was observed that, at 520 nematode eggs per pot, the number of leaves began to decline. This point was termed the tolerance limit. Sasanelli et al. (2006) reported that tolerance limits of 1280 nematode eggs and 780 juveniles per 1000 cm3 of soil were projected respectively for the grapevine cultivars 1103 Paulsen and Italia, which corroborated the findings of this experiment. But it seems that the variety Anab-i-sahib was more sensitive to nematode infection than the 1103 Paulsen and Italia varieties under equal conditions. This variety was introduced into West Africa from the Middle East, so it may be more sensitive in this region since it has to adapt afresh.
Sumita (2014) reported a decrease in growth parameters due to an increase in the nematode population density, which corroborated the findings of this present study. Root galling was at its lowest at low initial population densities and its greatest at 16000 nematode eggs and second-stage juveniles per 1000 cm3 of soil (Di Vito et al., 2004). As initial inoculum densities increased, the number of galls and egg masses increased as well on tomato (Khan, 2000). These research outcomes fully confirmed the current findings.
Another research corroborated the current findings. Carla et al. (2012) cultivated tomatoes sixty days after inoculation and observed decreased plant growth (shoot and root lengths, and fresh and dry root and shoot weights) with increasing Meloidogyne inoculum level on all four tomato genotypes. A study using three initial inoculum densities (2000, 4000, and 6000 M. incognita) while considering the influence of plant age on tomato in a greenhouse revealed a significant reduction in plant height, number of leaves, fresh and dry weight of shoot and root as the initial inoculum level was increased.
Under greenhouse conditions, by inoculating soil with different inoculum levels of M. incognita (200, 400, 800, and 1600 J2 juveniles per 1.5 kg of soil per pot), there was a significant (p≤0.05) progressive reduction in the growth and biochemical parameters of Vigna radiata as from 400 to 1600 J2 juveniles (Abbasi and Hisamuddin, 2014).
El-Sherif et al. (2007) reported a reduction in the total fresh weight of the plants (18.6-43.9%) as the inoculum level increased from 250 to 1000 nematode eggs per tomato plant, respectively. When the initial inoculum (Pi) level was increased up to 2000 nematode eggs per pepper plant, the percentage reduction of whole-plant fresh weight (73.2%) and shoot dry weight (55.3%) as well as the rate of nematode build-up (1.49) also increased.
The galls and egg masses per root system increased as Pi was increased on host plants. Root galling reached its lowest level at low initial population densities and highest at 16000 nematode eggs and second-stage juveniles per 1000 cm3 soil (Di Vito et al., 2004). These studies strongly confirmed the results of this study. In the present investigation, this reproduction factor revealed that grapevine is an excellent host to root-knot nematodes.
López-Gómez and Verdejo-Lucas (2017) revealed that, when plants were inoculated with 0, 25, 50, 100, 200, and 300 second-stage juveniles per plant, the increase in Pi was correlated with the penetration rates (R2 = 0.603, p<0.001) and total numbers of nematodes in the root (R2 = 0.963, p<0.001), but there was no correlation between Pi and the reproduction factor (nematode eggs/plant/Pi). This result seems to contradict those presented so far.
However, Sumita (2014) explicated that the growth parameters of green gram, except fresh and dry weight of roots, were inversely proportional to the inoculum level of M. incognita. At the initial inoculum level of 100 nematodes per kg of soil, significant reductions in plant growth parameters were recorded. The maximum numbers of galls and egg masses per root system were recorded at 100 and 1000 and the lowest at 10000 J2 per kg of soil. The nema tode population in soil increased together with the inoculum level. Meanwhile, the nematode reproduction rate was inversely proportional to the nematode inoculum level. This corroborates our findings, especially those on reproduction factor (Rf).
Gutierrez-Gutierrez et al. (2011) stated that root infection resulted in moderate to severe root galling in all grapevine rootstocks. The diameter of the shoot and main stem appeared to be the most sensitive variables, with reduction rates of 36-67% damage by Meloidogyne spp. The shoot height was not significantly affected by the root-knot nematodes, but the root fresh weight increased. This result is in agreement with the present findings.
Vega-Callo et al. (2021) evaluated six rootstocks and reported that the rootstocks were resistant to M. incognita, M. arenaria, andM. hapla (5000 nematode eggs + juveniles (J2)). Moosavi (2014) and Ndifon (2023) revealed that plant growth parameters and yield decreased due to an increase in the population densities of Meloidogyne species on eggplant. The highest nematode reproduction rate was seen at the lowest population density and was equal to 4379 nematodes. This result completely concurred with our findings in this present study.
CONCLUSION
A screen-house research was carried out to determine the effect of population densities of root-knot nematodes on the growth parameters of the grapevine variety Anab-i-sahib. It was observed that the performance of the grapevine was dependent on the density of nematodes used to inoculate the soil. It was concluded that control of root-knot nematodes should commence before the population of the pest in vineyards reaches 520 nematodes per liter of garden soil. It was thus concluded that the effects of higher initial populations of M. incognita on grapevine growth parameters were highly detrimental to the crop. Since the tolerance limit of grapevine to nematodes is low, it was concluded that urgent steps should be taken to inform farmers of the need to avoid and/or control this nematode in their nurseries and vineyards. Research on how to best control nematodes in vineyards should continue. The effect of higher nematode populations should also be further investigated.
REFERENCES
ABBASI, H.; HISAMUDDIN, N. 2014. Effect of different inoculum levels of Meloidogyne incognita on growth and biochemical parameters of Vigna radiata. Asian Journal of Nematology, 3, 15-20. DOI: 10.3923/ajn.2014.15.20
AL-HAZMI, A.S.; DAWABAH, A.A.M.; AL-NADHARI, S.N.; AL-YAHYA, F.A. 2017. Comparative efficacy of different approaches to managing Meloidogyne incognita on green bean. Saudi Journal of Biological Sciences, 24(1), 149-154. https://doi.org/10.1016/j.sjbs.2016.05.013
ASKARYS T.H.; KHALIL, A.; KHAN, A.A.; NAZIR, N. 2018. Population fluctuation of plant parasitic nematodes associated with grapevine under hi-tech and low-tech poly-house conditions. International Journal of Current Microbiology and Applied Science, 7(5), 2133-2140. https://doi.org/10.20546/ijcmas.2018.705.248
CHINDO, P.S. 1991. Management of pests and diseases of grapes. In: Grapevine production and control of pest and diseases of orchards. Proceeding Training Workshop Institute for Agricultural Research, Samaru. Pages 1-67.
CLAERBOUT, J.; NEUKERMANS, J.; VANDEVELDE, I.; DECOMBEL, A.; DE SUTTER, N.; DEEREN, A.; VENNEMAN, S.; BLEYAERT, P.; HÖFTE, M.; VIAENE, N. 2019. Pratylenchus penetrans, a potential risk in glasshouse-grown lettuce: population densities and damage threshold. Nematology, 22, 577-590.
DESHAR, A. 2019. Distribution of soil nematodes associated with grapevine plant in Central Horticultural Centre (CHC), Kirtipur, Kathmandu. Master of Science in Zoology, Tribhuvan University, Kathmandu, Nepal. 38 p.
DI VITO, M.; PARISI, B.; CATALANOL, F. 2004. Effect of population densities of Meloidogve incognita on common bean. Nematology Mediterranean, 32, 81-85.
EL-SHERIF, A.G.; REFAEI, A.R.; EL-NAGAR, M.E.; HAGAR, S.M.M. 2007. The role of eggs inoculum level of Meloidogyne incognita on their reproduction and host reaction. African Journal of Agricultural Research, 2(4), 159-163. (Available at: http://www.academicjournals.org/AJAR verified on November 21, 2023).
GHARABADIYAN, F.; JAMALI, S.; KOMEILI, H.R. 2013. Determining of root-knot nematode (Meloidogyne javanica) damage function for tomato cultivars. Journal of Agricultural Sciences, 58(2), 147-157. DOI: 10.2298/JAS1302147G UDC: 635.64-265.132
GÓMEZ-GONZÁLEZ, G.; CRUZ-LACHICA, I.; MÁRQUEZ-ZEQUERA, I.; VALDEZ-TORRES, J.B.; TOVAR-PEDRAZA, J.M.; OSUNA-GARCÍA, L.A.; GARCÍA-ESTRADA, R.S. 2021. Meloidogyne enterolobii egg extraction in NaOCl versus infectivity of inoculum on cucumber. Journal of Nematology, 53, e2021-57. DOI:10.21307/jofnem-2021-057
GUTIERREZ-GUTIERREZ, C.; PALOMARES-RIUS, J.E.; JIME´NEZ-DI´AZ, R.M.; CASTILLO, P. 2011. Host suitability of Vitis rootstocks to root-knot nematodes (Meloidogyne spp.) and the dagger nematode Xiphinema index, and plant damage caused by infections. Plant Pathology.Doi:10.1111/j.1365-3059.2010.02404.x
HUSSEY, R.S.; JANSSEN, G.J. W. 2002. Root-knot Nematodes: Meloidogyne Species. In: STARR, J.L.; COOK, R.; BRIDGE, J. (ed.). Plant Resistance to Parasitic Nematodes. CABI Publishing. New York. USA. 43-70 pp.
ILIYA, C.J.; LILIAN, D.S.; IBRAHIM, U.; ABRAHAM, P.; IBRAHIM, S. 2023. Survey on plant parasitic nematode associated with grapevine (Vitis vinifera L.) at Federal College of Horticulture Dadin-Kowa, Gombe, Nigeria. EQA - International Journal of Environmental Quality, 55, 33-41. https://eqa.unibo.it. DOI: 10.6092/issn.2281-4485/16767
JAMALI, S.; POURJAM, E.; SAFAI, N. 2012. Determining the relationship between population density of white tip nematode and rice yield. Journal of Agricultural Science and Technology, 14, 195-203.
KHAN, H.; RIAZ, A.; AKHTAR, A. S.; ARSHAD, M.; TAHIR, B.; NIAZ, T. 2000. Effect of inoculum density of Meloidogyne incognita and plant age on the severity of root-knot disease in tomato. International Journal of Agriculture and Biology, 2(4), 360-363.
KHAN, F. A.; CHINDO, P.S. 1988. Nematode pests of grapevine in northern Nigeria. Paper presented at the 25th Annual Conference of Agricultural Society of Nigeria, held at Federal University of Technology, Owerri, Nigeria.
LÓPEZ-GÓMEZ, M.; SOLEDAD, V. 2017. Penetration and post-infection development of root-knot nematodes in watermelon. Spanish Journal of Agricultural Research, 15(4), Article e1010, 8 p. https://doi.org/10.5424/sjar/2017154-11189
MAEITA, C.M.M.; CURTIS, R.H.C.; POWERS, R.HC.; ABRANTES, I.M.DE O. 2012. Inoculum levels of Meloidogyne hispanica and M. javanica affect nematode reproduction, and growth of tomato genotypes. Phytopathologia Mediterranea, 51(3), 566-576.
MAI, W.F.; LYON, H.H. 1975. Pictorial key to genera of plant parasitic nematodes, 4th Edition. Cornell Univ. Press, Ithaca. 219 p.
MEKETE, T.; DABABAT, A.; SEKORA, N.; AKYAZI, F.; ABEBE, E. 2012. Identification key for agricultural important plant-parasitic nematodes. Prepared for the international Nematode diagnosis and identification course 2012. A manual for nematology. CIMMYT. Mexico, D.F. 23 p.
MOOSAVI, M.R. 2014. Densities of damage to eggplant by Meloidogyne javanica. Cibtech Journal of Zoology, 3(3), 43-49.(Available at:http://www.cibtech.org/cjz.htm verified on November 21, 2023).
NADEEM, H.; KHAN, A.; GUPTA, R.; ANEES, A.; AHMAD, F. 2022. A Seinhorst model determined the host-parasite relationships of Meloidogyne javanica infecting Fenugreek cv. UM202. Journal of Nematology, 54. DOI:10.2478/jofnem-2023-0005
NDIFON, E.M. 2023. Host-parasite interactions between Solanum aethiopicum, Meloidogyne incognita, and Fusarium oxysporum f.sp. melongenae as portrayed by disease traits and crop yield. Yuzuncu Yil University Journal of Agricultural Sciences, 33(3), 461-477. https://doi.org/10.29133/yyutbd.1256166
RAHMAN, L.; ORCHARD, B.; WHITELAW-WECKERT, M.; HUTTON, R.J. 2012. Susceptibility of Vitis vinifera cv ‘Semillon’ and ‘Chardonnay’ to the root knot nematode M. javanica. Vitis, 51(1), 19-26.
SASANELLI. N.; D’ADDABBO, T.; LIŠKOVÁ, M. 2006. Influence of the root-knot nematode Meloidogyne incognita race 1 on growth of grapevine. Helminthologia, 43(3), 168-170.
SUMITA, K. 2014. Pathogenicity of root-knot nematode, Meloidogyne incognita in green gram. International Journal of Purend Applied Bioscience, 2(6), 182-184. (Available at: www.ijpab.com verified on November 21, 2023).
VAN BEZOOIJEN, J. 2006. Methods and techniques for nematology. Wageningen. 112 p.
VEGA-CALLO, R.A.; TAMO-ZEGARRA, J.J.; BELLÉ, C. 2021. Reaction of grapevine rootstocks and cultivars to Meloidogyne incognita, M. arenaria, and M. hapla.Agriscientia, 38:93-98. DOI: 10.31047/1668.298x.v38.n1.29368
WALLIS, C.M. 2022. Microplate bioassay to examine the effects of grapevine-isolated stilbenoids on survival of root knot nematodes. BMC Research Notes, 15, Article 220.
Información adicional
CONFLICT OF INTEREST: The author declares that there is no conflict of interest (financial, social or corporate) that can hinder the publication of this paper.