
Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial 4.0 Internacional.
DOI: https://doi.org/10.58149/d3qn-bx20
Abstract: Hirschfeldia incana (L.) Lagr.-Foss, an exotic weed in Argentina, is well adapted to different climate, soil, and crop conditions, and is particularly abundant in the southern region of Santa Fe province. The design of rational weed management strategies requires detailed information on biotypes behavior and growth characteristics. The objectives of this work were to study: 1) the phenology and growth, and 2) the germination response under varying light and temperature conditions of different populations (L1 and L2) of H. incana from the southern region of Santa Fe Province. Under semi-controlled conditions, seeds from L1 and L2 populations were used to assess phenological parameters, growth, and seed production. In two additional experiments, we evaluated germination responses: Experiment 1) assessed the effect of blue, green, red, and white light, and darkness on germination (populations L1 and L2); Experiment 2) evaluated the effect of temperature (7–29°C) on germination (populations L1, L2, and a third population designated FH1). Plants from populations L1 and L2 exhibited similar phenological development but differed in rosette diameter (20 ± 3.3 cm vs. 26 ± 2.4 cm, respectively), resulting in differences in leaf area and greater silique and seed production output in the L2 population. In both populations, a photoblastic index near zero indicated light-insensitive seeds. Seeds germinated across a broad temperature range above 10°C, with maximum germination occurring between 19°C and 30°C. Additionally, H. incana seed viability declined two years after harvest. Further research is needed to better understand the mechanisms regulating physiological dormancy in H. incana and to develop strategies to enhance its germination independently of light.
Keywords: mustard weeds, cruciferous, photoblastism.
Resumen: Hirschfeldia incana (L.) Lagr.-Foss es una maleza exótica en Argentina y se adapta bien a diferentes condiciones de clima, suelo y cultivos. Es particularmente abundante en el sur de la provincia de Santa Fe. Es de interés estudiar los biotipos locales que contribuyan a diseñar estrategias racionales de manejo de malezas. Los objetivos del trabajo fueron estudiar: 1) la fenología y el crecimiento, y 2) la germinación bajo diferentes condiciones lumínicas y térmicas, de diferentes poblaciones de H. incana pertenecientes a la región sur de la provincia de Santa Fe. Se tomaron plantas de H. incana de dos lotes localizados en el departamento Caseros, provincia de Santa Fe, para obtener semillas (poblaciones L1 y L2). En condiciones semicontroladas, dichas semillas se utilizaron para evaluar parámetros fenológicos, el crecimiento y la producción de semillas de ambas poblaciones. En dos experimentos adicionales se evaluaron: E1) el efecto de la luz azul, verde, roja y blanca, así como de la oscuridad (poblaciones L1 y L2), y E2) el efecto de la temperatura (entre 7 y 29 °C), sobre la germinación de las semillas utilizando una tabla de gradiente térmico (poblaciones L1, L2 y una tercera llamado FH1). Las plantas de las poblaciones L1 y L2 presentaron similar fenología, pero se diferenciaron en el diámetro de la roseta (20 ± 3,3 cm vs. 26 ± 2,4 cm, respectivamente), lo que generó un área foliar diferente, y una mayor respuesta de la población L2 en producción de silicuas y semillas. Para ambas poblaciones, el índice de fotoblastismo fue cercano a cero, lo que indica que las semillas son insensibles a la luz. Las semillas germinaron en un amplio rango de temperaturas superiores a 10 °C y con valores máximos de germinación de 19 °C a 30 °C. Además, la viabilidad de la semilla de H. incana disminuyó dos años después de la cosecha. Es importante continuar con estudios que permitan comprender mejor los mecanismos que están regulando la latencia fisiológica en H. incana y las estrategias para incrementar su germinación sin depender de la luz.
Palabras clave: nabolsas, nabillo, crucíferas, fotoblastismo..
INTRODUCTION
Hirschfeldia incana (L.) Lagr.-Foss is an annual, facultative biannual, or perennial species of Mediterranean origin, classified within the Brassicaceae family and considered exotic in Argentina (SENASA, 2018; Troiani, 2016; Marushia et al., 2012). Due to its morphological similarity to other species within the same botanical family, it is often grouped with members of the so-called “mustard family”, such as Brassica rapa L. (Turnip), Raphanus sativus L. (Radish), and Rapistrum rugosum L. (Bastard cabbage) (SENASA, 2018).
Hirschfeldia incana is worldwide associated with sub-humid and arid climates, typical occurring with sandy and loose soils, and not necessarily linked to cultivated areas (Cousens et al., 1993; Marushia et al., 2012; Hani et al., 2017). However, Cousens et al. (1993) noted that in Australia, the species predominates in the southern plateaus -regions characterized by lower-temperatures, high rainfall, and undisturbed soils. In Argentina, H. incana has been reported across much of the national territory (Flora del Cono Sur, 2024). Nevertheless, the literature highlights three main and geographically distant regions where the species is most frequently found: the southwest of Buenos Aires Province, the center-east of Córdoba Province, and the south of Santa Fe province (Prina, 1995; Vigna and Mendoza, 2015; Ustarroz, 2017). In the south of Santa Fe Province, Crespo (2025) reported that H. incana is primarily associated with agricultural fields under humid climates and silty soils, where it grows alongside with crops -mainly soybeans, corn, and wheat- almost entirely under no-till systems (Puricelli and Tuesca, 2017; AAPRESID, 2019).
Several strategies are carried out to control H. incana, with herbicide application being the most widely used tool in current agricultural production systems (Radosevich et al., 2007; Puricelli and Tuesca, 2017). However, relying almost exclusively on herbicides for weed control has led to changes in plant communities under no-till systems (Puricelli and Tuesca, 2017). Moreover, the increased use -both in quantity and frequency- of specific herbicides has contributed to the emergence and spread of herbicide-resistant biotypes (Vigna and Mendoza, 2015; Ustarroz, 2017). Despite its widespread global distribution, herbicide-resistant H. incana biotypes have only been documented in a few regions of Argentina. Biotypes resistant to 2,4-D and metsulfuron-methyl have been identified in the southwest of Buenos Aires Province (Vigna and Mendoza, 2015), while resistance to glyphosate and 2,4-D has been reported in the central-eastern region of Córdoba Province (Ustarroz, 2017). Recently, Crespo (2025) contributed to defining the distribution area of H. incana in the northern sector of the core region of the Argentine Humid Pampas, with its epicenter in Caseros County, southern Santa Fe Province. In this area, H. incana poses a significant threat to crop production (Vigna and Mendoza, 2015; Ustarroz, 2017). Therefore, studying the phenology and physiology of these biotypes is a key step for understanding the biological mechanisms driving population dynamics in the region. This knowledge is crucial for developing rational and effective management strategies for the species.
As in the vast majority of spermatophytes, the seed is the primary unit of propagation for H. incana. Once released from the mother plant, seeds may travel short or long distances before becoming buried and incorporated into the soil seed bank. From that point on, they are subject to environmental factors for variable periods until favorable conditions are given for germination and establishment. It is well known that environmental factors such as humidity, temperature, and light can modulate seed germination and subsequent plant development (Bewley, 1997; Mira et al., 2019). It has even been reported that within the same species, seeds from different populations or geographic origins may exhibit distinct germination requirements (Cousens et al., 1993; Acosta et al., 2021). Accordingly, studying the germination physiology of H. incana seeds in response to environmental stimuli is a primary step for identifying the conditions that promote successful germination and seedling establishment. This represents the starting point for understanding the species’ population dynamics within the seed bank, as well as its dispersal, colonization, and integrated management (Radosevich et al., 2007). The objectives of the present study were to investigate: 1) the phenology and growth, and 2) the germination response under different light and temperature conditions of H. incana populations from the southern region of Santa Fe Province. The results will contribute to a better understanding of local H. incana populations and support future research on its biology and management. Furthermore, advancing studies on morphological and phenological characterization of the different H. incana biotypes is not only important for expanding knowledge of the species itself but also critical for its effective management and control within agricultural production systems.
MATERIALS AND METHODS
Characterization of Hirschfeldia incana using phenological parameters
Seed collection
In September 2018, field surveys were conducted along rural roads in Caseros County, Santa Fe province, to randomly identify plots infested with H. incana. Plants from these plots were sampled to obtain seeds. Two highly infested agricultural plots (33° 3’53.50”S 61°24’12.48”W; 33° 2’17.99”S 61°21’25.51”W) were selected and ten rosette-stage plants -with no more than four fully expanded leaves- were collected from each site. These were designated as populations L1 and L2. Each plant was individually transplanted into a 10 L pots to complete its life cycle. The growing medium consisted of a 3:1 soil-to-perlite mixture. In February-March 2019, siliques were manually harvested from each plant, and were threshed to extract the seeds, which were then cleaned to remove silique residues. The seeds from each plant and population were stored separately at room temperature in a cool, dry place.
Experimental setup
In November 2019, 20 seeds from each of the populations (L1 and L2) were sown in 3.5 L pots. Once seedlings developed 3-4 fully expanded leaves, seven seedlings from L1 and eight seedlings from L2 were transplanted —root system and surrounding soil— into 15 L pots. These pots were placed outdoors under natural light and temperature conditions, with supplemental sprinkler irrigation, and were monitored until the end of the growth cycle. The substrate used was the same 3:1 soil-to-perlite mix. Between mid-February and mid-March, seeds from each population were manually harvested. Weather data from the Villarino Experimental Station (School of Agrarian Sciences, UNR, Zavalla) indicated average monthly temperatures of 22.8, 23.3, 24.1, 23.5, and 23.1°C from November 2019 to March 2020, respectively.
Data recording and analysis
For each population, the percentage of seedling emergence and days to emergence were recorded. For each individual plant, days to the onset of stem elongation and flowering (appearance of expanded/visible petals) were recorded from the day of germination. In addition, the following traits were measured: final rosette diameter, total number of leaves, number of basal branches, number of racemes on the main stem, and maximum plant height measured on the main stem. Moreover, the weight of three 100-seed samples (P100) per plant was determined to estimate the average seed production of L1 and L2.
Statistical analyses were conducted using InfoStat software, version 2017 (Di Rienzo et al., 2017). All data were an analyzed using analysis of variance (ANOVA) and mean comparisons were performed using Fisher’s least significant difference (LSD) test (p≤0.05) (Di Rienzo et al., 2017).
Effect of light type and temperature on Hirschfeldia incana seed germination
Experimental material
Seeds from populations L1 and L2 harvested in February-March 2019 were stored at room temperature in a dark, dry environment, protected from humidity and temperature extremes. These seeds were used to assess the effects of light and temperature on germination. In December 2020, additional H. incana rosettes were collected from the same area where populations L1 and L2 were collected, forming a third population designated FH1. These plants were cultivated under similar conditions to L1 and L2, and in March 2021, siliques were harvested and cleaned to obtain seeds.
Experimental setup
Two experiments (E1 and E2) were conducted to evaluate seed germination responses to different light wavelengths and temperature regimes.
E1: Conducted in December 2019, this experiment evaluated the germination of L1 and L2 seeds under four light treatments and darkness. Light was supplied by 6 W LED lamps emitting specific wavelengths: blue (440-500 nm), green (500-570 nm), red (620-720 nm), and white (400-750 nm) (table 1). Each treatment included three replicates of 30 seeds per population. Seeds were placed in Petri dishes on filter paper moistened with distilled water and incubated for 11 d at 25 ± 2°C with a 12-hour photoperiod. In the dark treatment, dishes were wrapped with aluminum foil and kept under the same temperature and moisture conditions. Each replicate received only one light treatment during the incubation period.
E2: Conducted in December 2020, this experiment assessed germination across a thermal gradient. Initially, L1 and L2 seeds harvested in February-March 2019 were tested, followed by a second run using seeds harvested in February-March 2020. Germination was evaluated at 10 temperatures (10, 13, 16, 19, 21, 24, 25, 26, 28 and 29°C) using a thermal gradient plate (table 1). Each treatment consisted of four replicates of 11 seeds per population, placed on moistened filter paper. This number of seeds was selected to ensure that the targeted regime temperature was achieved for each sample, thereby avoiding mixed temperature results. Distilled water was used in all germination experiments. Incubation lasted 7 days without supplemental light.
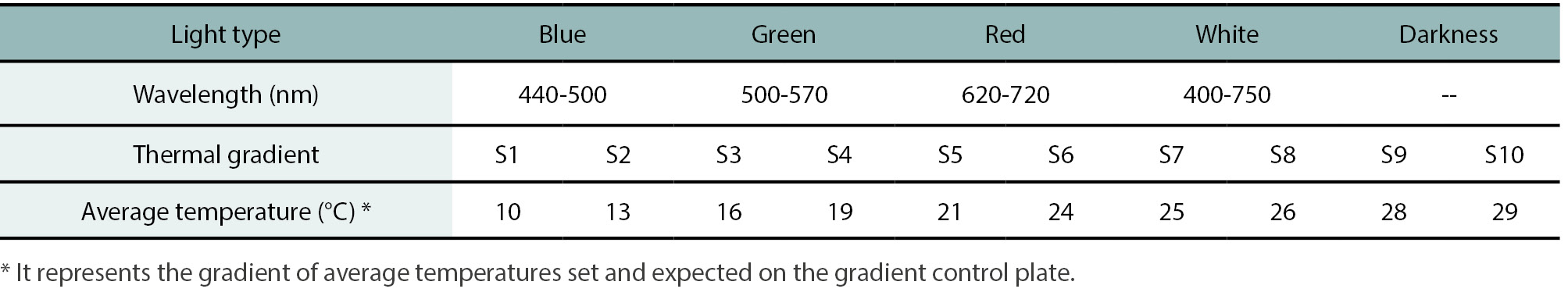
Table 1.
Light type and thermal gradient treatments used to germinate seeds of Hirschfeldia incana populations L1, L2 and FH1 for experiments 1 and 2.
Data collection and analysis
A seed was considered germinated when radicle protrusion through the seed coat was observed. In E1, germination was recorded at 2, 3, 6, 7 and 11 d after starting incubation. In the dark treatment, counting was performed on day 11 to avoid light exposure. In E2, germination was recorded at the end of the experiment (day 7). The results were expressed as the percentage of viable seeds that germinated (%G) (González-Amaya et al., 2018).
In E1, mean germination speed (VMG) and mean germination time (TMG) were calculated using the formulas suggested by Ranal and García de Santana (2013), cited in González-Amaya et al. (2018):
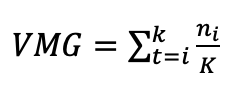
where
ni = number of seeds germinated on day i; K = duration of the germination test (days).
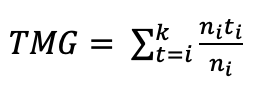
where, ni = number of seeds germinated on day i; ti: days after sowing when ni seeds germinated.
Additionally, the photoblastic index (FI) was calculated following Castillo et al. (2013):
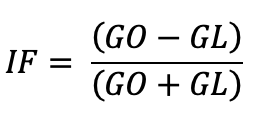
where, GO is the percentage of seeds germinated in darkness, and GL is the percentage of seeds germinated in light.
Results from E1 and E2 were analyzed by ANOVA, and mean comparisons were performed using Fisher’s LSD test (p≤0.05) with InfoStat version 2017 (Di Rienzo et al., 2017).
RESULTS AND DISCUSSION
Phenological characterization of Hirschfeldia incana
The phenological characterization was conducted on individual plants grown in pots and under semi-controlled conditions. Seedling emergence was low in both populations, reaching 35% and 40% for L1 and L2, respectively. This low emergence rate may indicate the presence of some degree of seed dormancy, suggesting that additional stimuli and/or mechanisms may be required to overcome germination constrains. No significant differences (p<0.05) were observed between populations in the number of days required to reach key phenological stages (emergence, stem elongation, and flowering) (table 2). However, phenotypic differences were observed in several morphological traits. The L1 population exhibited a significantly (p<0.05) lower average number of leaves per plant and a smaller average rosette diameter compared to L2, suggesting smaller plant biomass in L1 compared to L2 (not measured). No significant differences were found in plant height, the number of basal branches, or the number of nodes in the main stem (table 3). The average number of total siliques per plant differed significantly between populations (p<0.05), with L2 producing a higher number of siliques (figure 1). In contrast, the proportion of vain (aborted) siliques did not differ significantly between L1 and L2, accounting for 11.3 ± 4.5 and 10.6 ± 2.9% of total siliques, respectively. As a result, the L2 population showed a significantly higher total seed production than L1 (figure 1). Thus, a greater number of leaves and larger rosette diameter observed in L2 plants (table 3) may have contributed to increase leaf area and, consequently, enhanced light interception and photosynthetic capacity. Since whole plant biomass could not be measured due to the prolonged silique maturation period and silique dehiscence, estimating the harvest index was not feasible. However, rosette diameter and leaf number can serve as proxies for plant biomass to approximate a partition index. Using the number of leaves as a proxy, the L2 population showed slightly better performance in terms of seed weight. In contrast, using rosette diameter suggested that L1 was slightly more efficient in seed and silique production (data not shown). Further studies are needed to determine whether these trends reflect plasticity traits among populations.
The average P100 was 0.037 ± 0.0013 g and 0.0372 ± 0.0029 g for the L1 and L2 population, respectively, with no statistically significant difference between them (p<0.05). In the L1 population, P100 values ranged between 0.026 and 0.041 g, while in L2 they ranged from 0.022 to 0.045 g. Based on the P100 values and the seed production per population (figure 1), the average number of seeds per plant was calculated as 7,135 for L1 and 10,075 for L2. Hani et al. (2017) reported that H. incana can produce 14,000 seeds per plant when grown in isolation, with P100 values ranging from 0.03 to 0.07 g. The growth conditions in the present study differed somewhat from those in Hani et al. (2017), particularly because the plants in our experiment completed their cycle in pots, which may have restricted root development. Nevertheless, both studies report similar average P100 values and comparable seed production per plant. Although the average number of seeds per plant varied widely between the populations, P100 values remained similar. This may be explained by the capacity of H. incana to produce seeds in a wide range of sizes under certain conditions. The presence of a greater number of smaller seeds can increase the total seed number, while having little impact on the average seed weight (P100).
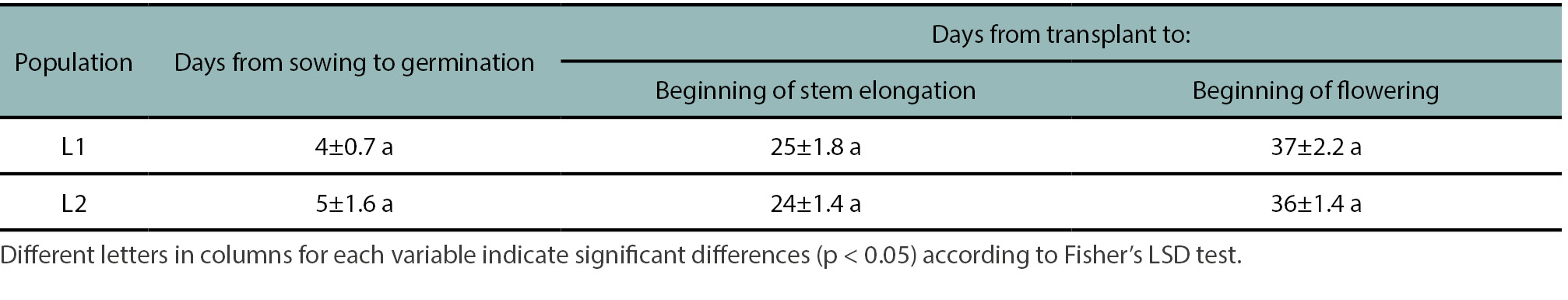
Table 2.
Days and standard error of mean to reach three different phenological stages of two Hirschfeldia incana populations (L1 and L2) from the south of Santa Fe province.

Table 3.
Morphological description of two Hirschfeldia incana populations (L1 and L2) from the south of Santa Fe province.
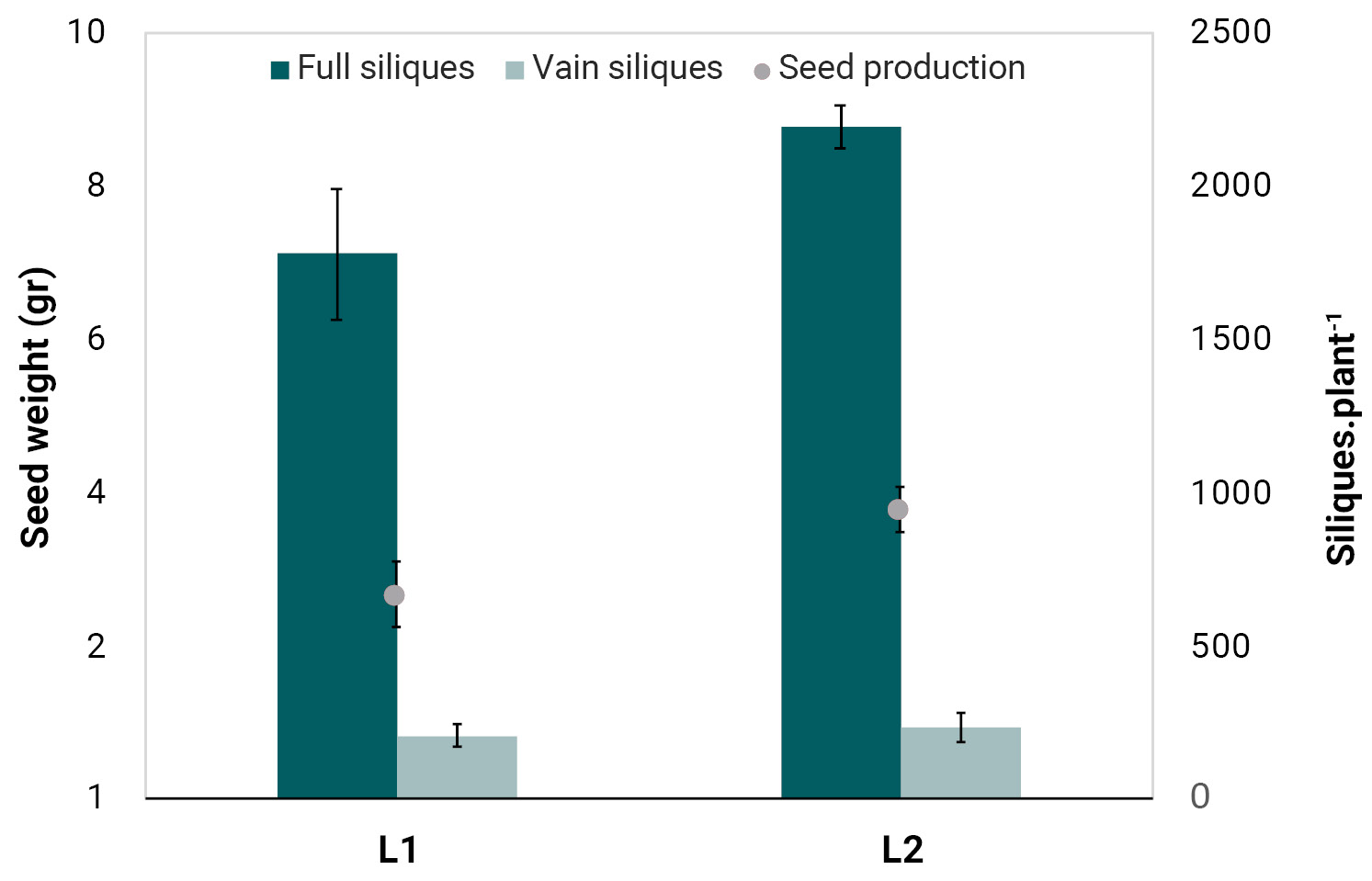
Figure 1.
Average seed production and number of full and vain siliques per plant of two Hirschfeldia incana populations (L1 and L2) from the south of Santa Fe province.
Light and temperature effects on seed germination of Hirschfeldia incana
Experiment 1: The germination response under light and dark conditions was evaluated. After 11 days of incubation under light, total germination differed significantly between populations (p<0.05), with higher germination in L2 (59 ± 4%) than in L1 (42 ± 3%). Germination was consistently higher in L2 across all light treatments; however, significant differences (p<0.05) between populations were only observed under blue and red light (table 4). In L1, the highest germination occurred under green light (53 ± 7%), whereas in L2, it was under blue light (71 ± 6%). Both values were significantly higher than those under dark conditions (L1: 38 ± 2% and L2: 49 ± 13%) (table 4). In a similar study on Brassicaceae species, Cousens et al. (1993) reported that germination in darkness was generally half or even less than that under light. Specifically, for H. incana, they found a light preference for germination, particularly in seeds from undisturbed soils (e.g., pastures), as opposed to seeds from cultivated soils.
The VMG (velocity of mean germination) followed the same trend as total germination (%G), being consistently higher in L2 than in L1 across all light treatments (table 4). Only in L2 did VMG values differed significantly among light treatments. The TMG (time to mean germination) was consistent with VMG patterns and was always lower in L2 than in L1 under all light conditions. Under darkness, TMG did not differ significantly from light treatments, although this varied between populations (table 4). In L2 no significant differences in TMG were found between light and dark treatments. In contrast, for L1, TMG under darkness was statistically similar (p>0.05) to green and white light treatments but differed significantly (p<0.05) from red and blue light treatments (table 4). Niño-Hernández et al. (2020) related small TGM differences in Amaranthus hybridus under different light conditions to simultaneous germination of seeds under both light and dark conditions. A similar pattern was observed in H. incana in our study: although seeds began germinating after 7 d in darkness and between 3 and 5 d under light, these values did not differ statistically (p<0.05) between treatments.
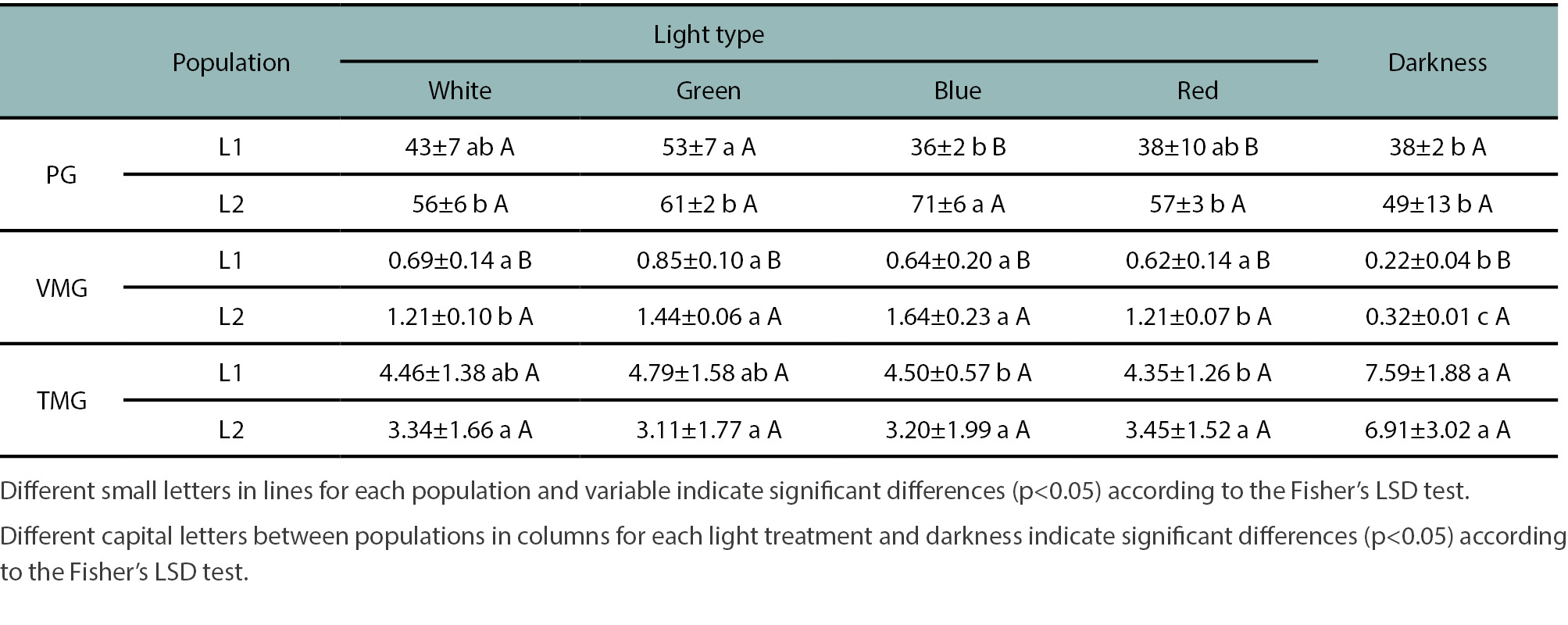
Table 4.
Effect of different types of light and darkness on the percentage of germination (PG), the average germination speed (germinated seeds/d) (VMG) and the average germination time (days) (TMG) of two Hirschfeldia incana populations (L1 and L2) from southern Santa Fe province.
Photoblastism is a phenomenon often associated with seed persistence in the soil, acting as a strategy whereby small seeds only germinate when close to the surface, where light quality is sufficient for successful emergence before reserves are depleted (Castillo et al., 2013; Osuna Fernández et al., 2017). The (IF) ranges from +1 (negative photoblastism, where light inhibits germination) to -1 (positive photoblastism, where light stimulates germination), with IF = 0 indicating not photoblastic (Castillo et al., 2013). In the present study, the IF values were very close to neutral for both populations (-0.171 to 0.03 in L1 and -0.185 to -0.064 in L2), suggesting low sensitivity to light and which supporting the similar germination percentages observed under dark and white light conditions. For comparison, seeds of Eschscholzia californica Cham. and Amaranthus hybridus, which had negative IF values, showed inhibited germination under light and improved performance in darkness (Castillo et al., 2013; Niño-Hernández et al., 2020). These species also showed a higher VMG in darkness (Niño-Hernández et al., 2020). In contrast, in the present study, H. incana seeds with near-neutral IF values exhibited lower VMG in darkness. These results suggest that H. incana may require additional environmental cues or specific light conditions for optimal germination, representing previously unreported behavior for this species (Castillo et al., 2013).
Experiment 2: This experiment evaluated the germination response to temperature. Germination of L1 and L2 seeds did not exceed 5% across all temperature treatments. Tetrazolium viability test (ISTA, 2016) confirmed that these ungerminated seeds were non-viable (data not shown). It is important to note that since the seed harvest (February-March 2019), germination had been tested at several point: during phenological characterization studies (November 2019), Experiment 1 (December 2019) and Experiment 2 (December 2019 and 2020, respectively). The low initial emergence (35% for L1 and 40% for L2) and the moderate germination observed in E1 (42% for L1 and 59% for L2) suggest the presence of intrinsic dormancy mechanisms and/or specific germination requirements. These results, together with the sharp decline in viability over time, indicate short seed longevity, with viability nearly absent in E2, two years post-harvest. These findings align with studies by Mira et al. (2019) and Castro et al. (2015). Mira et al. (2019) related H. incana seed viability loss to seed size and water content, highlighting that environmental conditions during seed development can influence seed composition (e.g., lipid content), and hereby affect water levels and longevity. Meanwhile, Castro et al. (2015) reported lower germination percentages in seeds harvested shortly before the experiment (47%) compared to those stored for two years (61%), suggesting a dual dormancy strategy in H. incana, where some seeds germinate soon after dispersal, while others remain dormant to ensure population persistence over time.
Given the low viability of the original pool of seeds, the Experiment 2, temperature response, was repeated using seeds harvested in February-March 2020. Germination increased with temperature, reaching a maximum of 100% at 29°C (treatment S10; table 5). Significant differences (p<0.05) were found across three thermal ranges: S01 (10°C): 11% germination (G); S02 - S03 (13-16°C): 70-77% G; and S04 - S10 (19-29°C): 90-100% G (table 5). Thus, the lowest and highest temperatures resulted in the lowest and highest germination percentages, respectively. Intermediate germination (~50%) was inferred between 10 and 13°C. Minimum and maximum temperature thresholds for germination could not be precisely defined under the conditions tested. Nonetheless, the methodology used allowed to characterize germination response to temperature. Notably, this experiment was conducted in the absence of light, suggesting that temperature and light may act independently in regulating H. incana germination, and confirming the species broad thermal germination range. Castro et al. (2015) reported consistent germination (80-82%) at temperatures between 20 and 35°C. However, these authors were unable to perform viability tests due to small seed size. Cousens et al. (1993) similarly found increasing germination of H. incana with temperature, reaching 90% at 25-30°C, and 98% at 35°C. Their comparison across Brassicaceae species demonstrate diverse temperature and light requirement for germination. For example, species like Sinapis arvensis and Sisymbrium erysimoides required light at low temperatures but maintained high germination in darkness at higher temperatures.
The results in this study enhance our understanding of H. incana, providing valuable information on plant phenology, seed longevity, and germination behavior under different light and temperature conditions. These environmental factors play a significant role in modulating germination and subsequent phenological stages (Cousens et al., 1993). Moreover, exploring the regulation and natural control exerted by such factors contributes to the development of integrated weed management strategies by improving predictions of population dynamics in response to environmental cues.
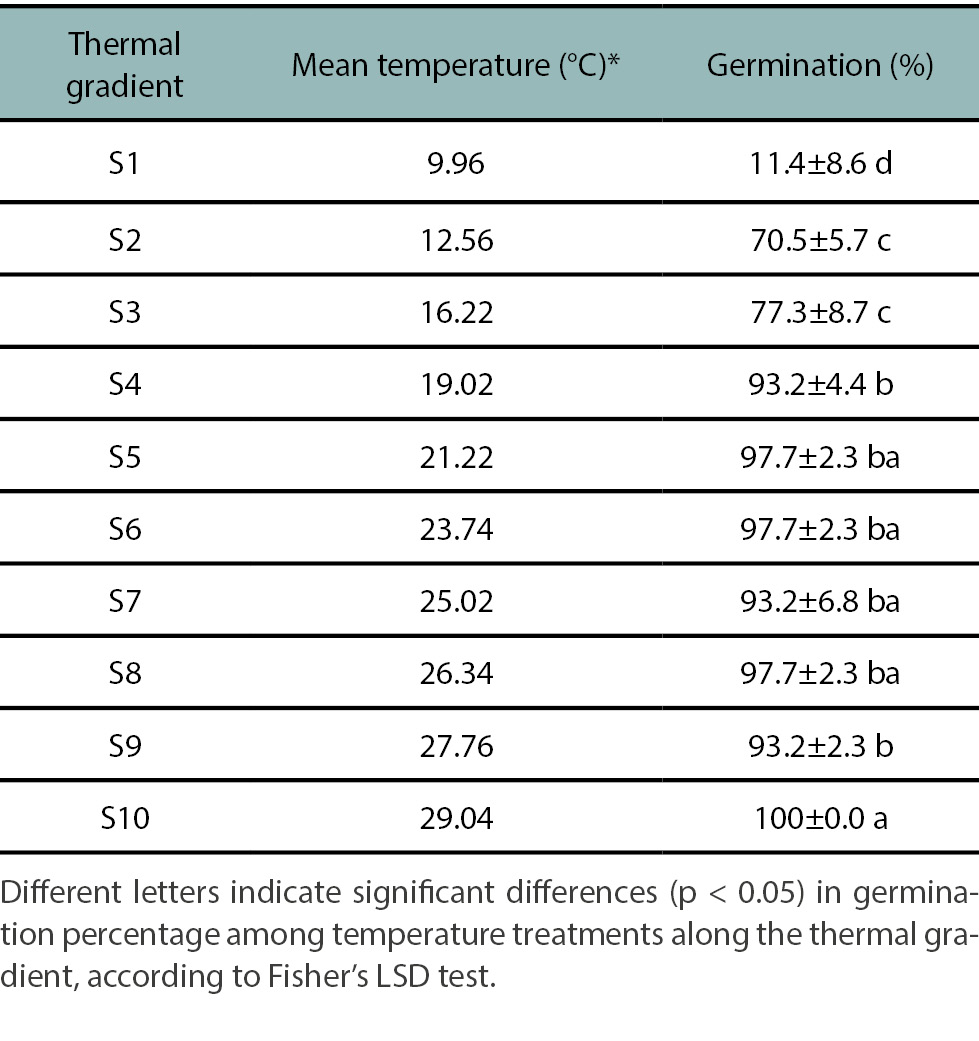
Table 5.
Average temperatures of each temperature measured on the gradient plate and percentage of germination of viable seeds of the Hirschfeldia incana FH1 population by treatment.
CONCLUSION
This study enabled the characterization of two H. incana populations from southern Santa Fe. Both populations exhibited similar phenological development; however, differences in vegetative leaf area may have contributed to variations in silique and seed production. An (IF) close to zero was recorded for both populations, indicating that the seeds are light-insensitive (i.e., neutral or non-photoblastic) and capable of germination regardless of light presence. The results further demonstrated that H. incana seeds can germinate over a broad temperature range above 10°C, with optimal germination occurring between 19°C and 30°C. Additionally, a marked decline in seed viability was observed two years after harvest. These findings underscore the importance of further research into the physiological dormancy mechanisms of H. incana as well as strategies to promote germination without reliance on light stimuli.
Acknowledgments
ACKNOWLEDGMENTS
This project was partially funded by the National University of Rosario (Project No 80020180100006UR). We thank Franco Herranz, Martina Meira, and the members of the Weed Science Team at the Faculty of Agricultural Sciences, National University of Rosario, for their valuable support and collaboration throughout the study. All content was critically reviewed and validated by the authors to ensure its scientific accuracy and integrity.
REFERENCES
AAPRESID. 2019. Evolución de siembra directa en Argentina campaña 2018/19. Asociación Argentina de Productores en Siembra Directa. Rosario, Argentina. (Available at: https://www.aapresid.org.ar/blog/evolucion-de-siembra-directa-en-argentina-campana-2018-19/ verified on October 28, 2025).
ACOSTA, M.C.; ALCARAZ, M.L.; SCARAMUZZINO, R.L.; MANFREDA, V.T. 2021. Fisiología de la germinación de Rhodophiala bifida. Revista FAVE Ciencias Agrarias 19 (1), 159-173.
BEWLEY, J.D. 1997. Seed germination and dormancy. Plant Cell. 9, 1055-1066.
CASTILLO, M.L.C.; BUSTAMANTE, R.O.; PEÑA-GÓMEZ, F.T.; GUTIÉRREZ, V.L.; REYES, C.A.; ARREDONDO-NÚÑEZ, A.; MAREY, M. 2013. Negative photoblastism in the invasive species Eschscholzia californica Cham. (Papaveraceae): Patterns of altitudinal variation in native and invasive range. Gayana. Botánica, 70 (2), 331-337. https://dx.doi.org/10.4067/S0717-66432013000200010
COUSENS, R.; BAWEJA, R.; VATHS, J.; SCHOFIELD, M. 1993. Comparative biology of cruciferous weeds: a preliminary study. Proceedings of the Tenth Australian Weeds Conference, Brisbane, 376-380.
CRESPO, R.J. 2025. Presencia de Hirschfeldia incana (L.) Fagr.-Foss. En el centro de la Pampa Húmeda Argentina. Malezas (ASACIM). Aceptado para publicación.
DI RIENZO, J.A.; CASANOVES, F.; BALZARINI, M.G.; GONZALEZ, L.; TABLADA, M.; ROBLEDO, C.W. 2017. InfoStat versión 2017. Centro de Transferencia InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. (Available at: http://www.infostat.com.ar verified on October 10, 2025).
FLORA DEL CONO SUR. 2024. Catálogo de las Plantas Vasculares. Buenos Aires: Instituto de Botánica Darwinion. (Available art: http://www.darwin.edu.ar/Proyectos/FloraArgentina/Especies.asp verified on October 28,2025).
HANI, M.; LEBAZDA, R.; FENNI, M. 2017. Studies of morphological characteristics and production of seeds weeds of species of family Brassicaceae (Cruciferous) in Setifian High Plateau, Algeria. Annual Research & Review in Biology, 12(5), 1-9.
ITSA. 2016. Reglas Internacionales para el Análisis de las Semillas. The International Seed Testing Association (ITSA). Bassersdorf, Suiza. (Available at: https://vri.umayor.cl/images/ISTA_Rules_2016_Spanish.pdf verified on October 28,2025).
NIÑO-HERNANDEZ, J.C.; MORENO, D.F.; RUIZ-BERRÍO, H.D.; BALAGUERA-LÓPEZ, H.E; MAGNITSKIY, S. 2020. Luz, giberelinas y profundidad de siembra inciden sobre la germinación de semillas de Amaranthus hybridus L. Rev. U.D.C.A Act. & Div. Cient. 23(2):e1545. http://doi.org/10.31910/rudca.v23.n2.2020.1545
OSUNA FERNÁNDEZ, H.R.; OSUNA FERNÁNDEZ, A.M.; FIERRO ÁLVAREZ, A. 2017. Manual de propagación de plantas superiores. Universidad Autónoma Metropolitana, México. (Available at: https://www.casadelibrosabiertos.uam.mx/contenido/contenido/Libroelectronico/manual_plantas.pdf verified on October 28, 2025).
PURICELLI, E.C.; TUESCA, D.H. 2017. Análisis de los cambios en las comunidades de malezas en sistemas de siembra directa y sus factores determinantes. Revista de la Facultad de Agronomía, La Plata. 102(1), 97-118.
RADOSEVICH, S., HOLT, J.; GHERSA, C. 2007. Ecology of Weeds and Invasive Plants: Relationship to Agriculture and Natural Resource Management. Wiley, New York.
SENASA (SERVICIO NACIONAL DE SANIDAD Y CALIDAD AGROALIMENTARIA). 2025. Hirschfeldia incana. Sistema Nacional de Vigilancia y Monitoreo de Plagas, SENASA, Argentina. (Available at: https://www.sinavimo.gob.ar/plaga/hirschfeldia-incana verified on October 28, 2025).
TROIANI, H. 2016. Hirschfeldia incana (L.) Lagr.-Foss. Descripción de la especie. In: FERNÁNDEZ, O.A.; LEGUIZAMÓN, E.S.; ACCIARESI, H.A.; TROIANI, H.O.; VILLAMIL, C.B. (Eds.). Malezas e invasoras de la Argentina Tomo II. Descripción y reconocimiento. Editorial de la Universidad Nacional del Sur. 385 p.
USTARROZ, D. 2017. Hirschfeldia incana “nabo o mostacilla” resistente a glifosato y 2,4-D. INTA Manfredi. (Available at: https://www.aapresid.org.ar/blog/alerta-amarillo-hirschfeldia-incana-nabo-o-mostacilla-a-glifosato-y-24-d/ verified on October 28, 2025).
VIGNA, M.R.; MENDOZA, J. 2015, September. Presencia de poblaciones de Hirschfeldia incana (l.) resistentes metsulfuron-metil en el SO de Buenos Aires, Argentina. xxii Congreso de la Asociación Latinoamericana de Malezas (ALAM). i Congreso de la Asociación Argentina de Ciencia de las Malezas (ASACIM). Buenos Aires, Argentina.
Información adicional
redalyc-journal-id: 864
