Abstract: The lygaeoid Orsillus depressus Dallas feeds on seeds of several species, mainly cypress. Since the end of the last century, it has extended its range from its native area in the Mediterranean Basin towards northeastern Europe and it is considered an invasive species. In the present work, the presence of O. depressus is reported for the first time from the New World and Southern Hemisphere. Host plants and distribution from central-southern Argentina are given. In addition, morphological characters that allow their identification are provided.
Keywords: Argentina, Cupressaceae, Invasive species, True bug.
Resumen: El ligeido Orsillus depressus Dallas se alimenta de semillas de varias especies, principalmente cupresáceas. Desde finales del siglo pasado ha extendido su área de distribución desde su área de origen en la cuenca mediterránea hacia el noreste de Europa, y se la considera una especie invasora. En el presente trabajo se reporta por primera vez la presencia de O. depressus en el Nuevo Mundo y en el Hemisferio Sur. Se brindan datos de sus plantas hospedadoras y su distribución desde el centro-sur de Argentina. Además, se aportan caracteres morfológicos que permiten su identificación.
Palabras clave: Argentina, Chinches, Cupressaceae, Especies invasoras.
Notas
First record of Orsillus depressus (Hemiptera: Lygaeidae: Orsillinae) from the New World and Southern Hemisphere
Primer registro de Orsillus depressus (Hemiptera: Lygaeidae: Orsillinae) de América y del Hemisferio Sur
Sociedad Entomológica Argentina
Recepción: 11 Junio 2022
Aprobación: 20 Septiembre 2022
The genus Orsillus Dallas (Hemiptera: Lygaeidae: Orsillinae) includes six species of medium to small-sized true bugs distributed in the Palearctic Region (Péricart, 1998, 2001; Dellapé & Henry, 2022). The nymphs and adults live in the cones of different species of conifers and use their stylets to feed on the seeds, causing damage to seed orchards, natural stands, and plantations (Roques et al., 1999; Battisti et al., 2000). In addition, when adults migrate in search of food resources or egg laying sites, they can carry spores of the fungus Seiridium cardinale Sutton & Gibson and S. unicorne (Cooke & Ellis) B. Sutton that causes cypress bark canker (Roques & Battisti, 1999; Ramos & Abrantes, 2000).
Orsillus depressus Dallas is the most widely distributed species in the genus (Péricart, 2001; Costas et al., 2018; Dellapé & Henry, 2022). Since the end of the last century, it has extended its range from its native area in the Mediterranean Basin towards northeastern Europe (Aukema, 1988; Hawkins, 1989; Korcz, 2007; Hebda et al., 2016; Rabitsch, 2008; Lis & Stolarczyk, 2018), and its presence in the Italian Alps is an example of its ability to adapt to harsher climates (Dioli, 1991). Orsillus depressus is considered a pest of conifers and an invasive species in Europe (Sweet, 2000; Wittenberg, 2005; CABI, 2022), and is on the list of quarantine pests in the USA (USDA, 2017).
Orsillus depressus appears to be less host-specific than other Orsillus species, being capable of feeding and ovipositing on species of Chamaecyparis Spach,Cupressus L., Juniperus L., Sequoiadendron giganteum (Lindley) J. Buchholz, Thuja L. (Cupressaceae), and Pinus L. (Pinaceae), including species introduced into its native area of the Mediterranean Basin (Péricart, 1998; Sweet, 2000; Rouault et al., 2005; Rabitsch, 2008). In Argentina, host conifers of O. depressus were introduced from the Northern Hemisphere for ornamental purposes (Parodi & Dimitri, 1972), being frequent in most parks, gardens and recreational areas. Some of these exotic species, such as Cupressus arizonica Greene and C. macrocarpa Hartw. ex Gord., are considered invasive in Córdoba Province, whereas others, such as Juniperus communis L., develop adventitiously in the Andean forests, (Giorgis & Tecco, 2014; Cantero et al., 2016) where there are also native Cupressaceae forests comprising Austrocedrus chilensis (D. Don) Pic. Serm. & Bizzarri, Pilgerodendron uviferum (D. Don) Florin, and Fitzroya cupressoides (Molina) I.M. Johnst.
Females of O. depressus lay their eggs in two or three-year-old cones, when they begin to open to disperse their seeds, or in the emergence holes of Megastigmus wachtli Seitner (Megastigmidae), a wasp whose larvae develop inside the cones (Roques & Battisti, 1999; Rouault et al., 2005). Another hymenopteran associated with O. depressus is the egg parasitoid Telenomus gr. floridanus (Scelionidae) (Rouault et al., 2000).
A female collected by the senior author on May 8, 2020, in Villa Regina town, Río Negro, Argentina, and identified as O. depressus triggered the search for more specimens. Between November 2021 and March 2022, samples were taken from trees in the family Cupressaceae to verify the presence of eggs, nymphs, and adults.
Surveys were carried out at 25 sites located in urban and suburban areas within a distance of 302 km in a north-south direction and 652 km in an east-west direction, covering 15 localities belonging to Buenos Aires, La Pampa, Neuquén, and Río Negro provinces. These localities are in most cases separated by extensive areas of grasslands and xerophytic shrubby plains.
Host trees belonging to the genera Chamaecyparis, Cupressus, and Thuja were identified at each site and 30 cones were collected, selecting those with open scales and still containing their seeds. On some occasions, samples were collected on different dates and hosts for the same site. In the laboratory, the samples from each site/date/host were placed in 1-liter plastic containers with cloth lids and observed with a Zeiss Stemi 2000-C 6.5x-50x stereomicroscope. Additionally, the cones were observed externally for holes that indicate the presence of species that provide oviposition sites, such as M. wachtli. Those cones that contained masses of unhatched eggs were kept in the laboratory at 25 ºC for 30 days under 16L:8D conditions to observe the emergence of oophagous parasitoids. The collection sites were georeferenced by GPS device and the data obtained were used to make a distribution map using the QGIS software version 3.8.1. The presence of O. depressus was communicated to the Argentine phytosanitary authorities through identification Nº 580 of Sistema Nacional de Vigilancia y Monitoreo (SINAVIMO) and the examined specimens were deposited in the Museo de La Plata (MLP) La Plata, Buenos Aires, Argentina and in Laboratorio Regional de Plagas of Servicio Nacional de Sanidad Agroalimentaria (SENASA) in Villa Regina, Río Negro, Argentina.
Following Péricart’s key to adults in the genus (1998), specimens were identified as O. depressus (Fig. 1). The first couplet in the key refers to “Rostrum not exceeding or slightly exceeding the middle of the abdomen; sternites III and IV with a more or less distinct longitudinal median groove.” to O. depressus or O. reyi Puton or “Rostrum reaching or nearly apex of abdomen; sternites III and IV without a longitudinal median groove; second antennal segment of the order of 1.5 times as long as fourth'' to O. maculatus (Fieber) and O. pinicanariensis Lindberg.

Fig. 1.
Orsillus depressus female.
Our specimens show variability on rostral length, ranging from attaining the base of sternite VI to almost attaining the pygophore in some males. Rouault et al. (2005) noted that in areas where C. sempervirens occurs, O. depressus and O. maculatus may occur on the same tree, but morphological variability of adults and nymphs often hinders accurate identification of the species. They measured the body length, rostral length, as well as their ratio (R:Bd) from several hundred specimens on different host plants, and found that the ratio was significantly lower in O. depressus (mean R:Bd < 0.85, although the values ranged from 0.62 to 0.89) than in O. maculatus (mean R:Bd > 0.85 in females and most males). Therefore, they argued that based on frequency distribution, this ratio does not seem to necessarily result in an accurate identification, particularly of males, and it is related to some degree with the host and the geographical location. According to these authors, the R:Bd ratio on fourth- and fifth-instar nymphs also could represent a diagnostic character for specific identification.
Besides the variability exhibited in rostral length and minor variation on coloration pattern, we believe that all specimens belong to the same specific unit because of their consistent morphology and vestiture. Most of the specimens lack a well-demarcated dark medial line on the pronotum, often used as diagnostic to identify O. depressus, and the dark area is more or less confined between the calli in most specimens, as described for O. maculatus, or extending somewhat diffusely over the posterior pronotal lobe. All specimens, however, show a more or less distinct longitudinal median groove on sternites III and IV, the pedicel is similar in length to the distiflagellomere and, the ratio body length-rostral length agrees to those found by Rouault et al. (2005) for O. depressus. We measured 20 males and 20 females from different localities and host, and we found a ratio from 0.72 to 0.90 in males (with the higher values from males collected on C. sempervirens), and a ratio from 0.67 to 0.83 in females. Additionally, we measured ten fifth-instar nymphs whose R:Bd ratio varied between 0.67 and 0.93, within the set of measurements recorded for O. depressus (Rouault et al., 2005: Fig. 7)
The species is recorded from Buenos Aires, La Pampa, Neuquén, and Río Negro provinces in Argentina (Fig. 2). These records represent the first occurrence of this genus and species in the New World and Southern Hemisphere.
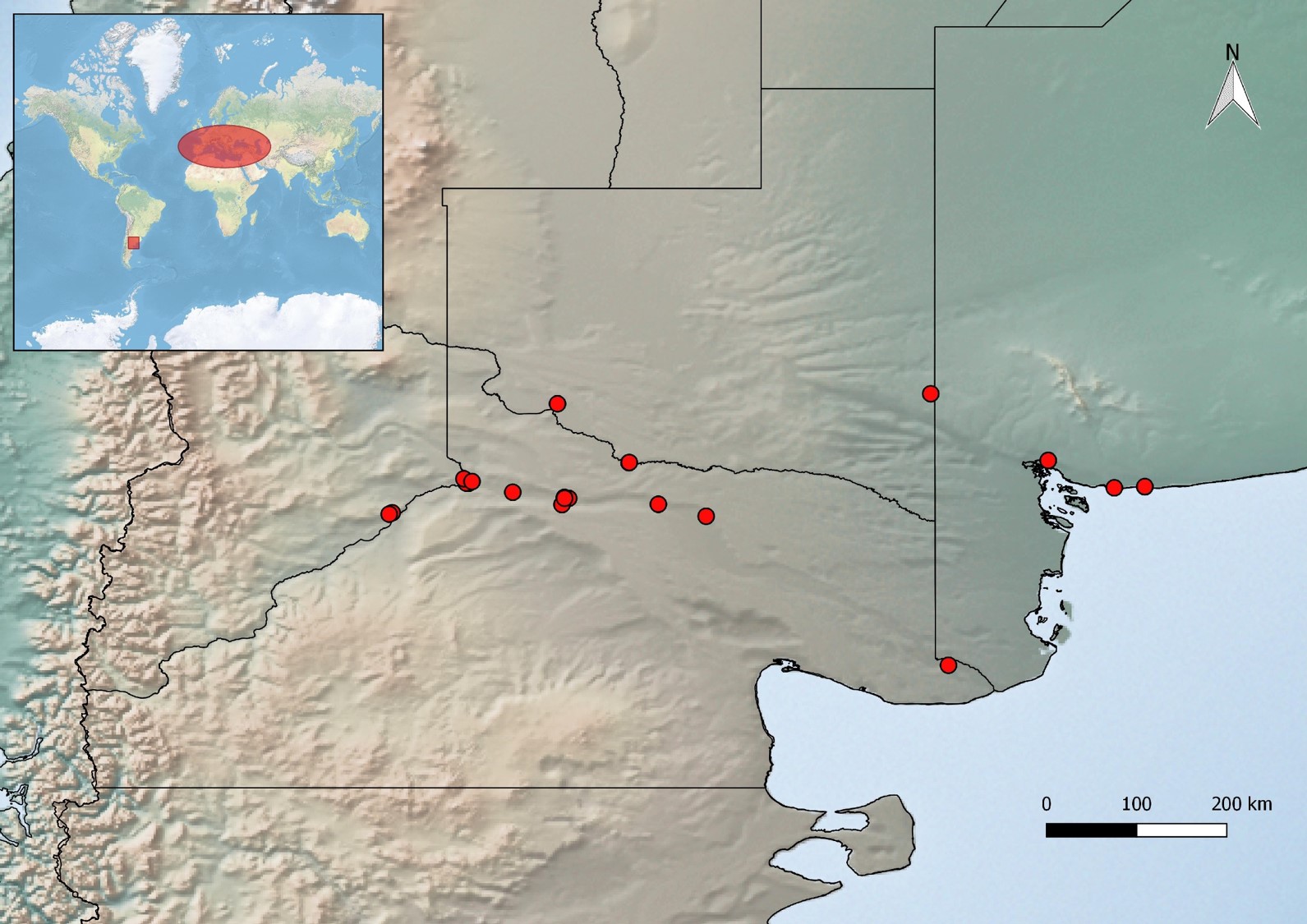
Fig. 2.
Geographical distribution of Orsillus depressus: sites where was collected (red dots).
Top left, area of origin in the Mediterranean basin and new distribution area in South America.
Of the 25 sites sampled, O. depressus was found in 18 (72%), which reveals its wide distribution in the study area (Table I). In addition, immature stages were observed in all these sites, indicating that this species is actively reproducing. No emergence holes or adults of the hymenopteran M. wachtli were found in the observed cones, nor were parasitoids obtained from the eggs.
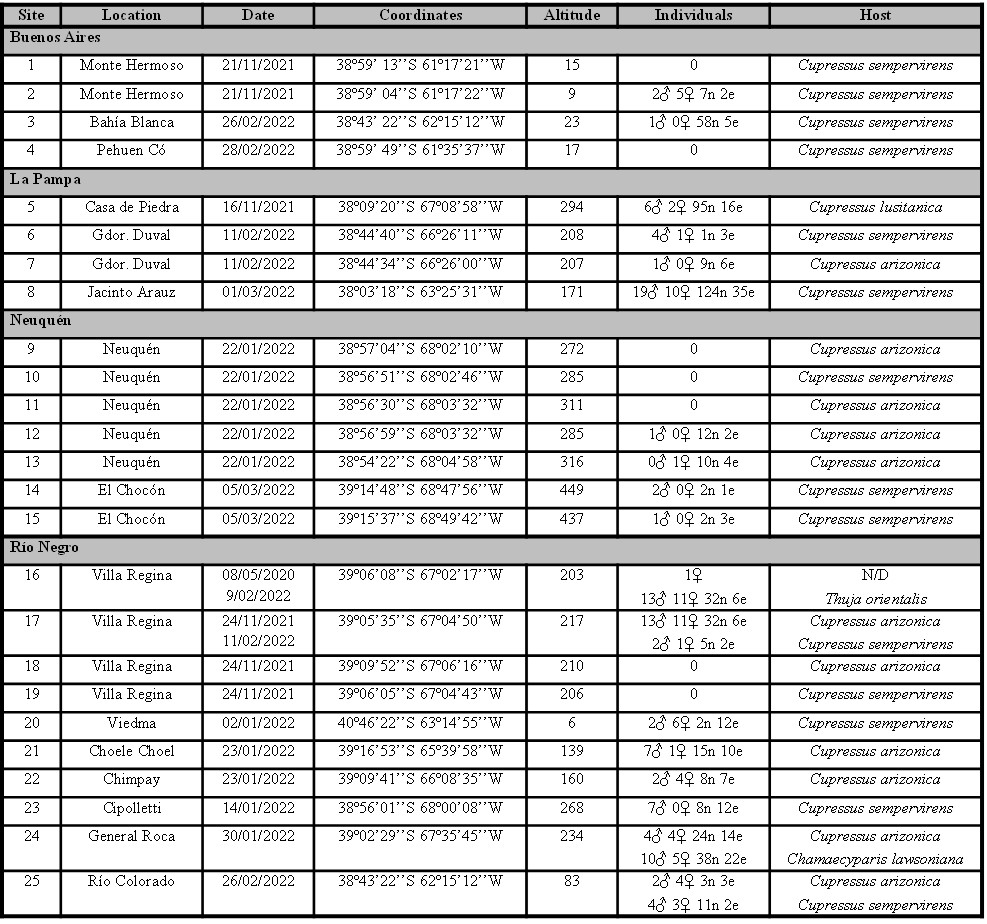
Table I.
Sites sampled in each province.
Indicated: location, date (dd/mm/yyyy), geographic coordinates, altitude (meters above sea level), number of males (♂), females (♀), nymphs (n), egg masses (e) present in each sample and host species.
Unlike what was reported in Europe where O. depressus spreads through the native vegetation, the arrival and dispersion of this species in Argentina requires human assistance in commerce to extend its range. Orsillus depressus can be considered as beneficial when feeding on the seeds of invasive exotic Cupressaceae. However, its feeding habits and ability to act as a vector of cryptogamic diseases reverse its role and makes it a serious threat to native cupressaceous forests considered vulnerable or endangered by the IUCN (2022).
We thank Thomas J. Henry (Systematic Entomology Laboratory, ARS, USDA c/o National Museum of Natural History (NMNH), Washington, DC), who kindly reviewed the manuscript and offered comments for its improvement.
pdellape@fcnym.unlp.edu.ar

Fig. 1.
Orsillus depressus female.

Fig. 2.
Geographical distribution of Orsillus depressus: sites where was collected (red dots).
Top left, area of origin in the Mediterranean basin and new distribution area in South America.

Table I.
Sites sampled in each province.
Indicated: location, date (dd/mm/yyyy), geographic coordinates, altitude (meters above sea level), number of males (♂), females (♀), nymphs (n), egg masses (e) present in each sample and host species.
