Insecticidal and fungicidal activity of eucalyptol against pest and fungal diseases of soybean
Insecticidal and fungicidal activity of eucalyptol against pest and fungal diseases of soybean
RIA. Revista de Investigaciones Agropecuarias, vol. 51, núm. 2, pp. 88-96, 2025
Instituto Nacional de Tecnología Agropecuaria

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial 4.0 Internacional.
Abstract: Soybean (Glycine max), a globally significant crop, faces significant economic losses from pests and fungi. Farmers often resort to synthetic pesticides, posing potential risks to human and environmental health. In this context, eucalyptol (1, 8 cineole) stands out in agriculture for its toxicity to various insects and fungi, with minimal environmental and human health impacts. This study aimed to assess in the lab the insecticidal activity of eucalyptol against Spodoptera frugiperda and Epicauta atomaria. Its fungicidal activity was studied against Cercospora kikuchii, C. sojina, and Sclerotium rolfsii. Preliminary exploration of the potential phytotoxic effect on soybean plants has been initiated. Eucalyptol demonstrated significant fumigant insecticidal activity against S. frugiperda 1st instar larvae (LC50 = 9.4 µL/L air) and E. atomaria adults (LC50 = 34.6 µL/L air), along with significant fungicidal activity against C. kikuchii and C. sojina (mycelial inhibition halo diameters of 6.0 and 10.0 mm, respectively, at a concentration of 5 µL/disk). Importantly, eucalyptol exhibited no phytotoxic effects on soybean. Eucalyptol potential as a biopesticide for soybean crops, providing an alternative to synthetic pesticides. Further research is needed to determine its economic viability and large-scale applicability.
Keywords: botanical insecticides, sustainable pest management, monoterpene, plant extract, phytotoxicity, fall armyworm, blister beetle, fungicide effect.
Resumen: La soja (Glycine max), un cultivo de importancia global, enfrenta pérdidas económicas significativas debido a plagas e infecciones fúngicas. Ante esto, los agricultores suelen recurrir a plaguicidas sintéticos, lo que puede generar riesgos para la salud humana y el medioambiente. En este escenario, el eucaliptol (1,8-cineol) se destaca en la agricultura por su toxicidad contra diversos insectos y hongos, con un impacto mínimo en el medioambiente y la salud humana. El objetivo de este estudio fue evaluar en laboratorio la actividad insecticida del eucaliptol contra Spodoptera frugiperda y Epicauta atomaria. Asimismo, se investigó su actividad fungicida contra Cercospora kikuchii, C. sojina y Sclerotium rolfsii. Adicionalmente, se comenzó a explorar su potencial efecto fitotóxico en plantas de soja. El eucaliptol demostró una significativa actividad insecticida fumigante contra larvas de primer instar de S. frugiperda (LC50 = 9.4 µL/L de aire) y adultos de E. atomaria (LC50 = 34.6 µL/L de aire). También se observó una notable actividad fungicida contra C. kikuchii y C. sojina, con diámetros de halo de inhibición micelial de 6.0 y 10.0 mm, respectivamente, a una concentración de 5 µL/disco. Es importante resaltar que el eucaliptol no mostró efectos fitotóxicos en la soja. En conclusión, el eucaliptol presenta un gran potencial como biopesticida para los cultivos de soja, ofreciendo una alternativa a los plaguicidas sintéticos. Sin embargo, se requiere investigación adicional para determinar su viabilidad económica y su aplicabilidad a gran escala.
Palabras clave: insecticidas botánicos, manejo sostenible de plagas, monoterpeno, extracto vegetal, fitotoxicidad, gusano cogollero, escarabajo vesicante, efecto fungicida.
INTRODUCTION
Soybean, Glycine max (L.) Merr (Fabales: Fabaceae), is a globally significant crop, playing a pivotal role in oil and protein production (Graham and Vance 2003; Hamza et al., 2024). Argentina is the world’s third-largest producer of soybeans, with an annual output of 51 million tonnes, accounting for 13% of global production (USDA, 2022). Nevertheless, various pests and fungal diseases can cause considerable damage to these crops in Argentina, resulting in significant annual economic losses (Jerez et al., 2023; Murúa et al., 2018; Ploper, 2004). The fall armyworm (FAW), Spodoptera frugiperda JE Smith (Lepidoptera: Noctuidae), and the blister beetle (BB), Epicauta atomaria Germar (Coleoptera: Meloidae), are phytophagous insects that affect various agricultural crops, including soybeans (De Freitas Bueno et al., 2011; Campos-Soldini et al., 2021; Overton et al., 2021). FAW can cause significant yield losses in soybean, especially at post-bloom stages, where intervention thresholds have been reported at 25% defoliation, compared to 50% at pre-bloom (Overton et al., 2021). In contrast, no established thresholds have been reported for BB. Additionally, Cercospora kikuchii (Tak. Matsumoto and Tomoy.) MW Gardner, Cercospora sojina K Hara (Mycosphaerellales: Mycosphaerellaceae), and Sclerotium rolfsii Sacc. (Amylocorticiales: Atheliaceae) are phytopathogenic fungi responsible for various diseases in soybean (Barro et al., 2023; Billah et al., 2017; Bhamra and Borah, 2022; Hartman et al., 1999; Sautua et al., 2019). Although soybean yield losses caused by these pathogens are difficult to estimate, as they, along with other pathogens, are part of the “late-season soybean diseases” (LSD). Collectively, these diseases account for an average of 10% of annual losses, but can reach up to 30%, depending on environmental conditions (Carmona et al., 2016). In Argentina, it was estimated that damage caused by C. kikuchii reduced soybean crop yields by 11% in 2018 and 2019 (Lavilla and Ivancovich, 2021). Consequently, farmers often turn to synthetic pesticides to mitigate these issues. These synthetic pesticides can be harmful to both human and environmental health, especially when used excessively or inappropriately (Alaoui et al., 2024; Aparicio and De Gerónimo, 2024; Rani et al., 2021). Therefore, it is essential to seek and promote new tools that facilitate the more sustainable control of these pests and diseases.
Eucalyptol (1, 8-cineole) is a monoterpene oxide comprising up to 85% of the total essential oils extracted from eucalyptus species (Campos and Berteina-Raboin, 2022). It is also present in essential oils from rosemary (Salvia rosmarinus Spenn.), lavender (Lavandula sp), and laurel (Laurus nobilis L.), albeit in smaller proportions (Borges et al., 2019; Cavanagh and Wilkinson, 2002; Chahal et al., 2017). Currently, there is increasing interest in eucalyptol, not only in the pharmaceutical and cosmetic industries (Cai et al., 2021; Hoch et al., 2023) but also in agriculture. This interest in eucalyptol stems from its notable toxicity against various pest insects and phytopathogenic fungi (Jiang et al., 2020; Tahiri et al., 2022; Tripathi and Mishra, 2016), coupled with its facile biodegradability and minimal impact on the environmental and human health (Batish et al., 2008).
So far, the potential of eucalyptol as a prospective active compound in biopesticide formulations for controlling the mentioned insects and phytopathogenic fungi is still understudied. However, some studies have revealed that essential oils rich in eucalyptol exhibit pronounced toxicity against these insects and fungi (Sekhar et al., 2020; Usseglio et al., 2022; Wagner and Campos-Soldini, 2022; Wagner et al., 2021). Based on these considerations, we aim to pursue the following research objectives: i) evaluate the insecticidal and fungicidal activity of eucalyptol against the mentioned species of insect and fungi, and ii) investigate the possible phytotoxic effect of eucalyptol on soybean plants.
MATERIALS AND METHODS
Chemical compound
Eucalyptol (1, 8-cineole), characterized by its analytical-grade quality (99% purity), was obtained from Merck-Sigma-Aldrich®, Argentina, and is commercially available at http://www.sigmaaldrich.com/. Chlorpyrifos (10.5% w/v) (Huagro Hormix, Huagro SA, Argentina) and difenoconazole (25% w/v) (Janfry®, Gleba SA, Argentina) and 2, 4-D (66.9% w/v) (Enlist®,Corteva Agriscience™, Argentina).
Insects and fungi
BB adults were collected manually from their host plants (Salpichroa origanifolia (Lam.) Baill. and Amaranthus hybridus L.) located in the vicinity of soybean fields near Diamante, Argentina, during the spring period (October and November) of 2022. FAW larvae were acquired from the moth colony in the CICYTTP insectarium, Diamante, established in 2020. Both species were maintained in the laboratory under controlled conditions at 27 ± 1°C, 70 ± 5% relative humidity, and a light-dark photoperiod of 16/8 h. BB adults were feed with fresh chard leaves, while FAW larvae were nourished using an artificial diet based on chickpea flour and wheat germ (Murúa et al., 2003).
The fungi C. kikuchii (strain NRBC 6713), C. sojina (strain NRBC 6715), and S. rolfsii (strain CCC 143-2018), were sourced from microbial collections affiliated with the Facultad de Bioquímica y Ciencias Biológicas at the Universidad Nacional del Litoral, Argentina, and the Centro de Referencia de Micología (CEREMIC) at the Universidad Nacional de Rosario, Argentina.
Insecticidal activity
The fumigant insecticidal activity of eucalyptol was evaluated following the protocol outlined by Baghouz et al. (2024), with slight adaptations. Briefly, groups of five insects (BB adults or FAW 1st instar larvae) were placed in 127 mL glass vials fitted with rubber lids. A 1 cm² Whatman filter paper disk was affixed to the underside of each rubber lid. The filter paper disks were impregnated with varying aliquots of pure eucalyptol, using an automatic pipette, to achieve concentrations of 19.7, 27.6, 35.4, 47.2, and 78.7 µL/L air (for experiments with BB) and 3.1, 6.3, 9.4, 12.6, and 19.7 µL/L air (for experiments with FAW). To support the filter paper on the lid and prevent direct contact of the insects with eucalyptol, a porous fabric mesh was used between the lids and the vials. To prevent vapour escape, the vials were hermetically sealed with Parafilm. Negative controls only contained a filter paper disk free of chemical substances, while positive controls incorporated a filter paper disk with chlorpyrifos at equivalent concentrations to those used with eucalyptol in both species. Each treatment was replicated five times under controlled conditions at a temperature of 27 ± 1°C and a light-dark photoperiod of 16/8 h. Each replication was conducted on different days. Mortality was recorded after 6 h of exposure, for BB and 24 h, for FAW. Insects were deemed dead if they remained motionless in response to stimuli provided by entomological brushes and forceps.
Fungicidal activity
The fungicidal activity of eucalyptol against C. kikuchii, C. sojina, or S. rolfsii was evaluated using the disc diffusion method, as described by Sequín et al. (2023), with some adaptations. Fungal hyphal suspensions were obtained by carefully collecting mycelium from 7-day-old colonies of C. kikuchii, C. sojina, or S. rolfsii grown on PDA (Potato Dextrose Agar). The hyphal fragments in each suspension underwent microbiological surface quantification and were subsequently adjusted to a concentration ranging from 4.0 × 102 to 4.3 × 103 CFU/mL. Following this, 100 µL of each suspension was inoculated at the centre of a 9 cm diameter Petri dish containing 10 mL of YMDA (4 g/L yeast extract, 4 g/L malt extract, 10 g/L dextrose, 15 g/L agar), ensuring even distribution across the medium surface.
Sterile 5 mm diameter Whatman No 4 filter paper discs were impregnated with 5, 2.5, and 1 µL of eucalyptol per disc, plus 5 µL of dimethyl sulfoxide (DMSO). Negative controls consisted solely of discs saturated with 5 µL of DMSO per disc, while positive controls contained 5 µL of difenoconazole (concentration: 0.2 µg of a.i for µL) from a commercial source, which was used as a positive control per disc. Subsequently, the impregnated discs were placed on the surface of the PDA-inoculated Petri dishes. The Petri dishes were flipped and incubated in an oven at 25 ± 1°C, maintaining a 16/8 h light-dark cycle for a duration of 7 days. Each treatment was replicated four times on different days. Mycelial inhibition halo diameter was measured at the experiment’s conclusion using a digital caliper, values expressed in millimetres (mm).
Phytotoxic activity
The potential phytotoxic effect of eucalyptol on soybean was evaluated, following a protocol similar to that of the International Seed Testing Association (1976), but with slight modifications. Briefly, single soybean seeds were planted inside plastic boxes containing a 2 cm-thick layer of sterilized sand (weight: 30 g). Each sand layer was treated with a 3 mL solution of eucalyptol (concentration: 1,150 µg/mL) for the experimental samples, or distilled water for negative controls, or 2, 4-D (1.25 µg/mL) for positive controls. In all cases, distilled water with 1% v/v Tween 20 as diluent was used.
Each box with its soybean seed was considered one replicate, totalling 12 replicates per treatment. The boxes were hermetically sealed with a lid and placed in a germination chamber at a constant temperature of 25 ± 1°C, with a photoperiod of 16/8 h light-dark cycle for six days. At the end of this period, the hypocotyl and radicle of each seedling were measured using a digital caliper. The dry weight of each seedling was also determined after dehydrating them in an oven at 50°C for 3 days until a constant weight was achieved.
Statistical analysis
For BB and FAW experiments, lethal concentration doses producing 50% (LC50) and 90% (LC90) mortality were determined using Probit analysis (Finney, 1971) with POLO-PLUS Software (LeOra Software, 2002–2014). Significant differences between LC50 and LC90 values were considered when the 95% confidence limits did not overlap. Insecticidal, fungicidal and phytotoxic activity were assessed using the Kruskal-Wallis test, followed by a Conover test for post hoc comparisons (Conover, 1999), utilizing InfoStat version 2018 statistical software, and with α = 0.05.
RESULTS
Insecticidal activity
Figure 1 illustrates the mortality percentages caused by the fumigant action of different eucalyptol concentrations against FAW and BB. Table 1 summarises the calculated LC₅₀ and LC₉₀ values for eucalyptol and chlorpyrifos (positive control) against both species. Eucalyptol exhibited significant fumigant insecticidal activity in a concentration-dependent manner against FAW 1st instar larvae (H = 24.61; df = 5; p < 0.05) and BB adults (H = 22.33; df = 5; p < 0.05). Median mortality reached 100% at concentrations equal to or higher than 9.4 µL/L air for FAW and 47.2 µL/L air for BB, respectively. The calculated LC₅₀ values were 6.1 µL/L air for FAW and 34.6 µL/L air for BB. Chlorpyrifos (positive control) showed potent fumigant activity with LC50 values below 3.1 and 19.7 µL/L of air against FAW and BB, respectively (table 1). Negative controls showed no mortality in either species.
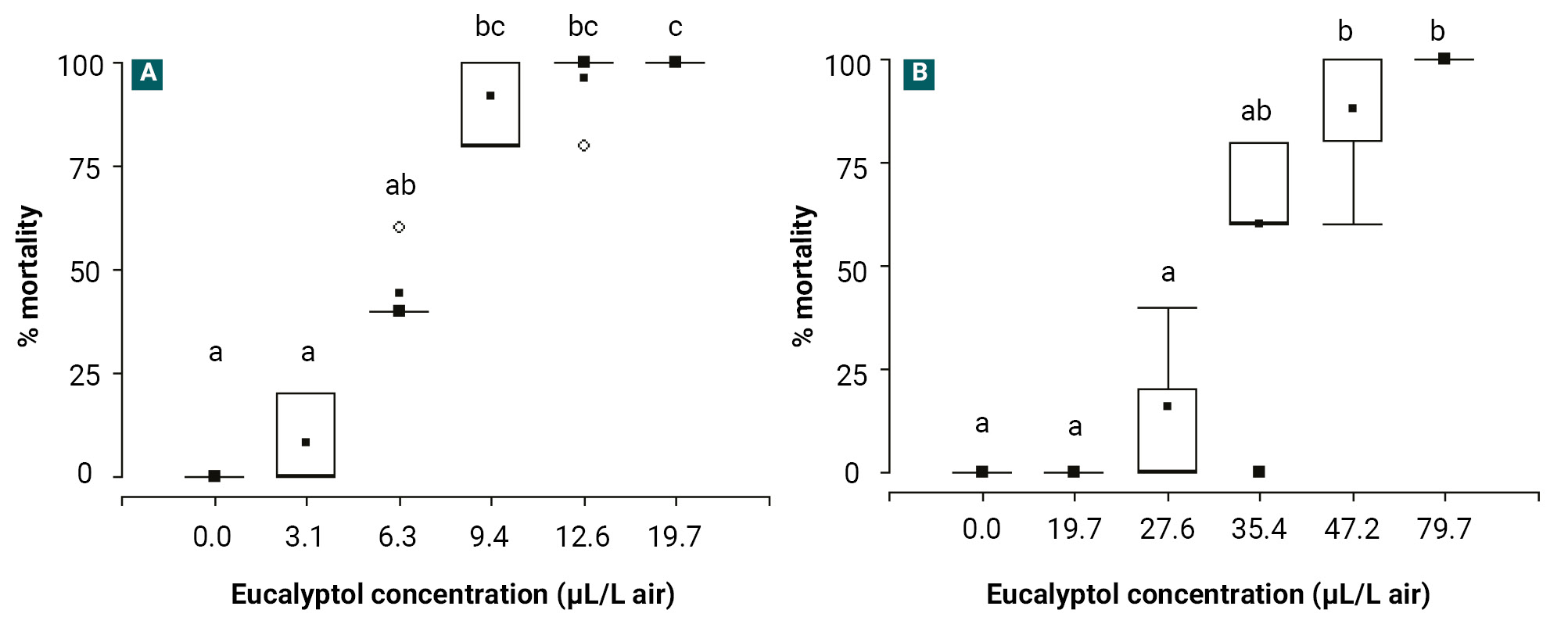
Figure 1.
Box and whisker plots showing the percentage mortality caused by the fumigant effect of different eucalyptol concentrations on (A) Spodoptera frugiperda first-instar larvae and (B) Epicauta atomaria adults. Each box represents the median (horizontal line), the mean (central point), and the interquartile range (percentiles 25 and 75). Whiskers extend to percentiles 10 and 90. Data analysis was conducted using a Kruskal-Wallis test followed by a Conover multiple comparison test (p < 0.05; n = 12). Different letters above the boxes indicate significant differences among treatments.
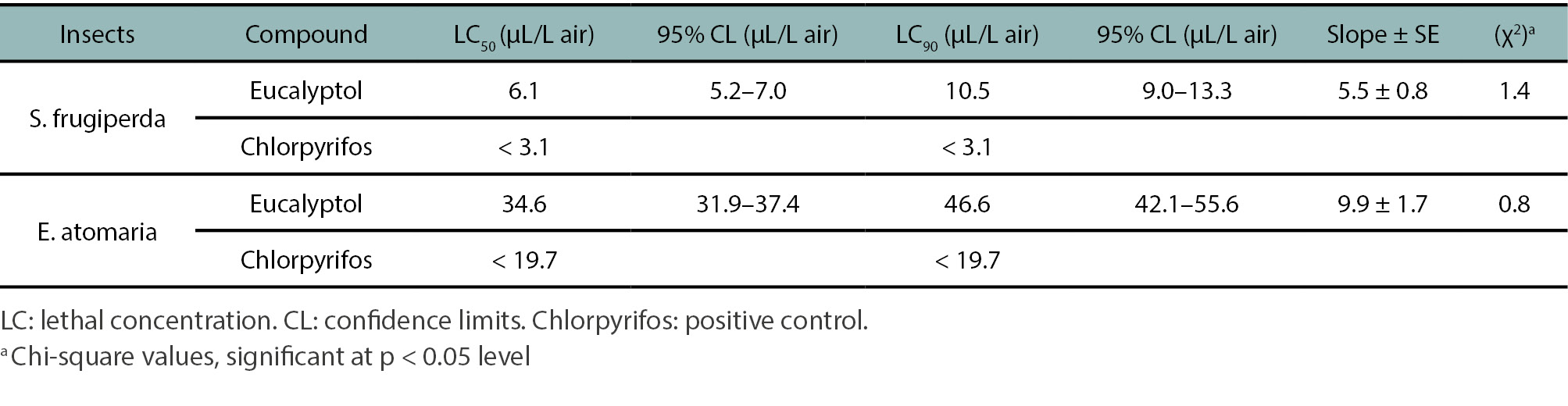
Table 1.
Fumigant toxicity of eucalyptol against Spodoptera frugiperda first-instar larvae and Epicauta atomaria adults.
Fungicidal activity
Table 2 presents the median values of the mycelial inhibition halo diameters caused by the fungicidal action of eucalyptol at different concentrations against C. kikuchii, C. sojina, and S. rolfsii. Eucalyptol demonstrated significant fungicidal activity against C. kikuchii and C. sojina. Specifically, only mycotoxicity was observed at the highest evaluated concentration (5 µL/disk). At this concentration, mycelial inhibition halo diameters of 6.0 ± 0.0 mm and 10.0 ± 2.5 mm (median ± interquartile range) were observed for C. kikuchii and C. sojina, respectively. These values differed significantly from the negative controls, where no mycelial inhibition was evident (table 2). In contrast, the positive control (difenoconazole: 5 µl/disk), applied to both C. kikuchii and C. sojina, exhibited mycelial inhibition halo diameters significantly similar to the maximum eucalyptol concentration. However, neither eucalyptol nor the negative control nor difenoconazole inhibited the growth of S. rolfsii mycelium.
Insecticidal activity
To date, the insecticidal activity of eucalyptol through fumigant action against FAW larvae and BB has not been reported. However, studies have shown that, when applied topically, eucalyptol exhibits toxicity (59.6% mortality) against FAW at a dose of ≈ 0.01223 µL/larva (Bibiano et al., 2022). In contrast, at lower doses (≈ 0.00326 µL of eucalyptol/mg of larva), its toxicity is practically negligible (Niculau et al., 2013). Within the mode of application by fumigation, previous studies reveal that essential oils from lavender and rosemary, rich in eucalyptol (34.33 and 18.72%, respectively), demonstrate outstanding activity against BB, with LC50 values of 28.9 and 23.3 µl/L air, respectively (Wagner and Campos-Soldini, 2022; Wagner et al., 2021). Although this better activity than eucalyptol could be attributed to the addition or synergy with other compounds present in the oils as it was suggested in the case of Tenebrio molitor (L) (Coleoptera: Tenebrionidae) and Spodoptera littoralis Boisd (Lepidoptera: Noctuidae) (Lima et al., 2011; Pavela, 2014).
Eucalyptol also shows fumigant toxicity in other beetle pests, such as stored grain insects Sitophilus oryzae L (Coleoptera: Curculionidae), Tribolium castaneum Herbst, Tribolium confusum Jacquelin du Val (Coleoptera: Tenebrionidae), and Rhyzopertha dominica Fabricius (Coleoptera: Bostrichidae) (Lee et al., 2004; Abdelgaleil et al., 2009; Kheloul et al., 2023).
As observed in other insect species, the toxicity induced by eucalyptol in FAW and BB could be attributed to the inhibition of acetylcholinesterase (AChE) enzyme activity, which disrupts the insect’s nervous system, leading to paralysis and eventual death (Abdelgaleil et al., 2009; Picollo et al., 2008).
Fungicidal activity
While previous studies highlight the significant potential of eucalyptol as a fungicide against various phytopathogenic fungi (Morcia et al., 2012; Shukla et al., 2012; Oxenham et al., 2005; Dammak et al., 2019; Jiang et al., 2020), this study is the first to report its fungicidal activity against C. kikuchii and C. sojina. However, our research group previously determined that, at the same concentration of 5 µl/disk, the essential oil extracted from lavender, rich in eucalyptol (34.33%), exhibits potent fungicidal activity against C. kikuchii and C. sojina, with mycelial inhibition halos of 34.0 and 29.5 mm, respectively (Wagner et al., 2021). This indicates a fungicidal activity much superior of the natural oil to that observed for pure eucalyptol. This enhanced activity is likely attributed to additive or synergistic effects with other major components present in the essential oil (Hassan et al., 2020; Yan et al., 2021).
On the contrary, in our experiments, eucalyptol does not exert a significant inhibitory effect on the mycelial growth of S. rolfsii. These results contradict findings by Kottearachchi et al. (2012), who report 100% inhibition of S. rolfsii mycelial growth when treated with high concentrations of eucalyptol (0.5 and 1.5%). However, the same authors note that at lower concentrations, the inhibition percentage decreases to less than 30%.
It is well-established that many essential oils, rich in terpenes, possess the ability to induce alterations in the fungal cell wall, plasma membrane, and mitochondria, thereby substantiating their toxicity (Kishore et al., 2007; Pawar and Thaker, 2006). Indeed, toxicity studies conducted against the phytopathogenic fungus Botrytis cinerea confirm the detrimental effects of eucalyptol on the organelles of this fungus’s cells (Yu et al., 2015). It is plausible that the observed mycotoxicity of eucalyptol against C. kikuchii and C. sojina may be attributed to some of these reasons.
Phytotoxicity
Previous studies have underscored the strong phytotoxicity of eucalyptol against various plant species as lettuce (Qiu et al., 2010), annual ryegrass, radish (Barton et al., 2014), redroot amaranth and annual bluegrass (Shao et al., 2018; Zhou et al., 2019), our preliminary findings suggest that even at high concentrations, eucalyptol is not phytotoxic to soybean seedlings. Consistent with our study on the herbicidal effects of eucalyptol, Vaughn and Spencer (1993), using a similar methodology,
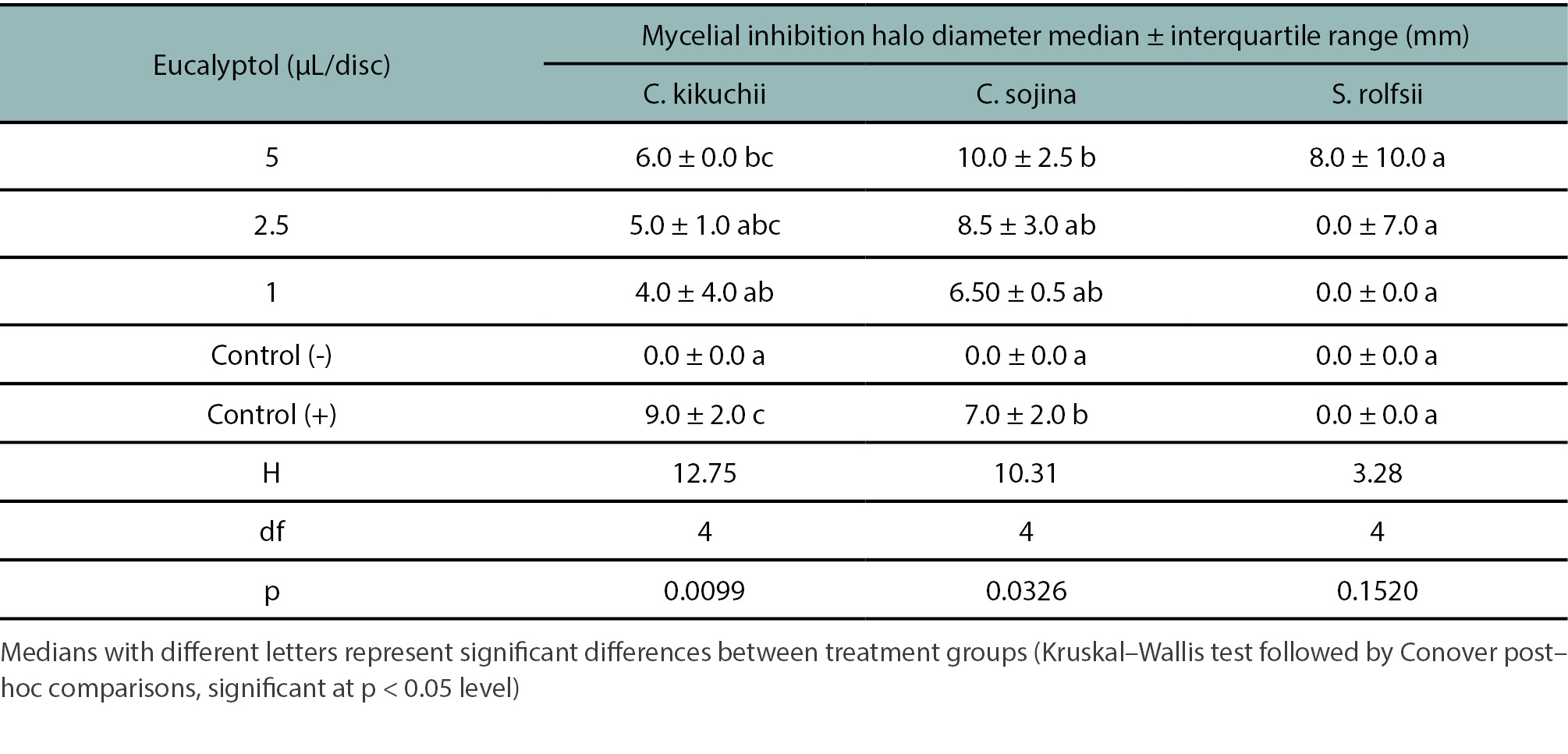
Table 2.
Mycelial inhibition halo diameters values recorded for eucalyptol against Cercospora kikuchii, Cercospora sojina, and Sclerotium rolfsii
Phytotoxic activity
Figure 2 shows the phytotoxic effect caused by eucalyptol on the radicle growth, hypocotyl growth, and dry weight of soybean seedlings. Eucalyptol at 1,150 µg/mL (≈ 1,249 µL/L) exhibited no phytotoxic activity on soybeans despite even at a concentration 1 × 103 times higher 2, 4-D (positive control: 1.25 µg/mL). In contrast, 2, 4-D demonstrated significant phytotoxicity, as evidenced by a significant reduction in both radicle length growth (H = 15.17; df = 2; p < 0.05) and seedling dry weight (H = 7.09; df = 2; p < 0.05). However, hypocotyl growth remained unaffected by both eucalyptol and 2, 4-D.
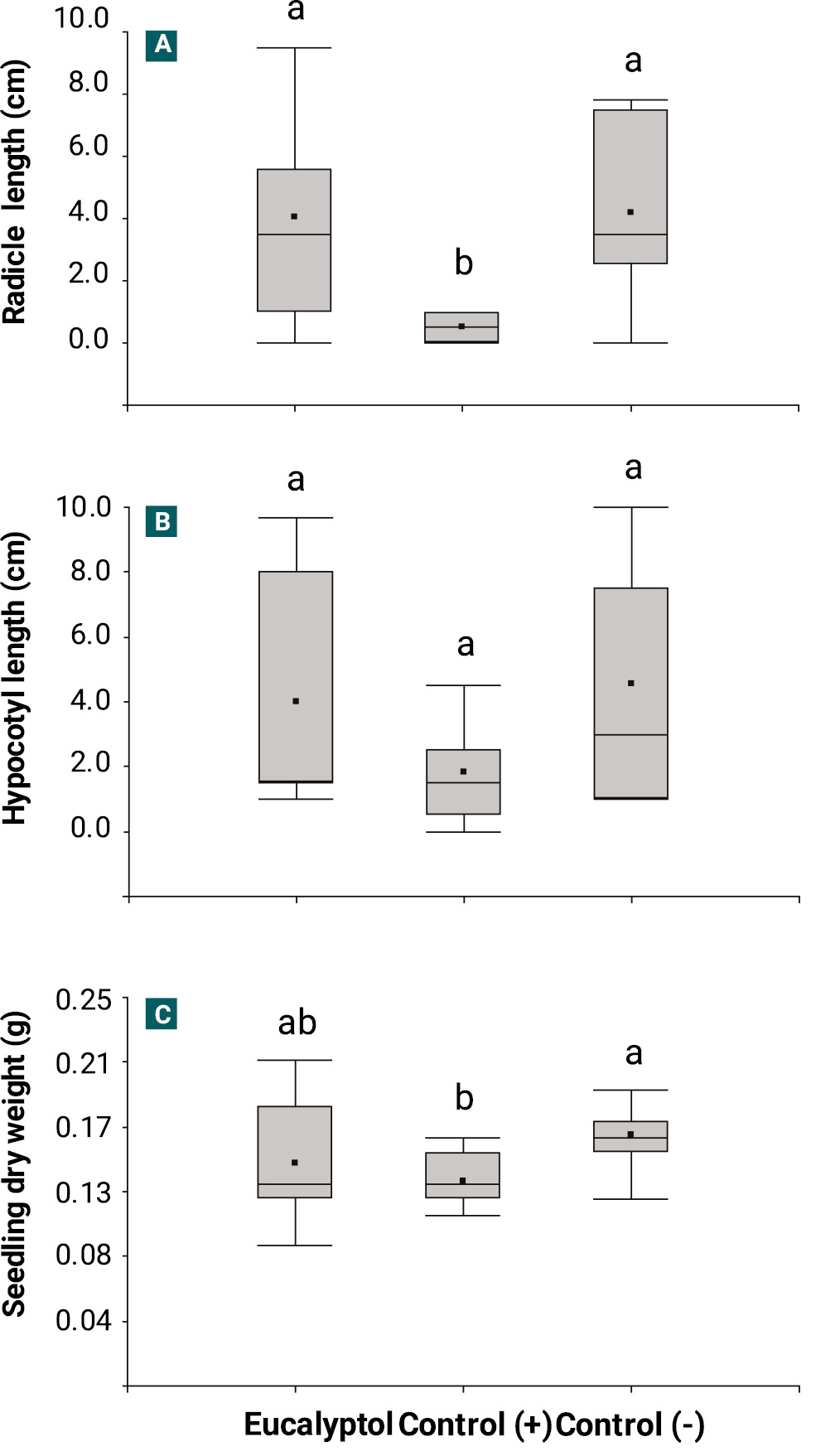
Figure 2.
Box and whisker plots represent the effect of eucalyptol at 1.15 µg/mL on A) radicle growth, B) hypocotyl growth, and C) dry weight of soybean seedlings. Positive control: 2, 4-D at 1.25 µg/mL, and negative control: distilled water with 1% v/v Tween 20. Each box represents the median (horizontal line), the mean (central point), and the interquartile range (percentiles 25 and 75). Whiskers extend to percentiles 10 and 90. Data analysis was conducted using a Kruskal-Wallis test followed by a Conover multiple comparison test (p < 0.05; n = 12). Different letters above the boxes indicate significant differences among treatments.
DISCUSSION
In general, eucalyptol exhibited fumigant insecticidal activity against FAW 1st instar larvae and BB adults, as well as fungicidal activity against C. kikuchii and C. sojina. Importantly, eucalyptol showed no phytotoxic effects on soybean.
Insecticidal activity
To date, the insecticidal activity of eucalyptol through fumigant action against FAW larvae and BB has not been reported. However, studies have shown that, when applied topically, eucalyptol exhibits toxicity (59.6% mortality) against FAW at a dose of ≈ 0.01223 µL/larva (Bibiano et al., 2022). In contrast, at lower doses (≈ 0.00326 µL of eucalyptol/mg of larva), its toxicity is practically negligible (Niculau et al., 2013). Within the mode of application by fumigation, previous studies reveal that essential oils from lavender and rosemary, rich in eucalyptol (34.33 and 18.72%, respectively), demonstrate outstanding activity against BB, with LC50 values of 28.9 and 23.3 µl/L air, respectively (Wagner and Campos-Soldini, 2022; Wagner et al., 2021). Although this better activity than eucalyptol could be attributed to the addition or synergy with other compounds present in the oils as it was suggested in the case of Tenebrio molitor (L) (Coleoptera: Tenebrionidae) and Spodoptera littoralis Boisd (Lepidoptera: Noctuidae) (Lima et al., 2011; Pavela, 2014).
Eucalyptol also shows fumigant toxicity in other beetle pests, such as stored grain insects Sitophilus oryzae L (Coleoptera: Curculionidae), Tribolium castaneum Herbst, Tribolium confusum Jacquelin du Val (Coleoptera: Tenebrionidae), and Rhyzopertha dominica Fabricius (Coleoptera: Bostrichidae) (Lee et al., 2004; Abdelgaleil et al., 2009; Kheloul et al., 2023).
As observed in other insect species, the toxicity induced by eucalyptol in FAW and BB could be attributed to the inhibition of acetylcholinesterase (AChE) enzyme activity, which disrupts the insect’s nervous system, leading to paralysis and eventual death (Abdelgaleil et al., 2009; Picollo et al., 2008).
Fungicidal activity
While previous studies highlight the significant potential of eucalyptol as a fungicide against various phytopathogenic fungi (Morcia et al., 2012; Shukla et al., 2012; Oxenham et al., 2005; Dammak et al., 2019; Jiang et al., 2020), this study is the first to report its fungicidal activity against C. kikuchii and C. sojina. However, our research group previously determined that, at the same concentration of 5 µl/disk, the essential oil extracted from lavender, rich in eucalyptol (34.33%), exhibits potent fungicidal activity against C. kikuchii and C. sojina, with mycelial inhibition halos of 34.0 and 29.5 mm, respectively (Wagner et al., 2021). This indicates a fungicidal activity much superior of the natural oil to that observed for pure eucalyptol. This enhanced activity is likely attributed to additive or synergistic effects with other major components present in the essential oil (Hassan et al., 2020; Yan et al., 2021).
On the contrary, in our experiments, eucalyptol does not exert a significant inhibitory effect on the mycelial growth of S. rolfsii. These results contradict findings by Kottearachchi et al. (2012), who report 100% inhibition of S. rolfsii mycelial growth when treated with high concentrations of eucalyptol (0.5 and 1.5%). However, the same authors note that at lower concentrations, the inhibition percentage decreases to less than 30%.
It is well-established that many essential oils, rich in terpenes, possess the ability to induce alterations in the fungal cell wall, plasma membrane, and mitochondria, thereby substantiating their toxicity (Kishore et al., 2007; Pawar and Thaker, 2006). Indeed, toxicity studies conducted against the phytopathogenic fungus Botrytis cinerea confirm the detrimental effects of eucalyptol on the organelles of this fungus’s cells (Yu et al., 2015). It is plausible that the observed mycotoxicity of eucalyptol against C. kikuchii and C. sojina may be attributed to some of these reasons.
Phytotoxicity
Previous studies have underscored the strong phytotoxicity of eucalyptol against various plant species as lettuce (Qiu et al., 2010), annual ryegrass, radish (Barton et al., 2014), redroot amaranth and annual bluegrass (Shao et al., 2018; Zhou et al., 2019), our preliminary findings suggest that even at high concentrations, eucalyptol is not phytotoxic to soybean seedlings. Consistent with our study on the herbicidal effects of eucalyptol, Vaughn and Spencer (1993), using a similar methodology,
observed that this terpene does not exhibit significant phytotoxicity in soybean seedlings.
It is acknowledged that the phytotoxicity of eucalyptol in other plant species, such as Arabidopsis, potato and onion, is attributed to various causes, such as disruptions in microtubule organization and the inhibition of key processes like mitochondrial respiration, mitosis, and phytohormone production, leading to a negative impact on plant development and growth (Baskin et al., 2004; Verdeguer et al., 2020). Nevertheless, it is conceivable that only very high concentrations of eucalyptol may be detrimental to soybean plants at the seedling stage, opening prospects for its future application in protecting this crop.
CONCLUSIONS
The protection of soybean crops against pest insects and phytopathogenic fungi within the framework of Integrated Pest Management (IPM), based on methods like biological control, planting resistant cultivars, and using biopesticides, has sharply declined in recent decades (Bueno et al., 2023; Bueno et al., 2021; Panizzi, 2013). This decline is primarily attributed to the prevalent use of synthetic pesticides as the main control method, despite the well-known adverse impacts on the environmental and human health. Our findings indicate that eucalyptol has notable potential as an active ingredient for future biopesticide formulations, aiming to reduce, at least partially, the use of synthetic pesticides in soybean cultivation. This potential is primarily based on its insecticidal and fungicidal properties found in our studies. However, as with essential oils, its possible high volatility and rapid degradation, induced by exposure to air, sunlight, moisture, and high temperatures, could significantly reduce its effectiveness. Therefore, the application of eucalyptol may face significant limitations, requiring more frequent and higher quantities of application to the crop, thus increasing costs. These stability issues could be addressed through the implementation of formulations based on nanoencapsulation, which protect the compounds from adverse environmental factors, thereby improving their stability and efficacy (Giunti et al., 2021; Campolo et al., 2018; Gupta et al., 2023; Šunjka and Mechora, 2022). Therefore, we believe further studies are needed to evaluate the economic feasibility and applicability of eucalyptol in large-scale cultivation areas.
Acknowledgments
ACKNOWLEDGMENTS
The authors express their sincere gratitude to the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, Argentina) and the Facultad de Ciencia y Tecnología de la Universidad Autónoma de Entre Ríos (FCyT-UADER, Argentina).
REFERENCES
ABDELGALEIL, S.A.; MOHAMED, M.I.; BADAWY, M.E.; EL-ARAMI, S.A. 2009. Fumigant and contact toxicities of monoterpenes to Sitophilus oryzae (L.) and Tribolium castaneum (Herbst) and their inhibitory effects on acetylcholinesterase activity. J Chem Ecol 35: 518-525. https://doi.org/10.1007/s10886-009-9635-3
ALAOUI, A.; CHRIST, F.; SILVA, V.; VESTED, A.; SCHLÜNSSEN, V.; GONZÁLEZ, N.; GEISSEN, V. 2024. Identifying pesticides of high concern for ecosystem, plant, animal, and human health: A comprehensive field study across Europe and Argentina. Science of the total environment, 948, 174671. https://doi.org/10.1016/j.scitotenv.2024.174671
APARICIO, V.; DE GERÓNIMO, E. 2024. Pesticide pollution in argentine drinking water: A call to ensure safe access. Environmental Challenges, 14, 100808. https://doi.org/10.1016/j.envc.2023.100808
BAGHOUZ, A.; BOUCHELTA, Y.; ES-SAFI, I.; EL BRAHIMI, R.; IMTARA, H.; ALZAIN, M. N.; GUEMMOUH, R. 2024. Biocidal activity of Ziziphora hispanica L and Satureja calamintha Scheele L essential oils against the Callosobruchus maculatus (Fabricius) pest on cowpea seeds during storage. Frontiers in Sustainable Food Systems, 8, 1329100. https://doi.org/10.3389/fsufs.2024.1329100
BARRO, J.P.; NEVES, D.L.; DEL PONTE, E.M.; BRADLEY, C.A. 2023. Frogeye leaf spot caused by Cercospora sojina: A review. Trop Plant Pathol1-12. https://doi.org/10.1007/s40858-023-00583-8
BARTON, A.F.; CLARKE, B.R.; DELL B.; KNIGHT, A.R. 2014. Post-emergent herbicidal activity of cineole derivatives. J Pest Sci87: 531-541. https://doi.org/10.1007/s10340-014-0566-6
BASKIN, T.I.; BEEMSTER, G.T.; JUDY-MARCH, J.E.; MARGA, F. 2004. Disorganization of cortical microtubules stimulates tangential expansion and reduces the uniformity of cellulose microfibril alignment among cells in the root of Arabidopsis. Plant Physiol 135(4): 2279-2290. https://doi.org/10.1104/pp.104.040493
BATISH, D.R.; SINGH, H.P.; KOHLI, R.K.; KAUR, S. 2008. Eucalyptus essential oil as a natural pesticide. For Ecol Manage256(12): 2166-2174. https://doi.org/10.1016/j.foreco.2008.08.008
BHAMRA, G.K.; BORAH, M. 2022. A review on collar rot disease of soybean caused by Sclerotium rolfsii sacc. Int. J Econ Plants9(3): 235-239. http://dx.doi.org/10.23910/2/2022.0470
BIBIANO, C.S.; ALVES, D.S.; FREIRE, B.C.; BERTOLUCCI, S.K.V.; CARVALHO, G.A. 2022. Toxicity of essential oils and pure compounds of Lamiaceae species against Spodoptera frugiperda (Lepidoptera: Noctuidae) and their safety for the nontarget organism Trichogramma pretiosum (Hymenoptera: Trichogrammatidae). Crop Prot 158: 106011 https://doi.org/10.1016/j.cropro.2022.106011
BILLAH, K.M.; HOSSAIN, M.B.; PRINCE, M.H.; SUMON, M.M. 2017. Pathogenicity of Sclerotium rolfsii on different host, and its over wintering survival; A mini review. Int J Adv Agric Sci2:(1).
BORGES, R.S.; ORTIZ, B.L.S.; PEREIRA, A.C.M.; KEITA, H.; CARVALHO, J.C.T. 2019. Rosmarinus officinalis essential oil: A review of its phytochemistry, anti-inflammatory activity, and mechanisms of action involved. J Ethnopharmacol 229: 29-45. https://doi.org/10.1016/j.jep.2018.09.038
BUENO, A.D.F.; PANIZZI, A.R.; HUNT, T. E.; DOURADO, P. M.; PITTA, R. M.; GONÇALVES, J. 2021. Challenges for adoption of integrated pest management (IPM): the soybean example. Neotropical Entomology, 50, 5-20. https://doi.org/10.1007/s13744-020-00792-9
BUENO, A.D.F.; SUTIL, W.P.; JAHNKE, S.M.; CARVALHO, G.A.; CINGOLANI, M.F.; COLMENAREZ, Y.C.; CORNIANI, N. 2023. Biological control as part of the soybean integrated pest management (IPM): Potential and challenges. Agronomy, 13(10), 2532. https://doi.org/10.3390/agronomy13102532
CAI, Z.M.; PENG, J.Q.; CHEN, Y.; TAO, L.; ZHANG, Y.Y.; FU, L.Y.; LONG, Q.D.; SHEN X.C. 2021. 1, 8-Cineole: A review of source, biological activities, and application. J Asian Nat Prod Res23(10): 938-954. https://doi.org/10.1080/10286020.2020.1839432
CAMPOLO, O.; GIUNTI, G.; RUSSO, A.; PALMERI, V.; ZAPPALÀ, L. 2018. Essential oils in stored product insect pest control. Journal of Food Quality, 2018(1), 6906105. https://doi.org/10.1155/2018/6906105
CAMPOS-SOLDINI, M.P.; ZAPATA, L.D.; WAGNER, L.S.; FERNÁNDEZ, E.N.; SAFENRAITER, M.E. 2021. Contribución al estudio de la ecología y biología de Epicauta atomaria (Coleoptera: Meloidae), insecto asociado a cultivos agrícolas en América del Sur. R.I.A 47(3): 367-375.
CAMPOS, J.F.; BERTEINA-RABOIN, S. 2022. Eucalyptol, an all-purpose product.Catalysts, 12(1): 48 https://doi.org/10.3390/catal12010048
CARMONA, M.A.; REIS, E.M.; SAUTUA, F.J. 2016. Sustainable chemical control of main soybean diseases in South America. Current Politics and Economics of South and Central America, 9(1), 107-149.
CAVANAGH, H.M.A.; WILKINSON, J.M. 2002. Biological activities of lavender essential oil. Phytother Res 16(4): 301-308. https://doi.org/10.1002/ptr.1103
CHAHAL, K.K.; KAUR, M.; BHARDWAJ, U.; SINGLA, N.; KAUR, A. 2017. A review on chemistry and biological activities of Laurus nobilis L. essential oil. J Pharmacogn Phytochem6(4): 1153-1161.
CONOVER, W.J. 1999. Practical nonparametric statistics (Vol. 350). John wiley and sons.
DAMMAK, I.; HAMDI, Z.; EL EUCH, S.K.; ZEMNI, H.; MLIKI, A.; HASSOUNA, M.; LASRAM, S. 2019. Evaluation of antifungal and anti-ochratoxigenic activities of Salvia officinalis, Lavandula dentata and Laurus nobilis essential oils and a major monoterpene constituent 1, 8-cineole against Aspergillus carbonarius. Ind Crops Prod128: 85-93. https://doi.org/10.1016/j.indcrop.2018.11.006
DE FREITAS BUENO, R.C.O.; DE FREITAS BUENO, A.; MOSCARDI, F.; POSTALI PARRA, J.R.; HOFFMANN‐CAMPO, C.B. 2011. Lepidopteran larva consumption of soybean foliage: basis for developing multiple‐species economic thresholds for pest management decisions. Pest Manage Sci 67(2): 170-174. https://doi.org/10.1002/ps.2047
FINNEY, D.J. 1971. Probit Analysis. Cambridge University Press, London.
GIUNTI, G.; CAMPOLO, O.; LAUDANI, F.; ZAPPALÀ, L.; PALMERI, V. 2021. Bioactivity of essential oil-based nano-biopesticides toward Rhyzopertha dominica (Coleoptera: Bostrichidae). Industrial crops and products, 162, 113257. https://doi.org/10.1016/j.indcrop.2021.113257
GRAHAM, P.H.; VANCE, C.P. 2003. Legumes: importance and constraints to greater use. Plant Physiol 131(3): 872-877. https://doi.org/10.1104/pp.017004.
GUPTA, I.; SINGH, R.; MUTHUSAMY, S.; SHARMA, M.; GREWAL, K.; SINGH, H.P.; BATISH, D.R. 2023. Plant essential oils as biopesticides: Applications, mechanisms, innovations, and constraints. Plants, 12(16), 2916. https://doi.org/10.3390/plants12162916
HAMZA, M.; BASIT, A.W.; SHEHZADI, I.; TUFAIL, U.; HASSAN, A.; HUSSAIN, T.; HAYAT, H.M. 2024. Global impact of soybean production: A review. Asian Journal of Biochemistry, Genetics and Molecular Biology, 16(2), 12-20. https://doi.org/10.9734/ajbgmb/2024/v16i2357
HARTMAN, G.L.; SINCLAIR, J.B.; RUPE, J.C. 1999. Compendium of soybean diseases. Soybean Disease Compendium; Agricultural Research Service: Washington, D. C., USA, 37-39.
HASSAN, H.A.; GENAIDY, M.M.; KAMEL, M.S.; ABDELWAHAB, S.F. 2020. Synergistic antifungal activity of mixtures of clove, cumin and caraway essential oils and their major active components.J Herb Med24: 100399. https://doi.org/10.1016/j.hermed.2020.100399
HOCH, C.C.; PETRY, J.; GRIESBAUM, L.; WEISER, T.; WERNER, K.; PLOCH, M.; VERSCHOOR, A.; MULTHOFF, G.; DEZFOULI, A.B.; WOLLENBERG, B. 2023. 1, 8-cineole (eucalyptol): A versatile phytochemical with therapeutic applications across multiple diseases. Biomed Pharmacother167: 115467. https://doi.org/10.1016/j.biopha.2023.115467
HUANG, Y.; TAN, J.M.W.L.; KINI, R.M.; HO, S.H. 1997. Toxic and antifeedant action of nutmeg oil against Tribolium castaneum (Herbst) and Sitophilus zeamais Motsch. J Stored Prod Res 33(4): 289-298. https://doi.org/10.1016/S0022-474X(97)00009-X
INTERNATIONAL SEED TESTING ASSOCIATION. 1976. Seed Sci. Technol. 4: 3-39.
JEREZ, P.G.P.; HILL, J.G.; PEREIRA, E.J.; ALZOGARAY, R.A.; VERA, M.T. 2023. Ten years of Cry1Ac Bt soybean use in Argentina: Historical shifts in the community of target and non-target pest insects. Crop Protection, 170, 106265. https://doi.org/10.1016/j.cropro.2023.106265
JIANG, H.; ZHANG, M.; QIN, L.; WANG, D.; YU, F.; LIANG, W.; SONG, C.; GRANATO, D. 2020. Chemical composition of a supercritical fluid (Sfe-CO2) extract from Baeckea frutescens L. leaves and its bioactivity against two pathogenic fungi isolated from the tea plant (Camellia sinensis (L.) O. Kuntze). Plants 9(9): 1119. https://doi.org/10.3390/plants9091119
KHELOUL, L.; ANTON, S.; BREARD, D.; KELLOUCHE, A. 2023. Fumigant toxicity of some essential oils and eucalyptol on different life stages of Tribolium confusum (Coleoptera: Tenebrionidae). Bot Lett 170(1): 3-14. https://doi.org/10.1080/23818107.2021.1982767
KISHORE, G.K.; PANDE, S.; HARISH, S. 2007. Evaluation of essential oils and their components for broad-spectrum antifungal activity and control of late leaf spot and crown rot diseases in peanut. Plant Dis91(4): 375-379. https://doi.org/10.1094/PDIS-91-4-0375
KOTTEARACHCHI, N.S.; SAMMANI, A.; KELANIYANGODA, D.B.; SAMARASEKARA, R. 2012. Anti-fungal activity of essential oils of Ceylon Eucalyptus species for the control of Fusarium solani and Sclerotium rolfsii. Arch. Phytopathol and Plant Prot45(17): 2026-2035. https://doi.org/10.1080/03235408.2012.720469
LAVILLA, M.; IVANCOVICH, A. 2021. Relación entre enfermedades y rendimiento de granos de soja. Agronomía Mesoamericana, 32(2), 479-486. https://doi.org/10.15517/am.v32i2.44057
LEE, B.H.; ANNIS, P.C.; CHOI, W.S. 2004. Fumigant toxicity of essential oils from the Myrtaceae family and 1, 8-cineole against 3 major stored-grain insects. J Stored Prod Res40(5): 553-564. https://doi.org/10.1016/j.jspr.2003.09.001
LEORA SOFTWARE. 2002–2014. POLO-PLUS: A User’s Guide to Probit or Logit Analysis. LeOra Software, Berkeley, CA.
LIMA, R.K.; CARDOSO, M.D.G.; MORAES, J.C.; CARVALHO, S.M.; RODRIGUES, V.G.; GUIMARÃES, L.G.L. 2011. Chemical composition and fumigant effect of essentialoil of Lippia sidoides Cham. and monoterpenes against Tenebrio molitor (L.) (Coleoptera: Tenebrionidae). Ciência e Agrotecnologia, 35, 664-671. https://doi.org/10.1590/S1413-70542011000400004
MORCIA, C.; MALNATI, M.; TERZI, V. 2012. In vitro antifungal activity of terpinen-4-ol, eugenol, carvone, 1, 8-cineole (eucalyptol) and thymol against mycotoxigenic plant pathogens. Food Addit. Contam Part A 29(3): 415-422. https://doi.org/10.1080/19440049.2011.643458
MURÚA, M.G.; DEFAGÓ, V.H.; VIRLA, E.G. 2003. Evaluación de cuatro dietas artificiales para la cría de Spodoptera frugiperda (Lep.: Noctuidae) destinada a mantener poblaciones experimentales de himenópteros parasitoides. Bol San Veg Plagas29: 43-51.
MURÚA, M.G.; VERA, M.A.; HERRERO, M.I.; FOGLIATA, S.V.; MICHEL, A. 2018. Defoliation of soybean expressing Cry1Ac by lepidopteran pests. Insects, 9(3), 93. https://doi.org/10.3390/insects9030093
NICULAU, E.D.S.; ALVES, P.B.; NOGUEIRA, P.C.D.L.; MORAES, V.R.D.S.; MATOS, A.P.; BERNARDO, A.R.; VOLANTE, A.C.; FERNANDES, J.B.; DA SILVA, M.F.G.F.; CORREA, A.G.; BLANK, A.F.; SILVA, A.C.; RIBEIRO, L. 2013. Atividade inseticida de óleos essenciais de Pelargonium graveolens l’Herit e Lippia alba (Mill) NE Brown sobre Spodoptera frugiperda (JE Smith). Quím Nova36: 1391-1394. https://doi.org/10.1590/S0100-40422013000900020
OVERTON, K.; MAINO, J.L.; DAY, R.; UMINA, P.A.; BETT, B.; CARNOVALE, D.; EKESI, S.; MEAGHER, R.; REYNOLDS, O.L. 2021. Global crop impacts, yield losses and action thresholds for fall armyworm (Spodoptera frugiperda): A review. Crop Prot 145: 105641. https://doi.org/10.1016/j.cropro.2021.105641
OXENHAM, S.K.; SVOBODA, K.P.; WALTERS, D.R. 2005. Antifungal activity of the essential oil of basil (Ocimum basilicum).J Phytopathol 153(3): 174-180. https://doi.org/10.1111/j.1439-0434.2005.00952.x
PANDIYAN, G.N.; MATHEW, N.; MUNUSAMY, S. 2019. Larvicidal activity of selected essential oil in synergized combinations against Aedes aegypti. Ecotoxicol Environ Saf174: 549-556. https://doi.org/10.1016/j.ecoenv.2019.03.019
PANIZZI, A.R. 2013. History and contemporary perspectives of the integrated pest management of soybean in Brazil. Neotropical entomology, 42(2), 119-127. https://doi.org/10.1007/s13744-013-0111-y
PAVELA, R. 2014. Acute, synergistic and antagonistic effects of some aromatic compounds on the Spodoptera littoralis Boisd.(Lep., Noctuidae) larvae. Industrial crops and products, 60, 247-258. https://doi.org/10.1016/j.indcrop.2014.06.030
PAWAR, V.C.; THAKER, V.S. 2006. In vitro efficacy of 75 essential oils against Aspergillus niger. Mycoses, 49(4): 316-323. https://doi.org/10.1111/j.1439-0507.2006.01241.x
PICOLLO, M.I.; TOLOZA, A.C.; CUETO, G.M.; ZYGADLO, J.; ZERBA, E. 2008. Anticholinesterase and pediculicidal activities of monoterpenoids. Fitoterapia, 79(4): 271-278. https://doi.org/10.1016/j.fitote.2008.01.005
PLOPER, L.D. 2004. Economic importance of and control strategies for the major soybean diseases in Argentina. (Available at: https://www.cabidigitallibrary.org/doi/full/10.5555/20043096400 verified on November 18, 2024).
QIU, X.; YU, S.; WANG, Y.; FANG, B.; CAI, C.; LIU, S. 2010. Identification and allelopathic effects of 1, 8-cineole from Eucalyptus urophylla on lettuce. Allelopathy J 26(2): 255-264.
RANI, L.; THAPA, K.; KANOJIA, N.; SHARMA, N.; SINGH, S.; GREWAL, A.S.; SRIVASTAV, A.L.; KAUSHAL, J. 2021. An extensive review on the consequences of chemical pesticides on human health and environment. J Cleaner Prod283: 124657. https://doi.org/10.1016/j.jclepro.2020.124657
SAUTUA, F.J.; GONZALEZ, S.A.; DOYLE, V.P.; BERRETTA, M.F.; GORDÓ, M.; SCANDIANI, M.M.; RIVAROLA, M.L.; FERNANDEZ, P.; CARMONA. M.A. 2019. Draft genome sequence data of Cercospora kikuchii, a causal agent of Cercospora leaf blight and purple seed stain of soybeans. Data Brief 27: 104693. https://doi.org/10.1016/j.dib.2019.104693
SEKHAR, J.C.; MISHRA, J.P.; PRASAD, R.; REDDY, V.P.; KUMAR, S.; THAKUR, A.; PAL, J. 2020. Isolation and in vitro evaluation of biocontrol agents, fungicides and essential oils against stem blight of tomato caused by Sclerotium rolfsii (Curzi) CC Tu and Kimber. J Pharmacogn Phytochem 9(3): 700-705.
SEQUIN, C.J.; APPELHANS, S.C.; HEIS, M.S.; TORRENT, W.A.; TROSSERO, J.A.; CATALÁN, C.A.; ACEÑOLAZA, P.G. 2023. Antifungal and toxicological evaluation of the alkaloids fraction from Neltuma nigra leaves. Biocatalysis and Agricultural Biotechnology, 54, 102914. https://doi.org/10.1016/j.bcab.2023.102914
SHAO, H.; HU, Y.; HAN, C.; WEI, C.; ZHOU, S.; ZHANG, C.; ZHANG, C. 2018. Chemical composition and phytotoxic activity of Seriphidium terrae‐albae (Krasch.) Poljakov (Compositae) essential oil. Chem Biodivers15(11): e1800348. https://doi.org/10.1002/cbdv.201800348
SHUKLA, R.; SINGH, P.; PRAKASH, B.; DUBEY, N.K. 2012. Antifungal, aflatoxin inhibition and antioxidant activity of Callistemon lanceolatus (Sm.) Sweet essential oil and its major component 1, 8-cineole against fungal isolates from chickpea seeds. Food Control 25(1): 27-33. https://doi.org/10.1016/j.foodcont.2011.10.010
ŠUNJKA, D.; MECHORA, Š. 2022. An alternative source of biopesticides and improvement in their formulation—recent advances. Plants, 11(22), 3172. https://doi.org/10.3390/plants11223172
TAHIRI, N.E.H.; SAGHROUCHNI, H.; HAMAMOUCH, N.; KHOMSI, M.E.; ALZAHRANI, A.; SALAMATULLAH, A.M.; BADIAA, L.; LRHORFI, L.A. 2022. Treatment with glyphosate induces tolerance of Citrus pathogens to glyphosate and fungicides but not to 1, 8-cineole. Molecules 27(23): 8300. https://doi.org/10.3390/molecules27238300
TRIPATHI, A.K.; MISHRA, S. 2016. Plant monoterpenoids (prospective pesticides). In Ecofriendly Pest Management for Food Security. Academic Press. 507-524 pp. https://doi.org/10.1016/B978-0-12-803265-7.00016-6
USDA. 2022. Soybean Explorer [Database]. (Available at: https://ipad.fas.usda.gov/cropexplorer/cropview/commodityView.aspx?cropid=2222000 verified on November 18, 2024).
Usseglio, V.L.; Dambolena, J.S.; Zunino, M.P. 2022. Can Essential Oils Be a Natural Alternative for the Control of Spodoptera frugiperda? A Review of Toxicity Methods and Their Modes of Action. Plants 12(1): 3. https://doi.org/10.3390/plants12010003.
VAUGHN, S.F.; SPENCER, G.F. 1993. Volatile monoterpenes as potential parent structures for new herbicides. Weed Sci 41(1): 114-119. https://doi.org/10.1017/S0043174500057672
VERDEGUER, M.; SÁNCHEZ-MOREIRAS, A.M.; ARANITI, F. 2020. Phytotoxic effects and mechanism of action of essential oils and terpenoids. Plants 9(11): 1571 https://doi.org/10.3390/plants9111571
WAGNER, L.S.; CAMPOS-SOLDINI, M.P. 2022. Fumigant insecticidal activity of plant essential oils against pest blister beetle Epicauta atomaria (Germar) (Coleoptera: Meloidae). J Plant Dis Prot 129(4): 783-789. https://doi.org/10.1007/s41348-022-00580-0
WAGNER, L.S.; SEQUIN, C.J.; FOTI, N.; CAMPOS-SOLDINI, M.P. 2021. Insecticidal, fungicidal, phytotoxic activity and chemical composition of Lavandula dentata essential oil. Biocatalysis and Agricultural Biotechnology, 35, 102092. https://doi.org/10.1016/j.bcab.2021.102092
YAN, J.; NIU, Y.; WU, C.; SHI, Z.; ZHAO, P.; NAIK, N.; YUAN, B. 2021. Antifungal effect of seven essential oils on bamboo. Advanced Composites and Hybrid Materials, 4(3), 552-561. https://doi.org/10.1007/s42114-021-00251-y
YU, D.; WANG, J.; SHAO, X.; XU, F.; WANG, H. 2015. Antifungal modes of action of tea tree oil and its two characteristic components against Botrytis cinerea.J Appl Microbiol 119(5): 1253-1262. https://doi.org/10.1111/jam.12939
ZHOU, S.; WEI, C.; ZHANG, C.; HAN, C.; KUCHKAROVA, N.; SHAO, H. 2019. Chemical composition, phytotoxic, antimicrobial and insecticidal activity of the essential oils of Dracocephalum integrifolium. Toxins 11(10): 598. https://doi.org/10.3390/toxins11100598
Información adicional
FUNDING: This research received no external funding.
Información adicional
redalyc-journal-id: 864