Abstract: Lolium perenne ssp. multiflorum (Lam) Husnot, commonly known as ryegrass, is a globally important weed with documented resistance to multiple herbicide modes of action. The germination and emergence responses of both susceptible and herbicide-resistant ryegrass phenotypes to environmental conditions may differ between them. To develop sustainable management strategies that reduce herbicide use, the inclusion of allelopathic crops such as Carthamus tinctorius (safflower) is promising. In the semi-arid temperate region of Argentina, specifically in the southwest of Buenos Aires Province, safflower is regarded as an alternative winter crop that is being cultivated for edible oil production. Therefore, understanding the effect of different safflower varieties on the morphophysiological seed traits of herbicide-resistant ryegrass phenotypes is crucial for optimizing management strategies. In this context, this study aims to: 1) evaluate the germination capacity of three ryegrass populations (Lp)—a susceptible population (1LOLG1), a population resistant to both ALS and ACCase inhibitors (1LOLG2), and a population resistant to EPSPS, ALS, and ACCase inhibitors (1LOLG3)— under alternate temperature and water potential regimes in the laboratory, and 2) to assess the effect of allelopathic substances, remaining in the soil or in residual leaves, from five safflower genotypes (CW99 OL, RCJ3, WSRC01, Montola 2000, and RM NAPOSTÁ) on the germination and initial growth (coleoptile and radicle length) of Lp. Seed weight differed among Lp (1LOLG1<1LOLG2<1LOLG3; p<0.01), while cumulative germination followed the opposite pattern (1LOLG1>1LOLG2>1LOLG3; p<0.05). 1LOLG3 exhibited the highest hydrotime constant and the lowest minimum water potential for germination values (p<0.05). The safflower genotypes differed in their allelopathic potential; CW99 OL leaves reduced germination by 20% in all Lp. Conversely, Montola 2000 soil stimulated germination. Coleoptile length was mostly affected in the Lolium resistant populations, indicating significant allelopathic effects of Montola 2000, WSRC01, and RM NAPOSTÁ. These results suggest that safflower can negatively impact the establishment of herbicide-resistant ryegrass seedlings, that could be useful information to integrated management strategies.
Keywords: ryegrass, Carthamus tinctorius, early growth.
Resumen: Lolium perenne ssp. multiflorum (Lam) Husnot, comúnmente conocido como raigrás, es una maleza de importancia mundial con resistencia documentada a múltiples modos de acción de herbicidas. Las respuestas de germinación y emergencia de los fenotipos de raigrás susceptibles y resistentes a herbicidas pueden diferir entre sí. Para desarrollar estrategias de manejo sostenibles que reduzcan el uso de fitosanitarios sintéticos, la inclusión de cultivos alelopáticos como Carthamus tinctorius (cártamo) es prometedora. En la región templada semiárida de Argentina, concretamente en el sudoeste de la provincia de Buenos Aires, el cártamo se considera un cultivo alternativo de invierno que se cultiva para la producción de aceite comestible. Por lo tanto, comprender el efecto de las diferentes variedades de cártamo sobre atributos morfofisiológicos de las semillas de los fenotipos de raigrás resistentes a herbicidas es fundamental para optimizar las estrategias de manejo. En este contexto, el objetivo de este estudio es: 1) evaluar la capacidad de germinación de tres poblaciones de raigrás (Lp) —una población susceptible (1LOLG1), una población resistente a los inhibidores de ALS y ACCasa (1LOLG2) y una población resistente a los inhibidores de EPSPS, ALS y ACCasa (1LOLG3)— bajo temperaturas alternas y un gradiente de potenciales hídricos en laboratorio, y 2) evaluar el efecto de las sustancias alelopáticas, que permanecen en el suelo o en las hojas, de cinco genotipos de cártamo (CW99 OL, RCJ3, WSRC01, Montola 2000 y RM NAPOSTÁ) sobre la germinación y el crecimiento inicial (coleóptilo y longitud de radícula) de Lp. El peso de las semillas difirió entre Lp (1LOLG1<1LOLG2<1LOLG3; p < 0,01), mientras que la germinación acumulada siguió el patrón opuesto (1LOLG1>1LOLG2>1LOLG3; p < 0,05). 1LOLG3 exhibió la constante de hidro tiempo más alta y el menor potencial agua base medio (p < 0,05). Los genotipos de cártamo difirieron en su potencial alelopático; las hojas de CW99 OL redujeron la germinación en un 20% en todos los Lp. Por el contrario, el suelo de Montola 2000 estimuló la germinación. La longitud del coleóptilo fue afectada principalmente en las poblaciones resistentes de Lolium, lo que indica efectos alelopáticos significativos de Montola 2000, WSRC01 y RM NAPOSTÁ. Estos resultados sugieren que el cártamo puede tener un impacto negativo en el establecimiento de plántulas de raigrás resistentes a herbicidas, lo que podría ser de utilidad al implementar estrategias de manejo integrado de malezas.
Palabras clave: raigrás, Carthamus tinctorius, crecimiento inicial.
Evaluating herbicide-resistant Lolium perenne ssp. multiflorum germination and their response to safflower’s allelopathic effects
Instituto Nacional de Tecnología Agropecuaria

Esta obra está bajo una Licencia Creative Commons Atribución-NoComercial 4.0 Internacional.
Chemical weed control is highly effective, but the excessive use of synthetic herbicides can negatively impact ecosystems and human health. In Argentina, the annual herbicide inputs per cropland area are notably high (Hulme, 2023), leading to significant herbicide contamination in rivers, streams, and drinking water (Aparicio and De Gerónimo, 2024; Mayora et al., 2024). Additionally, frequent and heavy herbicide use exerts selection pressure on weeds, resulting in the emergence of herbicide-resistant populations, including resistance to more than one herbicide with the same or different mechanisms of action (Chipomho et al., 2023).
Buenos Aires Province has the highest number of documented cases of herbicide resistance in Argentina, accounting for 35% of them, mainly in winter weeds, especially Lolium spp. populations (Oreja et al., 2024). In fact, Lolium genotypes in this region have shown resistance to multiple herbicide sites of action, including 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), acetolactate synthase (ALS), and acetyl coenzyme A carboxylase (ACCase) inhibitors (Vigna et al., 2013).
Loliumperenne ssp. multiflorum (Lam) Husnot is an important weed in cropping systems worldwide, particularly affecting winter crops (Matzrafi et al., 2020). It spreads mainly through seed production, with seeds exhibiting a partial physiological dormancy that breaks after a summer post-maturation period (Lodovichi and Yanniccari, 2018). The timing and number of seedling emergence are crucial for weed management, as they affect competition with crops. Emergence is influenced by dormancy, germination, coleoptile elongation, and environmental factors such as temperature and water availability (Forcella et al., 2000). Studies such as Vila-Aiub et al. (2005) on L. rigidum show that germination and emergence responses to environmental conditions differ between herbicide-susceptible and herbicide-resistant phenotypes. Understanding the morphophysiological characteristics of problematic weeds is essential for developing sustainable management strategies that minimize herbicide use.
In Integrated Weed Management (IWM), reducing herbicide use and employing a mix of preventive and control methods can help minimize environmental impacts. In this context, allelopathy is a promising approach (Scavo and Mauromicale, 2020). It can be applied through intercropping, crop rotation, mulching, or cover crops that negatively affect seed germination and plant growth (Khamare et al., 2022; Sobrero and Acciaresi, 2014). The effectiveness of allelopathy depends on factors such as the chemical properties of the allelochemicals, the organs that produce them and their route of release, the characteristics and phenological stage of the donor plant, environmental conditions such as soil moisture and temperature, and the phenological stage of the recipient species (Sobrero and Acciaresi, 2014; Zhang et al., 2021). Therefore, understanding both the donor and target species is crucial, as allelochemical release and susceptibility can vary within and between species.
Safflower (Carthamus tinctorius L.) is an annual crop grown mainly for its high-quality edible and industrial oil. In southwestern Buenos Aires, its cultivation is under study due to its potential for growth in semi-arid conditions (Franchini et al., 2021). Laboratory studies have demonstrated that safflower residues inhibit radish seedling growth, including radicle and hypocotyl length, and seed germination (Motamedi et al., 2016). Motamedi et al. (2020) proposed that C. tinctorius could be incorporated as an allelopathic crop and a natural herbicide for weed management. However, the allelopathic effects of safflower genotypes have not yet been studied in Argentina. Therefore, this work aims to: 1) assess the germination capacity of multiple herbicide-resistant L. perenne ssp. multiflorum populations under different temperature and water potential conditions, and 2) evaluate the potential allelopathic effects of five safflower genotypes on the germination and initial growth of the above-mentioned populations.
Lolium seed collection
Three populations of L. perenne ssp. multiflorum from the southwestern area of Buenos Aires Province, Argentina, were studied: one herbicide-susceptible (1LOLG1; 37°58′80″ S, 61°21′60″ W), one resistant to ALS and ACCase inhibitors (1LOLG2; 38°43′20″ S, 61°18′00″ W), and one resistant to EPSPS, ALS, and ACCase inhibitors (1LOLG3; 38°43′20″ S, 61°18′00″ W). Seeds were collected in December 2022, air-dried at room temperature, and stored in paper bags under lab conditions (16–22°C, 20–35% RH) until use in July 2023.
In two simultaneous experiments, the germination capacity of Lolium populations (Lp) was assessed. Two months later, a third experiment investigated the allelopathic effects of safflower. All experiments were conducted in 2023 at the Weed Laboratory of the Agronomy Department, Universidad Nacional del Sur, Argentina.
To determine the 1000 seed weight (W1000), eight replicates of 100 randomly selected seeds from each Lp were weighed.
Germination experiment
For each trial and experimental treatment, four replicates of 30 seeds from each Lp were placed in 9 cm diameter Petri dishes sealed with parafilm. Each dish was considered an experimental unit (EU) and incubated in a corresponding chamber at a 12-hour alternating temperature cycle with a 12-hour photoperiod (150 µmol m-2 s-1) in a completely randomized design. Seeds were scored daily for germination, using radicle protrusion as the evaluation criterion. At the end of the germination tests, non-germinated seeds that were rotten or empty were considered dead. The viability of non-germinated seeds was assessed by the crush test (Borza et al., 2007), and all viable but non-germinated seeds were classified as dormant (Maity et al., 2021). All evaluated parameters were calculated on the basis of viable seeds.
Cumulative germination was fitted to the logistic model described in Equation 1.
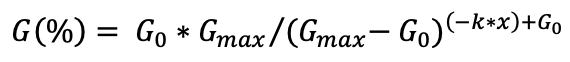
Equation 1
Where G indicates the cumulative germination over time x, Gmax is the maximum germination (%), G0 Is the initial germination, and k is the rate constant.
The germination rate (GR50) was also estimated, calculated as the inverse of the time required to reach 50% germination (1/t50; Bradford, 1990). Germination dynamics were evaluated using the Piper index (PI), defined as the number of days required for a seed to germinate (Petcu et al., 2017), and by comparison with the parameter k of equation 1.
Two simultaneous experiments were conducted in July 2023, following an after-ripening period. Experiment 1 evaluated the germinability of Lp under a thermal gradient, while Experiment 2 assessed it under a water potential gradient.
Experiment 1: In each UE, seeds were sown on two layers of Whatman No. 1 filter paper, moistened with 7 ml of distilled water, and placed in a germination chamber with a night/day temperature of 5/15, 10/20, 15/25, 20/30, or 25/35°C and a day/night cycle of 12/12 h for 15 days (N = 60).
The comparison of Lp was conducted using a thermal time model (García-Huidobro et al., 1982). Given the temperatures used in the experiment and the broad thermal window for Lolium seed germination, the analysis focused only on the suboptimal temperature range as defined by the following equation:
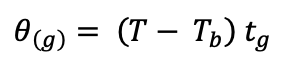
model, θ(g
In this model, θ(g) represents the suboptimal thermal time required for germination of a fraction g of the population; T is the incubation temperature; Tb is the base temperature below which germination does not occur; and tg is the time to germination in days for fraction g. The model assumes θ(g) follows a normal distribution with a mean of θ(50) and a standard deviation of σ(θ). Initially, Tb was determined and found to be consistent across Lp (p=0.59), so the average value was used for parameter calculations (Tb= 7.85°C). The average temperature value of each incubation regime was used.
Experiment 2: A water potential regime was simulated by applying 7 ml of polyethylene glycol solution (PEG 6000, Sigma-Aldrich), prepared according to Michel and Kaufmann, (1973), to Petri dishes lined with two layers of Whatman No. 1 filter paper. The treatments included osmotic potential (Ψ) of -0.2; -0.4; -0.8 y -1.2 MPa, using distilled water as control, i.e., Ψ=0 (N = 60: 5 Ψ * 3 Lp * 4 replicates). Seeds in PEG solutions were transferred to fresh solutions after the first 24 h and then weekly thereafter (Ni and Bradford, 1992). Petri dishes were kept in a germinator at 10/20°C for 15 days.
To analyze Lp seed germination responses to various Ψ values, a probit procedure described by Bradford, (1990) was employed. This method is based on three parameters: the hydrotime constant (θH) the base or minimum water potential for germination (Ψb) and the standard deviation of the base water potential within the seed population (σΨb). The base water potential for germination (Ψb, MPa) was estimated according to Equation 3:
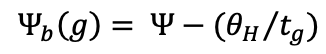
Equation 3
Where Ψ represents the water potential of the PEG solution, θH is the hydrotime constant, and tg is the time to radicle emergence for germination percentage g. To determine the optimal value of θH from all germination percentage data across all Ψs values, a single linear regression was performed (Bradford, 1990):
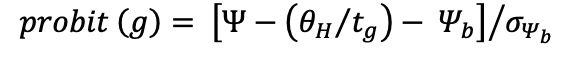
probit
The relationship between g and tg at any constant Ψ was analyzed using this probit transformation of g. For each constant Ψ Probit (g) was plotted against Ψ(θH/tg) using various values of θH until the best fit or minimal residual was achieved. The standard deviation in base water potential within the seed population (σΨb, MPa) is the inverse of the slope obtained from this plot.
Experiment 3: Allelopathy test
Five safflower plant genotypes (CW99 OL, RCJ3, WSRC01, Montola 2000 and RM NAPOSTÁ) were evaluated as donor plants. The accessions were grown at the Napostá Experimental Field, Buenos Aires, Argentina (38°25´30” S, 62°17’15” W) in April 2022. The soil was a Typic Haplustoll with a fine loamy texture, 3.98% organic matter, 0.18% total nitrogen and 15.2 ppm extractable phosphorus. Seeds were sown by hand at a depth of 3.5 cm. Plants were grown under rainfed conditions, and weeds and insects were controlled. A block design was followed (b=3), where each experimental unit measured 1 m * 2.8 m (N=15). Plant samples were harvested manually at maturity in January 2023. Soil samples were taken from a depth of up to 10 cm between plant rows in the center of each plot, while senescent steam leaves were collected randomly. All materials were air-dried at room temperature and stored in paper bags until use. For comparison, a control soil sample (sC) was taken from a natural field adjacent to the experimental area.
In October 2023, ten treatments were carried out in the laboratory to evaluate the potential effect of allelopathic substances remaining in the soil or in residual leaves from the crop according to Zhang et al. (2021). Each Petri dish was filled with 31.8 cm3 of sieved soil from plots corresponding to sCW99 OL, sRCJ3, sWSRC01, sMontola 2000, sRM NAPOSTÁ or sC; or with 0.55 g of leaf residues (Motamedi et al., 2016) from genotypes lCW99 OL, lRCJ3, lWSRC01, lMontola 2000 and lRM NAPOSTÁ on sC. Each dish was soaked with 12 ml of water and then incubated at 20/30°C for ten days.
The allelopathic effects on each Lp were studied in terms of germination, coleoptile length, radicle length, and seedling vigor index (VI) according to Mirmostafaee et al. (2020). Ryegrass sown in sC was used as the control. For the determination of radicle and coleoptile length, five 72 h-germinated seedlings were taken from each replicate. These seedlings were photographed and measured using Image J software.
Seedling VI was calculated according to germination percentage and seedling length:
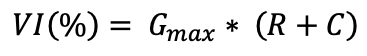
VI
where Gmax is the maximum germination achieved (%), R is radicle length (cm), and C is coleoptile length (cm). These traits were also reported relative to the control.
In parallel, the variables measured in the sC treatment were compared to assess possible morphological differences among Lp.
Statistical analysis
Logistic equations and k-comparisons using an extra-square F-test were performed using GraphPad Prism 8.0.1 software. The measured data and estimated parameters were analyzed and processed using Infostat® statistic software. In Experiments 1 and 2, each population was compared by one-way ANOVA, and means were separated using LSD multiple comparison test (α = 5%). Firstly, a two-way ANOVA was performed in Experiment 3 to determine the effect of Lp and safflower plant genotypes on germination and seedling parameters. Then, a one-way ANOVA was performed, partitioned by Lp. Gmax values were arcsine square root transformed to meet the assumptions of ANOVA.
Seed weight
The W1000 varied according to the population: 1LOLG1: 1,96a g < 1LOLG2: 2,18b g < 1LOLG3: 2,49c g (p<0.01).
Experiment 1: Effects of temperature on Lp germination
Since dormancy occurs under suboptimal environmental conditions (Bradford, 1990), germination among Lp was compared for each temperature condition, even though there was no interaction between the evaluated variables (p=0.36). The three Lp expressed the highest germination percentages at 20/30°C, with no difference in Gmax and GR50 among them (p>0.05, table 1; fig. 1). Under the remaining tested alternating temperatures, Gmax was highest for 1LOLG1 and lowest for 1LOLG3, with 1LOLG2 showing intermediate values (p<0.05, table 1; fig. 1). A similar trend was observed for GR50 with no differences in PI (p<0.05 and p>0.05 for GR50 and PI respectively). The germination dynamics of the three Lp tended to accelerate with increasing temperature (higher GR50). The mean time for a seed to germinate was 3.33 days at 25/35°C and 20/30°C, 4.6 days at 15/25°C and 10/20°C, and 7.8 days at 5/15°C (p<0.01). Only at 5/15°C, 1LOLG1 presented a steeper slope (p=0.03).
Results indicate a common mean baseline temperature of 5.4⁰C among populations (p>0.05, table 2). θ(50) values obtained from all the evaluated temperature regimes increased with the resistance level, with 1LOLG3 being significantly higher than the other two populations (p<0.01; table 2).
Experiment 2: Effects of osmotic potentials on Lp germination
All Lp exhibited similar germination behavior when subjected to osmotic potentials ranging from 0 to -0.4 MPa, but germination decreased to 50% and 10% at -0.8 and -1.2 MPa, respectively (fig. 2; p<0.01). Between 0 to -0.4 MPa, 1LOLG3 showed the lowest Gmax and GR50 values, while 1LOLG1 presented the highest Gmax and k (table 3, fig. 2a-c; p<0.05). The mean germination time was 4.5 days between 0 and -0.4 MPa, 6.2 days at -0.8 MPa, and 7.6 days at -1.2 MPa (p<0.01) for all Lp.
The θ(H) value remained constant across Lp (p= 0.67), but 1LOLG3 exhibited a lower Ψb (p=0.03) and higher σΨb values (p=0.04; table 4).
Experiment 3: Allelopathy test
In Exp. 3. no changes in the PI of ryegrass germination were observed (p>0.05) and the cumulative germination of Lp followed the same pattern as observed in Experiment 1 and 2 (1LOLG1>1LOLG2>1LOLG3; p=0.01). Although the interaction between safflower plant accessions and Lp was not significant (p=0.18), the LSD test showed that safflower genotypes affected germination differently among Lp. In 1LOLG1, senescent steam leaves from CW99 OL, Montola 2000 and RM NAPOSTÁ reduced germination by up to 15% and increased GR50 values in five days, compared to the control (p<0.05; fig. 3A). Germination of 1LOLG2 was inhibited by 20% by lCW99 OL treatment, but stimulated by sMontola 2000 and sRM NAPOSTÁ, reaching 100% (p<0.01; fig. 3B). Finally, in 1LOLG3, lCW99 OL reduced germination by 20% and delayed GR50 (p<0.05), while sMontola 2000 stimulated both parameters (p<0.05; fig. 4C).
There were no significant differences in the length of the Lp coleoptile or radicle. Safflower varieties had no effect on coleoptile length in 1LOLG1, but Montola 2000, WSRC01 and RM NAPOSTÁ reduced coleoptile length by 50-60% in 1LOLG2 and 1LOLG3 (fig. 4A). Effects on radicle length were variable: lMontola 2000 and lWSRC01 treatments reduced radicle length by 50% in 1LOLG1. In 1LOLG2, lCW99 OL increased radicle length by 25%, while lRM NAPOSTÁ decreased it by 15%. In 1LOLG3, lRCJ3 and sCW99 OL stimulated radicle length by about 30% (p<0.05; fig. 4B).
Finally, vigor index was reduced in 1LOLG1 by lMontola 2000 and lWSRC01 treatments. In 1LOLG2, VI was reduced by sCW99 OL, sRCJ3, lMontola 2000, s-lWSRC01 and s-lRM NAPOSTÁ treatments. In 1LOLG3, VI was reduced by the lCW99 OL, lMontola 2000 and lRM NAPOSTÁ and by both WSRC01 treatments (p<0.05; fig. 4C).
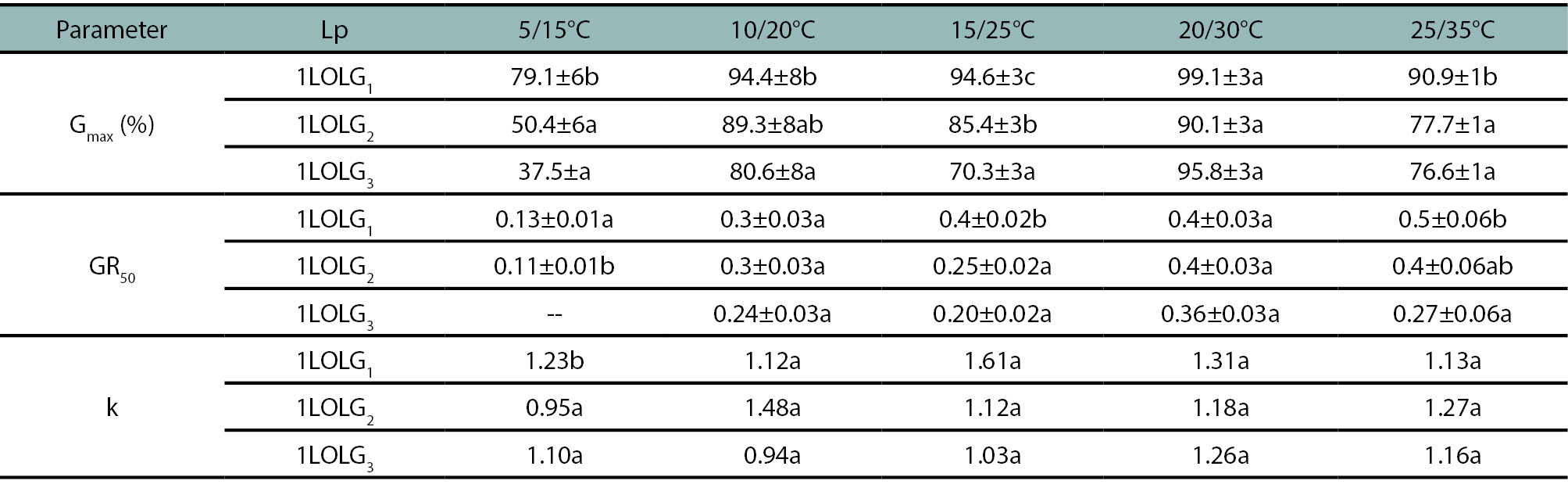
Table 1.
The estimated of the mean and standard errors for germination parameters of Lolium perenne ssp. multiflorum populations. 1LOLG1: an herbicide-susceptible, 1LOLG2: acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors-resistant and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) at a range of alternate temperatures (5/15, 10/20, 15/25, 20/30 and 25/35°C). Gmax represents the maximum germination achieved, the GR50 indicates the germination rate, calculated as the inverse of the time required to reach 50% germination and k is the logistic model rate constant. On each row, Gmax and GR50 are compared between Lp by LSD multiple range test and k by extra sum-of-squarer F test, identical letters are not significantly different at p>0.05.
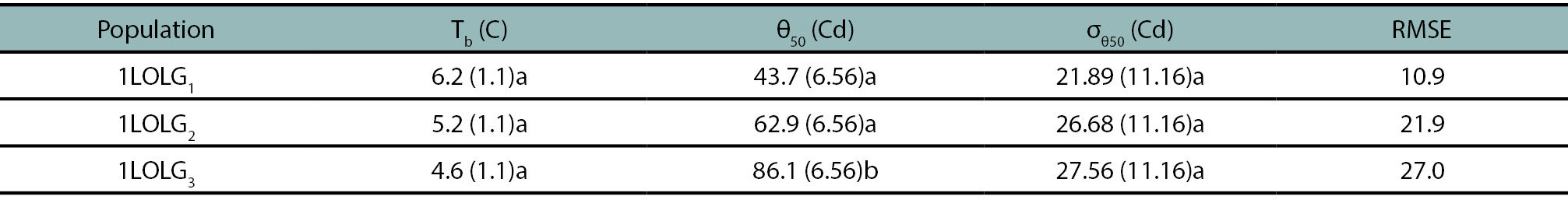
Table 2.
Estimated parameters of the suboptimal thermal time model (standard errors in brackets) describing seed germination of Lolium perenne ssp. multiflorum population. 1LOLG1: an herbicide-susceptible, 1LOLG2: acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors-resistant and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) at a range of alternate temperatures (5/15, 10/20, 15/25, 20/30 and 25/35°C). θ(50) and σ(θ50) are the mean and standard deviation, respectively, of the normal distribution of the suboptimal thermal time. Tb is the base temperature and RMSE is the root mean square error (goodness of fit). On each column, identical letters are not significantly different at p>0.05 by LSD multiple range test.
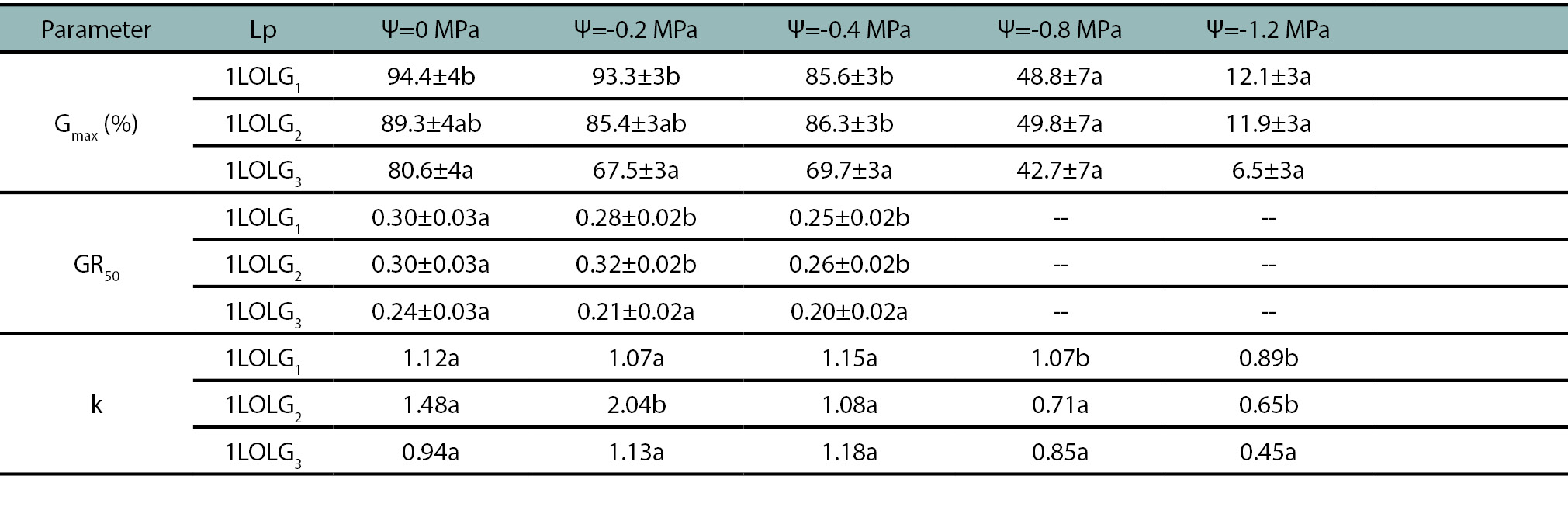
Table 3.
The estimated mean and standard error for germination parameters of Lolium perenne ssp. multiflorum populations. 1LOLG1: an herbicide-susceptible, 1LOLG2: acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors-resistant and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) under an osmotic potential gradient (Ψ= 0; -0.2; -0.4; -0.8 and -1.2 MPa). On each row, Gmax represents the maximum germination achieved, the GR50 indicates the germination rate, calculated as the inverse of the time required to reach 50% germination and k is the logistic model rate constant. Gmax and GR50 are compared between Lp by LSD multiple range test and k by extra sum-of-squarer F test, identical letters are not significantly different at p>0.05.
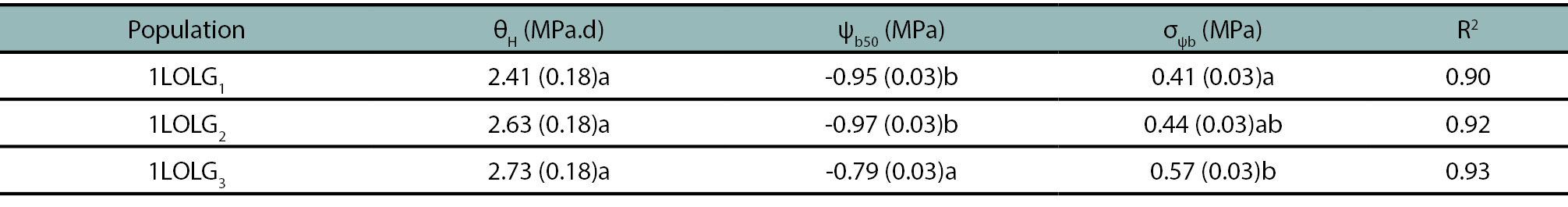
Table 4.
Estimated hydrotime model parameters (standard errors in brackets) describing seed germination of Lolium perenne ssp. multiflorum populations: A herbicide-susceptible (1LOLG1), an acetolactate synthase (ALS) and acetyl coenzyme-A carboxylase (ACCAsa) inhibitor-resistant (1LOLG2), and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) at a range of water potentials (0, -0.2, -0.4, -0.8 and -1.2 MPa). θ(H) is the hydrotime constant; ψb50 is the median base water potential; σψb is the standard deviation in base water potential and R2 is the coefficient of determination of the regression of probit germination against base water potential. On each column, identical letters are not significantly different at p>0.05 by LSD multiple range test.
The heaviest 1LOLG3 seed result is consistent with previous studies that have shown a positive correlation between L. perenne seed weight and survival to multiple herbicides and individually to pinoxaden (ACCAsa-inhibitor) or glyphosate (EPSPS-inhibitor; Maity et al., 2021). However, more research is needed to determine whether seed weight is directly related to herbicide resistance or other factor as environmental conditions during fruit set or resource availability (Lacey et al., 1997; Wulff et al., 1986).
This work shows that the optimal fluctuating temperature for germination of all Lp was 20/30°C (fig. 1D), similar to that observed by Javaid et al. (2022), which indicated that 25°C yields the highest germination percentages in L. perenne. Variations in germination among Lp were observed under suboptimal temperatures (fig. 1A-C), in agreement with Vila-Aiub et al. (2005) for L. rigidum, where ACCase-resistant populations exhibit deeper environmental dormancy (fig. 1-2). However, unlike Vila-Aiub et al. (2005) who attributed these differences to base temperature Tb, our results indicate that such differences are due to thermal time (θ50), which increased with herbicide resistance (p<0.01; table 2). The increment in thermal time indicates that more time is needed to complete germination under the same temperature conditions, explaining the higher GR50 observed in 1LOLG3 (Bradford, 2002; fig. 1). These results could lead to different seedling emergence patterns compared to herbicide-sensitive individuals (Vila-Aiub et al., 2005). Phenotypes that require more thermal time to germinate, resulting in delayed seedling establishment, may be ecologically disadvantageous due to the importance of rapid seedling growth to capture light and avoid shading (Vila-Aiub et al., 2005).
Similar to exp. 1, 1LOLG3 showed lower GR50 and Gmax values under optimal or moderate water stress conditions, probably related to the higher Ψ(b(50) (p<0.05, tables 3-4), which could be considered as an index of the mean dormancy status of the seed population (Chantre et al., 2010). Lower germination values at osmotic potentials below -0.4 MPa aligns with Javaid et al. (2022). Obtained high Ψ(b(50) values are associated with the tests being conducted during the peak emergence of Lolium in the field, as the seeds had already undergone a period of after-ripening being non-dormant (table 4). The dormancy release process exhibited a gradual decrease in Ψ(b(50) values with increasing after-ripening time (Chantre et al., 2010).
The lack of differences in Lp seedling dimensions (fig. 4), coupled with the fact that 1LOLG3 presented the heaviest seeds but the lowest GR50 (fig. 1,2; as discussed below), suggests that the variation in germination among Lp may be influenced by the thickness of the seed coat, which could hinder water absorption, contain germination inhibitors, or include polyphenols that induce hypoxia in the embryo (Brown, 1931). However, more studies are necessary to clarify such hypotesis. Given the critical importance of emergence timing for weed management, and the documented positive correlation between seed traits (such as seed weight and dormancy) and herbicide resistance, it is essential to investigate potential associations between seed morpho-physiological characteristics and herbicide resistance (Maity et al., 2021).
As observed by Motamedi et al. (2020) in radish seeds, different safflower genotypes exhibit varied allelopathic potential, resulting in different effects on Lp. Germination was only reduced by leaf debris treatments, with the 1LOLG1 population showing the greatest sensitivity to various safflower varieties. The lCW99 OL treatment reduced germination similarly in all Lp, suggesting a common site of action. This is consistent with the evidence that L. rigidum germination is reduced by leaf extracts from different mature safflower cultivars (Nikneshan et al., 2011). Although a 20% reduction in germination may contribute to ryegrass management in the field, the inclusion of safflower under zero-tillage, intended to leave leaf stubble on the soil surface due to its apparent allelochemical properties, should be complemented with additional weed control strategies. Further bioassays with extracts are needed to investigate this effect, as new compounds with allelopathic properties are needed for the development of natural herbicides. Conversely, sMontola 2000 treatment stimulated the germination of 1LOLG2-3, but the fact that the control 1LOLG1 germinated close to 100% did not allow us to corroborate whether germination of this population was stimulated by it (fig. 3). Tests with dormant seeds could elucidate this.
Several studies have found that safflower root exudates stimulate the germination of parasitic weed species, acting as a signal to indicate the presence of their host (Rial et al., 2020). This has led to the concept of ‘suicidal germination’, where a compound that induces parasitic seed germination is applied to soil. If the parasitic plant germinates without its host, it cannot complete its life cycle and dies, reducing the seed bank (Rial et al., 2020). However, no such effect has been reported in the literature for Lolium. Based on the results obtained here, it may be possible with further research to develop a formulation that stimulates Lolium germination for application in the field to synchronize emergence and reduce subsequent control efforts. If most seeds germinated in the first autumn cohort, the soil seed bank would be significantly reduced, leaving fewer seeds for future emergence. In an ideal scenario, control could be concentrated on a single date, with even pre-sowing mechanical tools used to manage resistant populations.
Coleoptile length was the most affected seedling parameter in 1LOLG2 and 1LOLG3, consistent with the findings of Nikneshan et al. (2011) in L. rigidum. The lack of difference between leaf and soil treatments suggests that the effect may be due to a compound in the leaves that leaches into the soil, although further studies are needed to confirm this. Trials in which Lolium seedlings were watered with leaf extracts could assess potential growth reduction after post-emergence application. The small increase in radicle length observed in some cases (fig. 4) could be due to a hormesis effect.
The allelopathic potential of Montola 2000, WSRC01, and RM NAPOSTÁ cultivars on seedling growth was demonstrated. While the reduction in germination was not strong, the negative effect of safflower on ryegrass establishment, especially in 1LOLG3 (fig. 4C), is evident when both are considered in the VI index. This index takes into account reductions in both germination and seedling growth, which directly affect emergence timing and seedling competitiveness (Forcella et al., 2000). In the case of rainfall, late emergence would hinder competition with neighboring seedlings due to delayed germination and/or shorter coleoptile emergence, and especially if the radicle is smaller and less able to absorb nutrients. This allows a vigorous crop to outcompete the weeds.
As the germination behavior and emergence patterns of different populations of the same species are influenced by a wide range of internal and environmental factors, generalizations and predictions are difficult. However, it is important to understand the similarities and differences in early morpho-physiological characteristics of problematic populations within a region, particularly in the context of herbicide resistance. The results obtained should be validated under field conditions, so that the knowledge generated can contribute to optimizing management strategies and reducing dependence on agrochemicals.
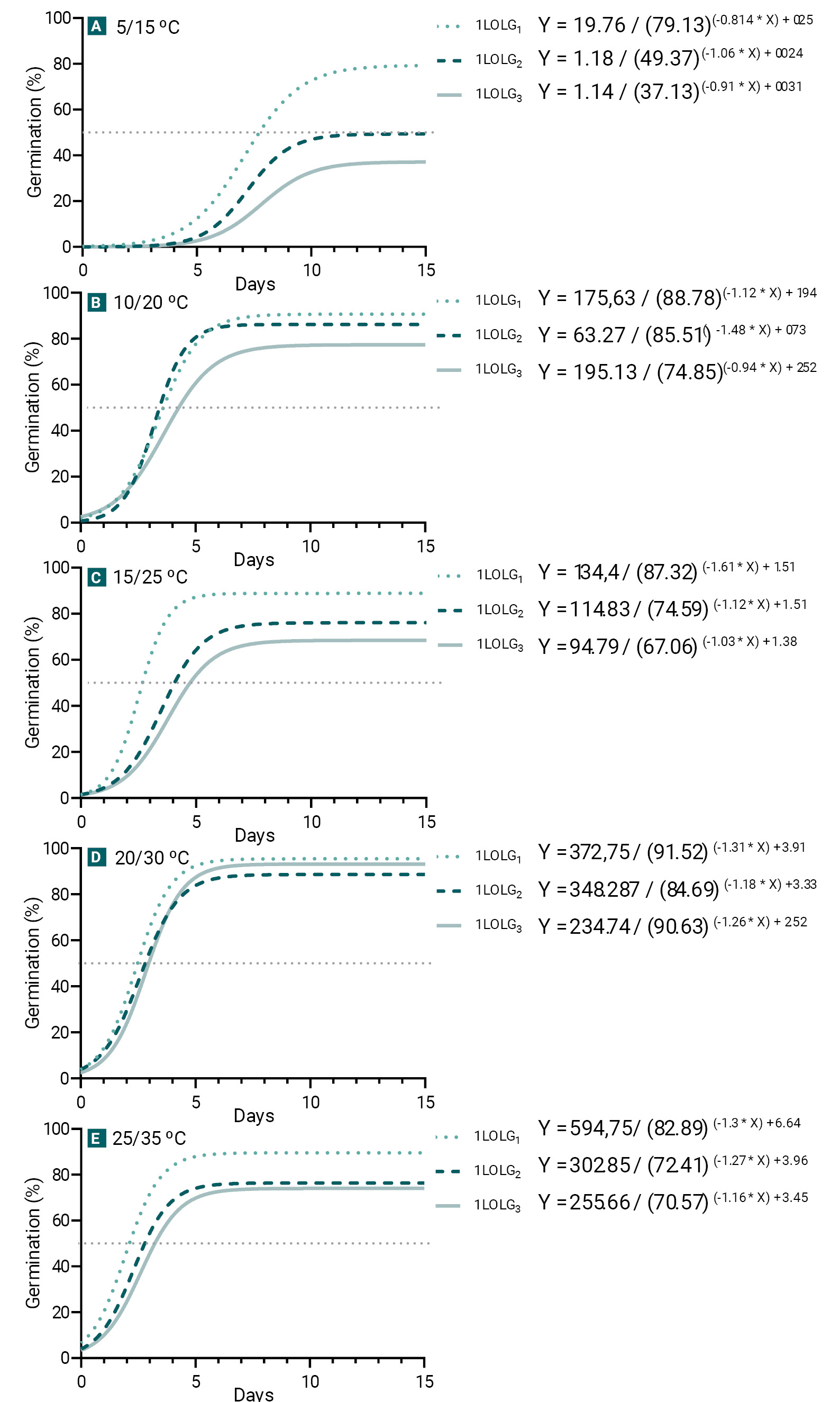
Figure 1.
Germination of Lolium perenne ssp. multiflorum populations under different alternating temperature regimes (Experiment 1). Susceptible to herbicides (1LOLG1; dotted line), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2; dashed line) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3; continuous line).
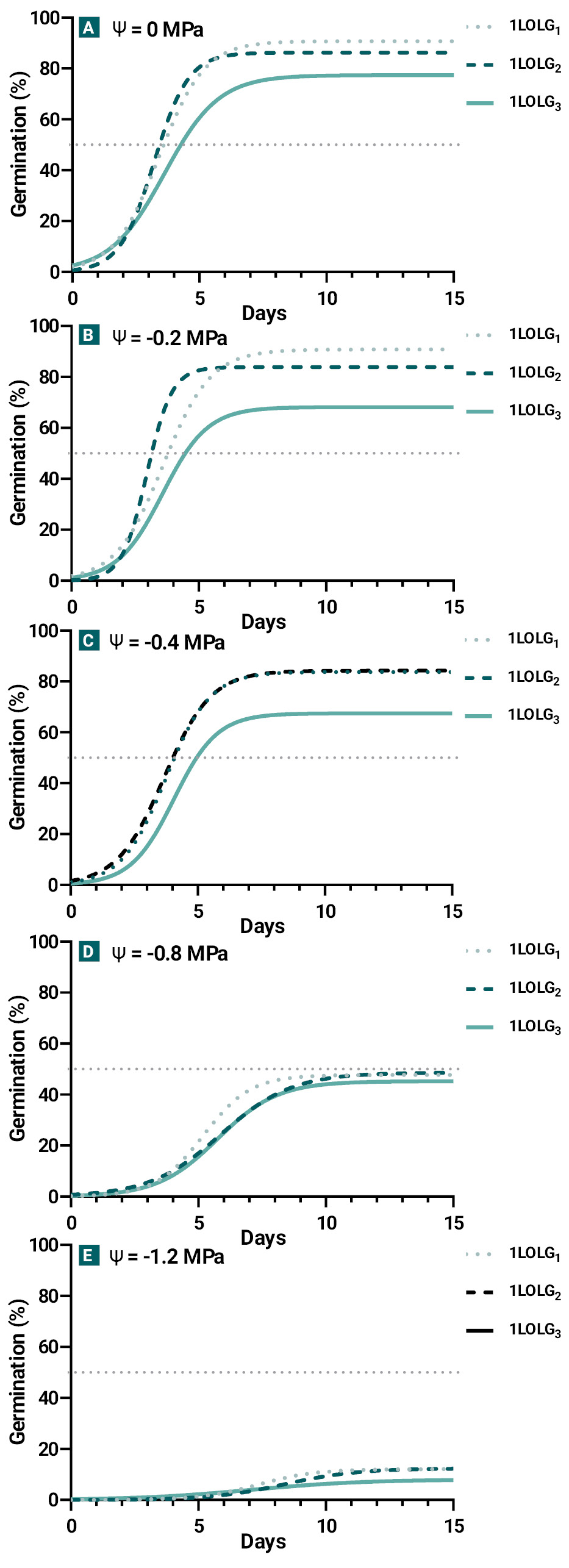
Figure 2.
Germination of Lolium perenne ssp. multiflorum populations under an osmotic potential gradient (Experiment 2): susceptible to herbicides (1LOLG1; dotted line), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2; dashed line) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3; continuos line).
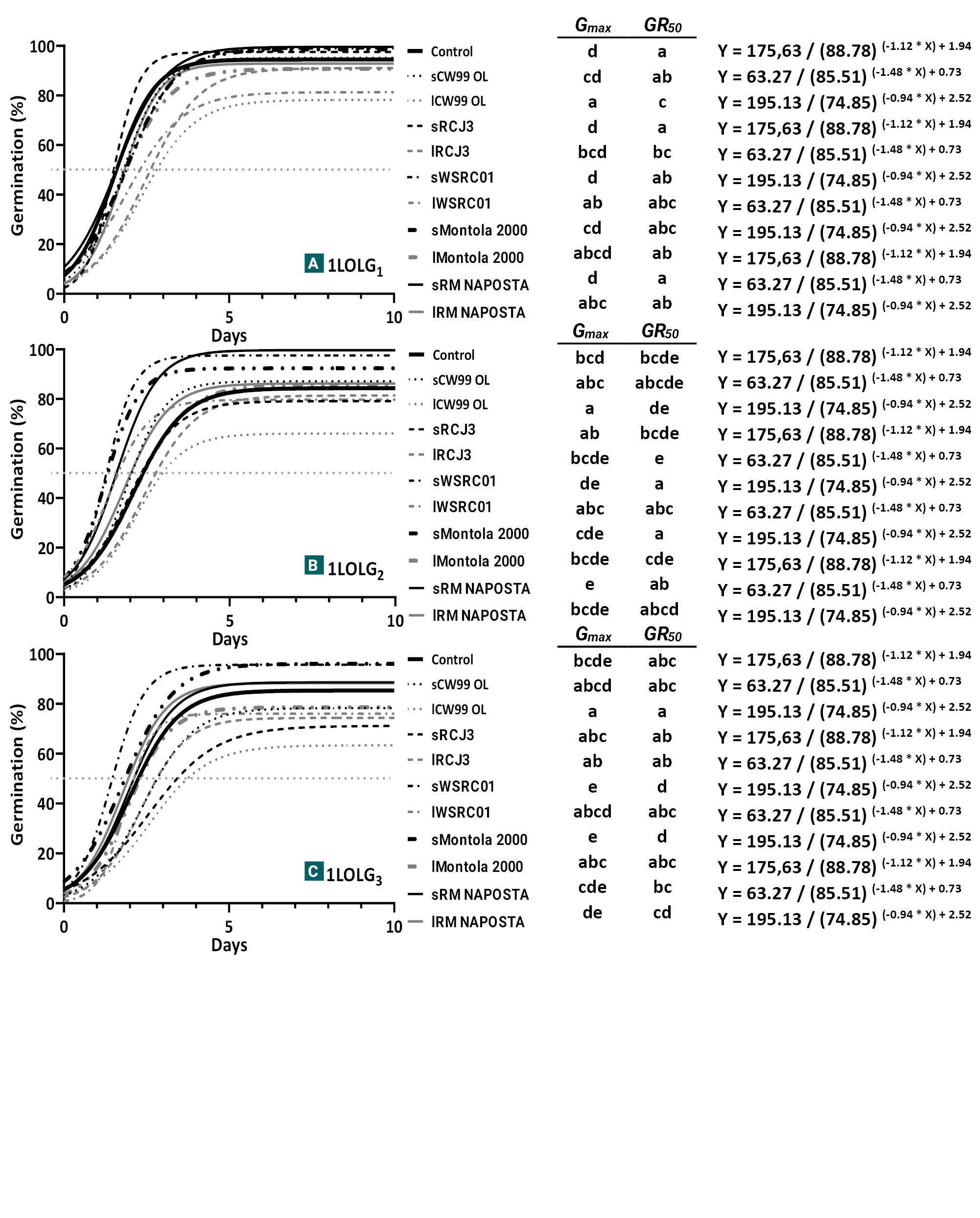
Figure 3.
Germination of Lolium perenne ssp. Multiflorum populations under safflower plant accessions treatments: susceptible to herbicides (1LOLG1), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3). Identical letters are not significantly different at p>0.05 by LSD multiple range test.
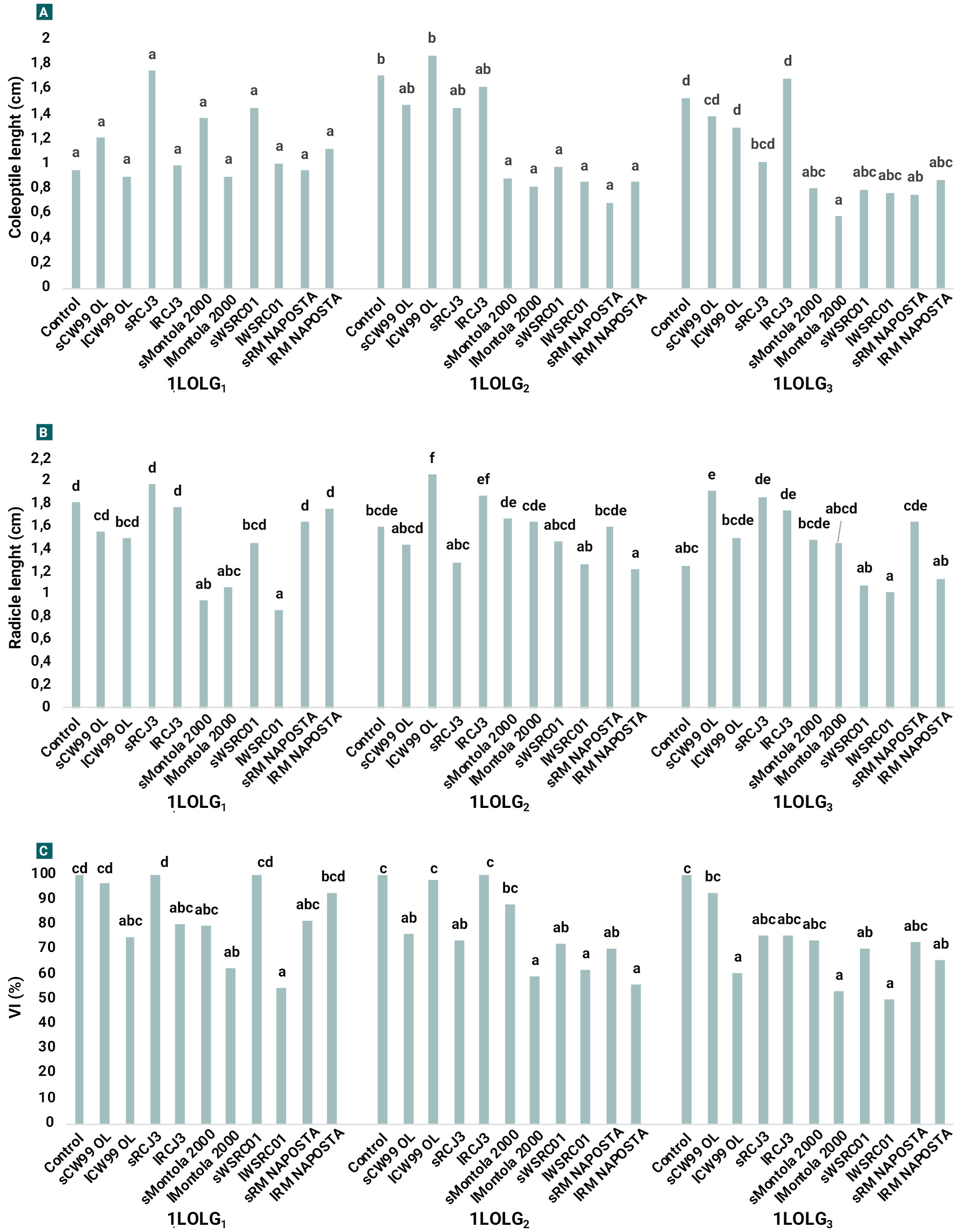
Figure 4.
Characteristics of Lolium perenne ssp. multiflorum populations under treatments with safflower plant accessions: susceptible to herbicides (1LOLG1), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3). A. coleoptile length, B. radicle length and C. vigor index. Comparisons were among treatments within each Lp, identical letters are not significantly different at p>0.05 by LSD multiple range test.
The observed differences in seed dormancy associated with herbicide resistance play a key role in determining the timing of resistant seedling emergency. This has important implications for the development of targeted management strategies aimed at limiting the spread of resistant populations. The safflower genotypes differed in their allelopathic potential; CW99 OL leaves reduced germination by 20% in all Lp. Conversely, Montola 2000 soil stimulated germination. Coleoptile length was mostly affected in the Lolium resistant populations, indicating significant allelopathic effects of Montola 2000, WSRC01, and RM NAPOSTÁ. These results suggest that safflower can negatively impact the establishment of herbicide-resistant ryegrass seedlings, that could be useful information to integrated management strategies.
The results presented here suggest new research avenues for the identification, isolation, and evaluation of allelochemicals through dose-response bioassays. This research trajectory could result in the development of eco-friendly bioherbicides with novel modes of action or compounds that stimulate seed germination. However, the efficacy of these substances on seeds with higher dormancy levels remains uncertain. Furthermore, the role of these substances as dormancy breakers or terminators, their residual activity in the soil, and the potential environmental side effects associated with high application rates require further investigation.
redalyc-journal-id: 864
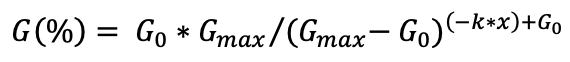
Equation 1
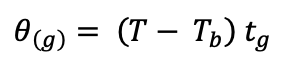
model, θ(g
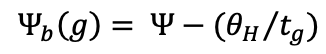
Equation 3
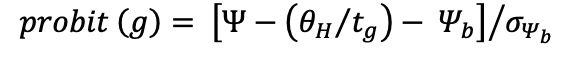
probit
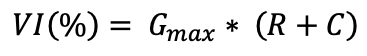
VI

Table 1.
The estimated of the mean and standard errors for germination parameters of Lolium perenne ssp. multiflorum populations. 1LOLG1: an herbicide-susceptible, 1LOLG2: acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors-resistant and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) at a range of alternate temperatures (5/15, 10/20, 15/25, 20/30 and 25/35°C). Gmax represents the maximum germination achieved, the GR50 indicates the germination rate, calculated as the inverse of the time required to reach 50% germination and k is the logistic model rate constant. On each row, Gmax and GR50 are compared between Lp by LSD multiple range test and k by extra sum-of-squarer F test, identical letters are not significantly different at p>0.05.

Table 2.
Estimated parameters of the suboptimal thermal time model (standard errors in brackets) describing seed germination of Lolium perenne ssp. multiflorum population. 1LOLG1: an herbicide-susceptible, 1LOLG2: acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors-resistant and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) at a range of alternate temperatures (5/15, 10/20, 15/25, 20/30 and 25/35°C). θ(50) and σ(θ50) are the mean and standard deviation, respectively, of the normal distribution of the suboptimal thermal time. Tb is the base temperature and RMSE is the root mean square error (goodness of fit). On each column, identical letters are not significantly different at p>0.05 by LSD multiple range test.

Table 3.
The estimated mean and standard error for germination parameters of Lolium perenne ssp. multiflorum populations. 1LOLG1: an herbicide-susceptible, 1LOLG2: acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors-resistant and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) under an osmotic potential gradient (Ψ= 0; -0.2; -0.4; -0.8 and -1.2 MPa). On each row, Gmax represents the maximum germination achieved, the GR50 indicates the germination rate, calculated as the inverse of the time required to reach 50% germination and k is the logistic model rate constant. Gmax and GR50 are compared between Lp by LSD multiple range test and k by extra sum-of-squarer F test, identical letters are not significantly different at p>0.05.

Table 4.
Estimated hydrotime model parameters (standard errors in brackets) describing seed germination of Lolium perenne ssp. multiflorum populations: A herbicide-susceptible (1LOLG1), an acetolactate synthase (ALS) and acetyl coenzyme-A carboxylase (ACCAsa) inhibitor-resistant (1LOLG2), and a 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; glyphosate), ALS- and ACCAsa inhibitors-resistant (1LOLG3) at a range of water potentials (0, -0.2, -0.4, -0.8 and -1.2 MPa). θ(H) is the hydrotime constant; ψb50 is the median base water potential; σψb is the standard deviation in base water potential and R2 is the coefficient of determination of the regression of probit germination against base water potential. On each column, identical letters are not significantly different at p>0.05 by LSD multiple range test.

Figure 1.
Germination of Lolium perenne ssp. multiflorum populations under different alternating temperature regimes (Experiment 1). Susceptible to herbicides (1LOLG1; dotted line), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2; dashed line) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3; continuous line).

Figure 2.
Germination of Lolium perenne ssp. multiflorum populations under an osmotic potential gradient (Experiment 2): susceptible to herbicides (1LOLG1; dotted line), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2; dashed line) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3; continuos line).

Figure 3.
Germination of Lolium perenne ssp. Multiflorum populations under safflower plant accessions treatments: susceptible to herbicides (1LOLG1), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3). Identical letters are not significantly different at p>0.05 by LSD multiple range test.

Figure 4.
Characteristics of Lolium perenne ssp. multiflorum populations under treatments with safflower plant accessions: susceptible to herbicides (1LOLG1), resistant to acetolactate synthase (ALS) and acetyl coenzyme A carboxylase (ACCAsa) inhibitors (1LOLG2) and resistant to a 5-enolpyruvylsikimate-3-phosphate synthase (EPSPS) inhibitor (glyphosate), ALS and ACCAsa (1LOLG3). A. coleoptile length, B. radicle length and C. vigor index. Comparisons were among treatments within each Lp, identical letters are not significantly different at p>0.05 by LSD multiple range test.
